Previous Issues Volume 1, Issue 1 - 2016
Pulsatile but not Tonic Secretion of Oxytocin Plays the Role of Anti-Precancerous Lesions of the Mammary Glands in Rat Dams Separated from the Pups during Lactation
Xiaoyu Liu1, Shuwei Jia1, Ying Zhang1, Yu-Feng Wang1
1Department of Physiology, School of Basic Medical Sciences, Harbin Medical University, Harbin, China.
Corresponding Author: Yu-Feng Wang, Department of Physiology, School of Basic Medical Sciences, Harbin Medical University, 157 Baojian Rd, Nangang Dist, Harbin, Heilongjiang 150086, China, Tel: +86-451-86674538; E-Mail: [email protected] Received Date: 01 Feb 2016 Accepted Date: 23 Feb 2016 Published Date: 14 Mar 2016 Copyright © 2016 Yu-Feng Wang Citation: Xiaoyu Liu, Shuwei Jia, Ying Zhang and Yu- Feng Wang. (2016). Pulsatile but not Tonic Secretion of Oxytocin Plays the Role of Anti-Precancerous Lesions of the Mammary Glands in Rat Dams Separated from the Pups during Lactation. Mathews J Neurol. 1(1): 002.
ABSTRACT
Lactation interruption can increase maternal stress and breast tumorigenesis; disorders in hypothalamic oxytocin(OXT)- secreting system and lactation failure could account for these effects. The question is whether abnormal activity of the OXT -secreting system is causally related to precancerous lesions of maternal mammary glands that could underlie breast tumorigenesis. To answer this question, we observed effects of lactation interruption on maternal behaviors, milk availability, OXT neuronal excitability and the histological features of mammary glands of lactating rats that were intermittently separated from the pups. Separation of lactating dams from pups for four days with four hour consecutive contacts per day caused losses of maternal interests in the offspring and lactation failure. The separation also caused early involution of the mammary glands. By simulating OXT release during suckling in isolated rat mammary glands, we determined that the pulsatile pattern of OXT release was the most effective way to suppress hydroxide peroxide-induced expressions of phosphorylated extracellular signal-regulated protein kinase 1/2 and cyclooxygenase 2, two proliferative biomarkers. Dam-pup separation also disrupted the electrical activity of OXT neurons in the supraoptic nucleus and their responses to excitatory stimuli in brain slices. These findings indicate that lactation failure results from lack of pulsatile OXT release during suckling, which is causally related to a series of maternal mental disorders and precancerous lesions of the mammary glands.
KEYWORDS
Lactation; Mammary Glands; Oxytocin; Postpartum Depression; Supraoptic Nucleus.
ABBREVIATIONS
Cox-2, cyclooxygenase 2; IMP, intramammary pressure; L-I, lactation interruption; MER, milk-ejection reflex; OXT, oxytocin; OXTR, oxytocin receptor; pERK, phosphorylated extracellular signal-regulated protein kinase; SON, supraoptic nucleus.
INTRODUCTION
Breastfeeding has many beneficial effects on mothers and the babies [1]. Lactation failure is associated with the high incidence of breast cancer, type 2 diabetes and obesity in the mothers and autism, sudden death, and deficiency in maternal behaviors in the babies [2]. Lactation failure is also associated with increased incidence of postpartum depression and premenopausal breast cancer [3-5]. Successful lactation is based on coordinated actions of milk production, secretion and ejection controlled by a series of hormonal events. These hormones synchronize the development of mammary glands and hypothalamic plasticity to meet requirements of lactation [6].
Milk ejections are the most sensitive event in the lactation and are achieved through the milk-ejection reflex (MER) [7]. During suckling, afferent inputs from nipples are integrated in the brain into intermittent activation of hypothalamic oxytocin (OXT) neurons in forms of burst firing [8,9]. Synchronized bursts among OXT neurons cause release enough OXT to trigger a milk-ejection from the mammary glands. OXT neuronal activity is directly modulated by various cellular components and neurochemical around OXT neurons. During lactation, the supraoptic nucleus (SON) undergoes significant plastic changes which form the basis of burst discharges[10].
Somatodendritic release of OXT is another dramatic feature in the SON, which plays permissive and auto-regulatory roles in neurochemical modulation of OXT neuronal activity [11- 13]. OXT receptors (OXTRs) are localized on both neurons and astrocytes in the supraoptic nucleus (SON) [14]. By activation of OXTRs, OXT can change the morphology and functions of supraoptic cells by activation of Gaq/11 type of G protein coupled receptors and by mobilization of a series of cellular signals, e.g., cyclooxygenase 2 (Cox-2) and phosphorylated extracellular signal-regulated protein kinase (pERK) 1/2 [14- 17]. Moreover, the findings that mother-baby separation can decrease blood OXT levels and OXT has anxiolytic effects suggest the involvement of the OXT-secreting system in the occurrence of lactation failure[18,19]. However, it remains a question about the critical loci in the MER pathway that are responsible for lactation failure. Moreover, it is not answered whether and how lactation failure is causally related to the breast tumorigenesis.
Lactation failure is associated with many factors, such as mother-baby separation, lacking social supports, obesity, babies' sickness, poor breast conditions, cesarean section, mental disorders, and early usages of bottle feeding and milk substitutes, etc. Among them, mother-baby separation is the common cause of lactation failure [20-27]. Thus, to establish causal relationship between lactation failure and postpartum health issues in women and to clarify neural mechanisms underlying lactation failure-associated breast cancer, we used a maternal separation model to observe effects of lactation interruption (L-I) on maternal behaviors, OXT neuronal excitability and proliferative activity of mammary glands of lactating rats. Lastly we examined different patterns of OXT applications on the expression of pERK 1/2 and Cox-2 of the mammary glands in response to oxidative stress in vitro. The preliminary results have been published in an abstract form [28].
MATERIALS AND METHODS
All procedures in these experiments were in accordance with the guidelines on the use and care of laboratory animals set by NIH and approved by the Institutional Animal Care and Use Committees of the University of California, Riverside, and Harbin Medical University, respectively.
Preparation of L-I Rats Primiparous Sprague-Dawley rats were used during lactation as described previously with minor modification[29]. Within 24 h of parturition, litters were adjusted to 10 pups. Dams had 4 h contacts per day with their pups, beginning on lactation day 8-9; separated pups were nursed by foster mothers at all other times. On postpartum day 11-12, observations were made of the following: maternal behaviors (e.g., pup retrieval, ano-genital licking, and suckling), signs of anxiety and depression, litter and dam body weight gains, the latency and duration of MER. This model was also used in observations of firing activity of OXT neurons in whole animals and in brain slices, measurement of intramammary pressure (IMP), and examination of functional proteins as described in the followings.
Western Blots Mammary glands and brain tissues from the SON were sampled. For examining effects of the separation on mammary gland development and OXT modulation of the responsive features of mammary glands to oxidative stress, primiparous rats were used on postpartum day 11-12 (i.e., separation day 4). Methods for in vitro preparation and incubation of rat mammary glands are modified from a previous report [30]. In brief, mammary glands were removed immediately after decapitation. Mammary tissues were dissected in icecold Krebs-Ringer solution under visual observation through a stereomicroscope. In the L-I rats, the tissues, each piece weighing 3-5 mg, were directly put in a tissue lysis buffer, and proteins were extracted and assayed. The lysis buffer was composed of 50 mMTris-HCl (PH 7.4), 150 mMNaCl, and 1% Triton X-100 and protease inhibitors. In weaning rats, the tissues with same size and loci were assigned to different groups as described in the Results and maintained for 1 h at 37°C in 30 ml of the solution. The Krebs-Ringer solution contained (in mM): 141 NaCl, 5.6 KCl, 3.0 CaCl2, 1.4 KH2PO4, 1.4 MgSO4, 24.6 NaHCO3, 11 glucose, pH 7.4, equilibrated with 95% O2-5% CO2 At the end of treatments, the tissues were transferred into the lysis buffer to obtain proteins. The methods for extracting proteins from the SON have been reported in details [16,17]. To run Western blots, 60 μg of protein were separated on 10% SDS-PAGE gels and transferred onto PVDF membranes. Protein membranes were pretreated with 5% milk solids for 1 h at room temperature, and then incubated with antibodies against target proteins as described in the Results. Loading controls were set using antibodies against actin (rabbit, 1: 500) or total ERK2 (rabbit antibody 1: 1000, 1 h at room temperature). Bands were visualized using horseradish peroxidase-conjugated secondary antibodies and an enhanced chemiluminencence system (GE Healthcare).
Immunohistochemistry
In different animal models, morphology of mammary glands and immunostaining of the SON were observed. While we collected samples for Western blots, mammary gland tissues were also sampled and then fixed with 4% paraformaldehyde for 24 h. After cryosection (10 μm-thick), conventional Hematoxylin and Eosin staining was carried out. Images of the sections were taken using a Nikon microscopy and stored in computer for further analysis. To reduce the variability of results from different loci, sections of equivalent location in the mammary glands from different groups were used for comparisons.
In vivo Electrophysiology and Measurement of IMP
In studying OXT neuronal activity in different types of rats, lactating rats were used with or without separation for 4 days. The methods were a modification of that used previously [31]. Dams were separated overnight from a litter of 10 pups but one, and anesthetized with urethane (1.2 g/ kg body weight, i.p.) on the following morning. Animals were placed in a stereotaxic frame according to a rat brain atlas, and body temperature was maintained at 36.5-38°C [32]. Venous cannulation was made of a femoral vein. To measure IMP, a cannula inserted into an inguinal mammary teat was connected to an electromagnetic pressure transducer and IMP changes were recorded in on a computer via an AD/DA board. After exposure of the dorsal surface of the cortex, a stimulating electrode was implanted into the neural stalk/ posterior pituitary, via the dorsal approach, and fixed on the skull with dental cement around pre-anchored screws. Three hours after the operation, extracellular recordings were made from the SON neurons. A glass micropipette (tip diameter, 2-3 μm; 4-6 MO) filled with a 2.5% Pontamine Sky Blue solution in 0.5 M sodium acetate was lowered into the SON. To facilitate burst generation, subcutaneous injection of xylazine (tranquillizing drug and a2-adrenoceptor agonist, 0.2 mg/kg body weight) was applied 20 min before suckling tests. Antidromically-identified SON neurons that showed burst firing patterns followed by ejection of milk were classified as OXT neurons. If no burst or milk ejection occurred, continuous firing pattern and excitatory responses to application of cholecystokinin octapeptide (CCK; 20 μg/kg, i.v.) were taken to judge the chemical nature of neurons in the SON [33]. Electrical signals from the SON were collected through a conventional amplifier and the digitized analog signals were recorded on a 16-channel data acquisition system. After recordings, the stimulation sites were marked by passing positive current through the metal electrode (10- 50 μA for 10-15 min). The last recording site was marked with an iontophoretic deposit of Pontamine Sky Blue dye (-20 μA, continuous current for 15 min). Correct loci were then verified by conventional histology and microscopy.
In vitro Electrophysiology
Following 4 day dam-pup separation, brain slices and patchclamp recordings were performed as previously described [34]. In brief, coronal brain slices containing the SON were cut after decapitation. Whole cell patch-clamp recordings were obtained from magnocellular neurons under visual guidance through an upright microscope during perfusion of artificial cerebrospinal fluid. The artificial cerebrospinal fluid contained (in mM): 126 NaCl, 3 KCl, 1.3 MgSO4, 2.4 CaCl2, 1.3 NaH2PO4, 26 NaHCO3, 10 Glucose, 0.2 ascorbic acid, pH 7.4 and was gassed with 95% O2/5% CO2. Patch-pipette filling solution contained the following (in mM): 145 K-gluconate, 10 KCl, 1MgCl2, 10 HEPES, 1 EGTA, 0.01 CaCl2, 2 Mg-ATP, 0.5 Na2-GTP, pH 7.3, adjusted with KOH. To further identify the chemical nature of recorded neurons, 0.05% Lucifer Yellow (K+ salt) was added to the pipette solution to mark the patched neurons. An Axopatch 200B amplifier (Molecular Devices) was used for collecting electrical signals that were filtered and sampled at 5 kHz by Clampex 10 software through an analog-to-digital converter (Molecular Devices). Data were stored in a computer for offline analysis.
Drugs, Antibodies and Agents Used
All drugs were from Sigma (St. Louis, MO) except as otherwise noted. All primary antibodies were products of Santa Cruz Biotechnology, Inc. (Santa Cruz, CA). All reagents for Western blots were from GE Healthcare (Piscataway, NJ).
Data Collection and Analysis
Data analyses of Western blots and patch-clamp recordings have been described in previous reports [16,17]. Lactation failure was defined as a reduction in milk availability of more than 80% of the control per unit time (one hour). To evaluate maternal behaviors, dams were observed throughout the period of dam-pup separation. Signs of anxiety were evaluated by increases in frequencies of ambulation and rearing; signs of depression mainly include loss of interests in the pups and maternal body weight gain [35]. The interests of dams to pups were shown by the latency and frequency of licking pups after whole litter retrieval. ANOVA, paired or non-paired t test, or Wilcoxon rank test and ?2 test were used for statistical analyses where available as instructed by SigmaStat program (SPSS, Chicago, IL), and p < 0.05 was considered significant. All measures were expressed as mean ± SEM, except as otherwise indicated in results.
RESULTS
Effects of L-I on the MER and Maternal Behaviors
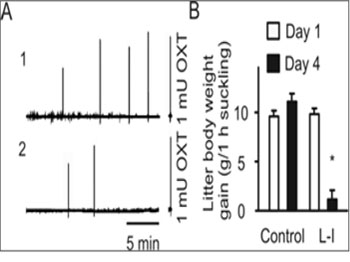
Maternal separation from the infant is one major cause of lactation failure and maternal mental disorders; therefore, we adopted a rat experimental model of dam-pup separation to study effects of L-I on the MER and maternal behaviors[29]. Compared to the MER of the controls (n = 8), the first MER of the L-I rats (n = 8) occurred after a longer latency (10- 24 min, versus 6-10 min in control) and milk ejections stopped earlier (30-45 min versus > 1 h in control). In two pairs of rats from the two groups, IMP was measured (see also the IMP recordings at 7 rats during in vivo recordings of OXT neuron activity). The MER in the L-I rat was obviously weaker than that in the control rat as shown in the magnitude and frequency of changes in the IMP (Figure 1).
Figure 1: Effects of lactation interruption (L-I) on the milk-ejection reflex. A. Traces indicating changes in the intramammary pressure (IMP) observed at day 4 of the control rat (A1) versus that at day 4 of the L-I dam (A2). Note that the right insets/axes indicate the IMP responses to 1 mU OXT (i.v.) as a biologic assay of the relative amount of OXT released in response to suckling stimulation. B. Bar graph summarizing the effect of L-I on milk availability to the litters. *, P < 0.05 compared to day 1.
However, injection of OXT (1 mU, i.v.) in L-I rats still caused an increase in the IMP and stretch responses of pups, similar to that in normally lactating dams, indicating that the failure in the MER was not occurring at the level of mammary glands. Correspondingly, body weight gains in the eight litters were significantly lower than those before the separation and of the control rats. These results indicate that dampup separation can significantly reduce milk availability by reducing OXT secretion but not significantly interrupting the contractile capacity of myoepithelial cells. In addition, milk was still available from the mammary glands.
During the separation, the mother rats exhibited a series of signs of psychiatric disorders. On the first two days, dams showed significantly increases in the frequency of ambulation and rearing, which reached a peak level within the first hour after the separation (6-10 times/min in the first 10 min). On the fourth day of the separation, maternal behaviors could still be observed; however, the dams showed loss of interests in the pups, a typical sign of maternal depression [35]. That is, despite the maintenance of suckling behaviors, the dams spent longer times before starting to lick them. Latency to the first licking after whole litter retrieval and the times of licking during suckling were significantly different from the control dams. Meanwhile, the body weight of those L-I dams was not increased that appeared in the control group. In addition, L-I dams appeared social isolation as shown by lack of interests to male intruders, e.g., less frequent (>80%) approaching, attacking, and ano-genital probing, etc. As a whole, dam-pup separation resulted in a mixed behavior disorder, with signs of anxiety-dominance at beginning and signs of depressiondominance in later stages. Quantitative evaluation of some of these mood/behavior changes is summarized in Table 1. These results indicate that while dam-pup separation can disrupt normal MER, it also evokes maternal depression intermixed with signs of anxiety, which are consistent with previous reports [29,36].
Table 1: Effects of Dam-pup separation on maternal behaviors and milk availability
| Parameters/Groups | Control day 1 | Control day 4 | L-I day 1 | L-I day 4 |
|---|---|---|---|---|
| Latency of licking (min) | 1.38 ± 0.32 | 1.25 ± 0.18 | 1.31 ± 0.21 | 4.88 ± 0.94 |
| Licking times/one hour | 5.1 ± 0.5 | 5.5 ± 0.4 | 5.4 ± 0.3 | 3.5 ± 0.3** |
| Dam's body weight (g) | 356.8 ± 11.2 | 366 ± 10.6** | 364.4 ± 13.5 | 359 ± 14.8 |
| Litter's weight gain (g) | 10.1 ± 0.46 | 11.9 ± 0.74 | 10.8 ± 0.53 | 1.2 ± 0.33** |
Note: Statistics are based on 8 litters in each group, *p< 0.05; **P< 0.01 compared to day 1 with two-tailed, paired t-test; L-I, lactation interruption.
Effects of L-I on the Histology of Mammary Glands
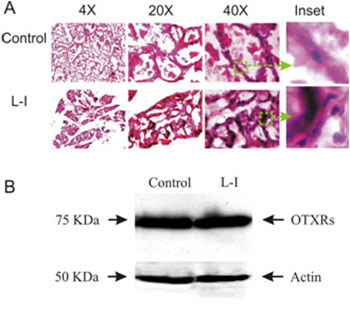
Abnormal OXT production can seriously influence mammary gland development, and form a basis of carcinogen susceptibility [37]. Since normal MER in L-I rats was disrupted, we examined whether dam-pup separation also causes early involution of mammary glands in three pairs of rats. After dissection, part of the mammary tissue was fixed, cryosectioned and Hematoxylin and Eosin stained. Microscopic observation revealed that the mammary glands in normal dams were rich in alveoli, which were much distended with milk. Most lobules were filled with secretory contents after 4 h separation from the pups. The interlobular connective tissue septa are thin and compressed (Figure 2A1). In the mammary glands of L-I dams, alveoli were smaller, whereas the number of body cells surrounding the alveoli and interlobular connective tissue increased significantly ((351 ± 45 % of the control, P < 0.05). In addition, many alveoli were without secretory materials (Figure 2A2). This result indicates that an early involution was evoked in the L-I dams. OXT is the major hormone to maintain mammary gland development during lactation, and which depends on OXTRs in the mammary glands [38]. Thus, changes in OXTR expression could be associated with the early involution of mammary glands. Therefore, in another part of mammary tissue samples, OXTR expression was examined in Western blots (Figure 2B). The results showed no significant (P > 0.05, n = 3) change in OXTR expression in the L-I dams (117.3 ± 8.9% of control). Together with the IMP changes and the restoring effects of exogenous OXT, these results indicate that the reduced milk ejections were due to decreases in the amount and frequency of OXT secretion, but not to changes in OXTR expression or the contractility of the myoepithelial cells in the mammary glands although the storage of milk was decreased obviously.
Figure 2: Effects of the L-I on the histology of mammary glands. A. Exemplary microscopic images of Hematoxylin and Eosin stained cryosections of mammary glands from control (top panel) and L-I rats (bottom panel). From left to right, the images are at 4X, 20X, and 40X magnifications, respectively; insets (green squares and arrows) were enlarged from 40X images. B. Western blots showing OXTR expression (top row) in the control (left) and L-I (right) rats and loading controls with actin (bottom bands).
OXT Effects on Oxidative Stress-Evoked Proliferative Responses in Mammary Glands
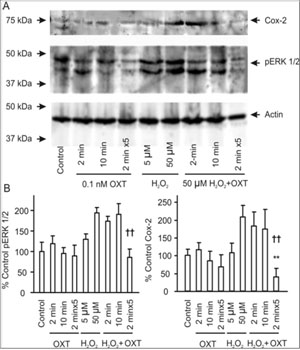
OXT can promote mammary gland differentiation and directly inhibit tumor growth [39]. Since lactation is negatively correlated with breast tumorigenesis, we hypothesized that OXT release during suckling may make unique contributions to the refractoriness of mammary glands to carcinogens[4]. Both pERK 1/2 and Cox-2 have been implicated in early breast tumorigenesis [40,41]. If lactation can reduce mammary tumor susceptibility, OXT should also inhibit carcinogenevoked expression of pERK 1/2 and Cox-2. To test this hypothesis, we examined influences of different patterns of OXT application on the effects of oxidative stress-evoked expression of pERK 1/2 and Cox-2 in mammary glands by applying H2O2. OXT (0.1 nM, 2 min, 10 min, or 2 min for 5 times with intervals of 5 min between two applications) did not significantly influence the expression of pERK 1/2 and Cox-2, while H2O2 (50 µM, 40 min) significantly increased the expression of pERK 1/2 and Cox-2. Simultaneous application of H2O2 with OXT produced different results depending on the patterns of OXT application. It is only the pulsatile application of OXT that significantly reduced H2O2-induced pERK 1/2 and Cox-2 expression (Figure 3). This result clearly indicates that the pulsatile OXT actions rather than tonic OXT actions have strong anti-oxidative effects.
Figure 3:Effects of OXT on hydrogen peroxide (H2O2)-evoked expression of phosphorylated extracellular signal-regulated protein kinase (pERK) 1/2 and cyclooxygenase (Cox-2) in mammary glands from weaning rats. A. Exemplary bands of Western blots showing Cox- 2, pERK 1/2 protein, and loading control (from top to the bottom). From left to the right, the bands show vehicle control, 0.1 nM OXT for 2 min, 10 min and 2 min X 5 with an interval of 5 min between two neighboring OXT applications (pulsatile OXT), 5 µM and 50 µM H2O2 for 40 min, and 50 µM H2O2for 40 min plus OXT for 2 min, 10 min and pulsatile OXT (the last panels), respectively. B. Bar graphs summarizing influences of different patterns of OXT applications on H2O2-evoked pERK 1/2 (left) and Cox-2 (right) expressions. Note that each point represents samples from five rats; *, P < 0.05 and **, P < 0.01 compared to control; ††, P < 0.01 compared to 50 µM H2O2.
Effect of the Dam-Pup Separation on the Firing Activity of OXT Neurons in vivo and in vitro
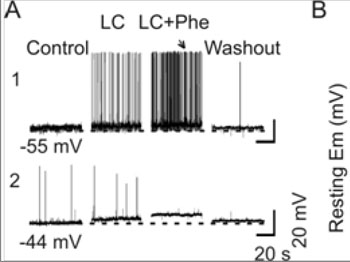
Examination of changes in the MER and mammary gland developments revealed that the lactation failure in L-I rats is likely due to the failure of OXT secretion but not its actions. OXT secretion from the posterior pituitary is directly controlled by burst firing in OXT neurons [42]. Thus, we examined the potential link of the L-I on the lactation. As shown in our preliminary study, in 7 L-I rats, none of 18 putative OXT neurons was elicited typical milk-ejection bursts although 2 of the 7 rats showed weak but detectable milk ejections (i.e., 0.2-0.6 mU OXT) during 1 h suckling[28]. In contrast, in 19 nonseparated lactating rats, 62 OXT neurons showed 197 typical bursts in our previous observations [34]. Statistically, there is significant difference between L-I and non-separated rats in the ratio of burst firing (Chi-square test, P < 0.001). These findings suggest that the ability of OXT neurons to discharge burst was reduced significantly. Moreover, direct electrical stimulation of the posterior pituitary in burst frequency (50 Hz, 0.2 mA, 0.5 s for 4 s) could still evoke full-sized milk ejections (~1 mU OXT) in 4/4 dams tested, indicating that the suppression of MER in L-I rats occurred at the levels of OXT neurons or their presynaptic neurons but not at the posterior pituitary or mammary glands. The suppression of burst firing in OXT neurons could result from malfunctions in OXT neurons themselves or from persistent inhibitory inputs from stress-associated brain structures. Because in stress responses, corticoids can inhibit OXT neuronal activity by increasing GABAergic inputs while reducing glutamatergic neurotransmission [43]. To identify whether the malfunctions in OXT-secreting system is at OXT neurons or remote modulating structures, the burst capacity of OXT neurons was further observed in brain slices from L-I rats. Phenylephrine (1 µM, 5 min, three times with intervals of 15 min washout) was applied in the low Ca2+ (0.7 mM) artificial cerebrospinal fluid as previously described [34]. In normal lactating rats, phenylephrine further increased the firing rate in OXT neurons on the basis of low Ca2+ excitatory actions, and evoked burst firing in 38.9% (14 of 36) OXT neurons as detailed in previous experiments [34].
Figure 4:Effects of L-I on burst firing in OXT neurons. A. Patch-clamp recordings of OXT neuronal activity in brain slices from a control rat (A1) and from an L-I rat (A2) in response to phenylephrine (Phe, 1 µM) in the low Ca2+artificial cerebrospinal fluid (LC, 0.7 mM). Note that the numbers under the traces are the initial membrane potentials (uncorrected for liquid junction potentials of 8-11 mV), which are extended in dashed lines. A burst was evoked in the slice from the control (arrow) but not the L-I rat. B. Bar graphs summarizing influences of L-I on the membrane potential of OXT neurons under resting condition. Note that, *, P < 0.05 compared to day 4 controls.
In contrast, most of OXT neurons (12/16) in the L-I rats had less negative resting membrane potentials (13.4 ± 1.9 mV more depolarized than normal dams, P < 0.01) and lacked spikes. Even in the four neurons showing a few spikes (0.01-0.2 Hz), which was low in amplitude (25-50 mV) and refractory to phenylephrine stimulation. However, a depolarizing response could still be elicited by phenylephrine (Figure 4). These results strongly support the hypothesis that lactation failure results from malfunctions in OXT neurons
DISCUSSION
To set a solid background to investigate neural mechanisms underlying lactation failure-associated maternal health issues, we first evaluated the influence of dam-pup separation on the MER and the maternal behaviors. Through measuring IMP, we identified a critical role of OXT-secreting system in the lactation failure. Moreover, we established the causal link between lactation failure and malignant transformation potentials of mammary glands by simulating the pulsatile release of OXT during suckling and its influence on oxidative stress. Finally, we identified the target of L-I on the MER by electrical recordings of OXT neuronal activity in the SON in vivo and in vitro. These findings allow us to propose that L-I could promote the precancerous lesions of the mammary glands by decreasing the excitability of OXT neurons and the pulsatile release of OXT following suckling stimulation.
Influences of L-I on Maternal Behaviors and the MER
Our result indicates that the lactation failure is due to lack of OXT from OXT-secreting system in the L-I rats since supplying exogenous OXT could evoke milk ejections. By disrupting normal mother-infant interactions, malfunctions of the OXT-secreting system could cause the disorders in maternal behaviors. During lactation, suckling-evoked OXT release is not only essential for the survival of the offspring but also for positive maternal mood [44,45]. OXT can reduce stressinduced response by reducing the adrenocorticotropin and cortisol secretion [45]. However, strong psychiatric impacts like mother-infant separation can still disrupt the "defense system" built on the anxiolytic effects of endogenous OXT, leading to lactation failure and disorders of maternal mood [46]. The maternal stress, anxiety, depression, and even mental disorders during the mother-infant separation can in return worsen the mother-baby relationship [3]. Actually, subcutaneous OXT supplement did increase milk yield and remove signs of anxiety and depression in the L-I rats (our unpublished observation).
Mother-baby separation can reduce afferent inputs from mammary glands, increase retention of milk in the mammary glands, and reduce release of prolactin and OXT that can contribute to maternal depression [18,47]. Mother-baby separation also increases the activity of sympathetic nervous system and hypothalamic-pituitary-adrenal axis that inhibit OXT neuron activity, and thus disrupts the MER [43,48]. Thus, cortisol and sympathetic outflows during the separation stress could contribute to the lactation failure while worsening maternal mood.
Influences of L-I on Mammary Gland Proliferation by Changing OXT Secretion.
In parallel with the behavioral disorders, lactation failure can also increase the susceptibility of mammary glands to carcinogens. Mammary glands in the L-I rats presented increased numbers of undifferentiated epithelial cells but not myoepithelial cells that are markedly resistant to malignant transformation and able to suppress the transformation of neighboring luminal cells [49]. These precancerous lesions could also facilitate breast tumorigenesis. Since OXT plays a dominant role in the generation of the MER the precancerous lesions of the mammary glands could result from lacking OXT during the separation. This proposal is consistent with that involution of the mammary glands after the cessation of sucking is greatly delayed by the administration of OXT to the mother [50]. Since OXTRs on mammary glands and their responses to OXT remain normal, this study provides experimental basis for therapeutic consideration that appropriate supplements of exogenous OXT to keep the mammary glands from tumorigenesis.
OXT can inhibit mammary cell proliferation and tumor growth in mice, rats and MDA-MB231 human breast cancer cell lines [39]. In animals with high mammary tumor incidence, circulating OXT during suckling is low [51,52]. The present study further presented that the intermittent, but not the infrequent (single 2 min) or tonic (10 min) OXT actions dramatically suppressed H2O2-evoked proliferative activity in mammary glands. This finding is the first evidence that directly indicates that suckling-evoked intermittent pulsatile release of OXT plays a key role in the reduction of mammary proliferative activity by lactation.
Mechanisms Underlying the Anti-Precancerous Lesion Effect of OXT
OXT suppression of proliferative activity is likely related to OXTR signaling in the mammary glands. It has been reported that oxidative stress can induce migration of MCF- 7 cells by up-regulation of pERK 1/2 signaling via activating calmodulin kinase II [40,53]. The anti-proliferative effect of OXT is achieved by mobilization of cAMP-protein kinase A [54]. Since cAMP/protein kinase A signaling and pERK 1/2 signaling are mutually antagonistic, OXT may exert anticancer effects by suppressing pERK 1/2 signaling and subsequent tumorigenesis following oxidative stress[15]. Similar to pERK 1/2, Cox-2 as a downstream signal of pERK 1/2, will also be inhibited following the inhibition of pERK 1/2 and reduction of Ca2+ signaling [15,17]. Because the proinflammatory microenvironment within the mammary gland of L-I rats could render an "ideal niche" for oncogenic events, by suppressing the expression of pERK 1/2 and Cox-2, OXT could inhibit the potential mutation by interruption of proliferative signaling cascades involving STAT3 activity, B cell lymphoma 2, caspases and others [55,56]. In human breast cancer tissue, the activity of insulin-regulated aminopeptidase increases significantly, which can decrease oxytocin levels in mammary tissues [57]. Thus, the anti-cancer effect of OXT is turned down due to the oxytocinase hydrolysis of local OXT in the mammary tissues.
Nevertheless, normal lactation will destine the epithelial cells in mammary glands to be resistant to carcinogens during lactation and late stages of female reproduction. The high efficiency of intermittent or pulsatile, rather than tonic, actions of OXT on mammary glands could be due to less desensitization of OXTRs and the evading of circulating OXT from the decomposition of local oxytocinase, which guarantees a more effective result of anti-oxidative stress. The translational potential of this finding is that clinician may consider adopting a therapy with an intermittently delivered, moderate amount, rather than single large dose, of OXT, to restore the MER and mammary gland differentiation in women experiencing lactation failure.
Mechanisms Underlying the Malfunctions of OXT-secreting System
Despite the extensive involvement of other hormones and neural processes, abnormal OXT neuron activity is clearly the major reason of lactation failure. Stimulation of the posterior pituitary or venous supplemental OXT could restore milk ejections immediately, and expression of OXTRs in the mammary glands did not reduce after the separation. Thus, the major disorders should occur at OXT neurons. This proposal is supported by the electrical features of OXT neurons in vivo and in vitro, and by the cellular and molecular changes in the SON that have been presented in our preliminary observations [28]. Mechanistically, abnormal electrical activity of OXT neurons in L-I rats is the primary cause of lactation failure. That suckling failed to excite OXT neurons including the burst firing suggests lack of efficient excitatory inputs or appropriate responses from OXT neurons. Recordings from OXT neurons in brain slices further clarified that, both basal and reactive electrical features were disrupted in L-I rats. Thus, the reduced OXT availability following the separation is due to the failure of OXT neurons to respond to suckling stimulation. The second neurochemical disorder is the uncoupling of OXTR signaling cascade. In L-I rats, OXTRs and their downstream effectors, Gaq/11 subunits, increased significantly while pERK 1/2 decreased significantly. Importantly, molecular associations between OXTRs, Gaq/11 subunits and ERK protein decreased in L-I rats. Thus, external signals could not be delivered into OXT neurons efficiently even the expression of OXTRs and the Gaq/11 subunits increased significantly. Certainly, some other links in the afferent pathway could also be involved in the malfunctioning of the OXT-secreting system, particularly the putative synchronization center in the mammillary body complex, which remain to be investigated[8].
CONCLUSION
The present study revealed that the dam-pup separation can cause maternal depression, lactation failure and precancerous lesions of the mammary glands. Moreover, pulsatile fashion of OXT secretion rather than tonic OXT actions powerfully suppresses oxidative stress-evoked proliferative activity in mammary glands. Lastly, the abnormal activity of OXTsecreting system originates from cellular and molecular disorders in OXT neurons rather than the sensitivity of the OXTRs on the mammary glands. This study, from an integrative view, reveals for the first time that malfunctions of the OXT-secreting system are causally associated with the precancerous lesions of the mammary glands. The present results highlight the importance of a pulsatile pattern of OXT actions in suppressing breast tumorigenesis. Further clarification of the neural mechanisms underlying lactation failure should trigger better therapeutic strategies for lactation failure and prevention of breast cancer.
ACKNOWLEDGEMENT
We thank the advice and supports of late Dr. Glenn I. Hatton. Research was also sponsored by the National Natural Science Foundation of China (grant No. 31471113, YFW), University of California, Riverside, College of Natural and Agricultural Sciences, and the higher education talents funds of Heilongjiang province (grant No. 002000154, YFW).
REFERENCES
- Jonas W, Woodside B. (2016). Physiological mechanisms, behavioral and psychological factors influencing the transfer of milk from mothers to their young. HormBehav. (77), 167- 81.
- Ip S, Chung M, Raman G, Chew P, et al. (2007). Breastfeeding and maternal and infant health outcomes in developed countries. Evid Rep Technol Assess (153), 181-186.
- Orhon FS, Ulukol B and Soykan A. (2007). Postpartum mood disorders and maternal perceptions of infant patterns in wellchild follow-up visits. ActaPaediatrica. 96, 1777-1783.
- Stuebe AM, Willett WC, Xue F and Michels KB. (2009). Lactation and incidence of premenopausal breast cancer: a longitudinal study. Arch Intern Med. 169(15), 1364-1371.
- Anothaisintawee T, Wiratkapun C, Lerdsitthichai P, Kasamesup V, et al. (2013). Risk factors of breast cancer: a systematic review and meta-analysis. Asia Pac J Public Health. 25(5), 368-387.
- Neville MC, McFadden TB and Forsyth I. (2002). Hormonal regulation of mammary differentiation and milk secretion. J Mammary Gland BiolNeoplasia. 7(1), 49-66.
- Hatton GI and Wang Y-F. (2008). Neural mechanisms underlying the milk ejection burst and reflex. In: Neumann ID, Landgraf R, editors. Progress in Brain Research. Nonapeptide Receptors: Regulation and Signalling. 170, 155-166.
- Wang YF, Negoro H and Higuchi T. (2013). Lesions of hypothalamic mammillary body desynchronise milk-ejection bursts of rat bilateral supraoptic oxytocin neurones. J Neuroendocrinol. 25(1), 67-75.
- Lincoln DW and Wakerley JB. (1975). Factors governing the periodic activation of supraoptic and paraventricularneurosecretory cells during suckling in the rat. J Physiol. 250(2), 443-461.
- Hatton GI, Yang QZ and Cobbett P. (1987). Dye coupling among immunocytochemically identified neurons in the supraoptic nucleus: increased incidence in lactating rats. Neuroscience. 21(3), 923-930.
- Moos F, Freund-Mercier MJ, Guerne Y, Guerne JM, et al. (1984). Release of oxytocin and vasopressin by magnocellular nuclei in vitro: specific facilitatory effect of oxytocin on its own release. J Endocrinol. 102(1), 63-72.
- Moos F and Richard P. (1989). Paraventricular and supraoptic bursting oxytocin cells in rat are locally regulated by oxytocin and functionally related. J Physiol. 408, 1-18.
- Wang YF, Ponzio TA and Hatton GI. (2006). Autofeedback effects of progressively rising oxytocin concentrations on supraoptic oxytocin neuronal activity in slices from lactating rats. Am J PhysiolRegulIntegr Comp Physiol. 290(5), R1191- 1198.
- Wang YF and Hatton GI. (2006). Mechanisms underlying oxytocin-induced excitation of supraoptic neurons: prostaglandin mediation of actin polymerization. J Neurophysiol. 95(6), 3933-3947.
- Zhong M, Yang M and Sanborn BM. (2003). Extracellular signal-regulated kinase 1/2 activation by myometrial oxytocin receptor involves Galpha(q)Gbetagamma and epidermal growth factor receptor tyrosine kinase activation. Endocrinology. 144(7), 2947-2956.
- Wang Y-F and Hatton GI. (2007). Dominant role of betagamma subunits of G-proteins in oxytocin-evoked burst firing. Journal of Neuroscience. 27(8), 1902-1912.
- Wang Y-F and Hatton GI.(2007). Interaction of Extracellular Signal-Regulated Protein Kinase 1/2 with Actin Cytoskeleton in Supraoptic Oxytocin Neurons and Astrocytes: Role in Burst Firing. Journal of Neuroscience. 27(50), 13822-13834.
- Tops M, van Peer JM and Korf J. (2007). Individual differences in emotional expressivity predict oxytocin responses to cortisol administration: relevance to breast cancer? Biol Psychol. 75(2), 119-123.
- MezzacappaSE and Endicott J. (2007). Parity mediates the association between infant feeding method and maternal depressive symptoms in the postpartum. Arch WomensMent Health. 10(6), 259-266.
- Trickey H and Newburn M. (2014). Goals, dilemmas and assumptions in infant feeding education and support. Applying theory of constraints thinking tools to develop new priorities for action. Matern Child Nutr. 10(1), 72-91.
- Stuebe AM, Horton BJ, Chetwynd E, Watkins S, et al. (2014). Prevalence and risk factors for early, undesired weaning attributed to lactation dysfunction. J Womens Health (Larchmt). 23(5), 404-412.
- Lawrence RM. (2013). Circumstances when breastfeeding is contraindicated. PediatrClin North Am. 60(1), 295-318.
- Tang L, Lee AH, Qiu L and Binns CW. (2014). Mastitis in Chinese Breastfeeding Mothers: A Prospective Cohort Study. Breastfeed Med. 9(1), 35-38.
- Orun E, Yalcin SS, Madendag Y, Ustunyurt-Eras Z, et al. (2010). Factors associated with breastfeeding initiation time in a Baby-Friendly Hospital. Turk J Pediatr. 52(1), 10-16.
- Figueiredo B, Dias CC, Brandao S, Canario C, et al. (2013). Breastfeeding and postpartum depression: state of the art review. J Pediatr. 89(4), 332-338.
- Jiang H, Li M, Yang D, Wen LM, et al. (2012). Awareness, intention, and needs regarding breastfeeding: findings from first-time mothers in Shanghai, China. Breastfeed Med. 7(6), 526-534.
- Suwalsky JTD, Klein RP, Zaslow MZ, Rabinovich BA, et al. (2006). Dimensions of naturally occurring mother-infant separations during the first year of life. Infant Mental Health Journal. 8(1), 1-18.
- Wang Y-F and Hatton GI. (2009). Oxytocin, lactation and postpartum depression. Frontiers in Neuroscience. 3, 252- 253.
- Lau C and Simpson C. (2004). Animal models for the study of the effect of prolonged stress on lactation in rats. PhysiolBehav. 82(2-3), 193-197.
- Tovar AR, Avila E, DeSantiago S and Torres N. (2000). Characterization of Methylaminoisobutyric Acid Transport by System A in Rat Mammary Gland. Metabolism. 49(7), 873- 879.
- Wang YF, Negoro H and Honda K. (1996). Milk ejection bursts of supraoptic oxytocin neurones during bilateral and unilateral suckling in the rat. J Neuroendocrinol. 8(6), 427- 431.
- Paxinos G and Watson C. (1998). The rat brain in stereotaxic coordinates. Fourth ed. New York: Academic Press.
- Leng G, Dyball RE and Way SA. (1992). Naloxone potentiates the release of oxytocin induced by systemic administration of cholecystokinin without enhancing the electrical activity of supraoptic oxytocin neurones. Exp Brain Res. 88(2), 321-325.
- Wang YF and Hatton GI. (2004). Milk ejection burst-like electrical activity evoked in supraoptic oxytocin neurons in slices from lactating rats. J Neurophysiol. 91(5), 2312-2321.
- Wang D, Noda Y, Tsunekawa H, Zhou Y, et al. (2007). Behavioural and neurochemical features of olfactory bulbectomized rats resembling depression with comorbid anxiety. Behav Brain Res. 178(2), 262-273.
- Gonzalez-Mariscal G, Melo AI, Chirino R, Jimenez P, et al. (1998). Importance of mother/young contact at parturition and across lactation for the expression of maternal behavior in rabbits. DevPsychobiol. 32(2), 101-111.
- Theil PK, Sejrsen K, Hurley WL, Labouriau R, et al. (2006). Role of suckling in regulating cell turnover and onset and maintenance of lactation in individual mammary glands of sows. J Anim Sci.84(7), 1691-1698.
- Zingg HH and Laporte SA. (2003). The oxytocin receptor. Trends EndocrinolMetab. 14(5), 222-227.
- Cassoni P, Sapino A, Papotti M and Bussolati G. (1996). Oxytocin and oxytocin-analogue F314 inhibit cell proliferation and tumor growth of rat and mouse mammary carcinomas. Int J Cancer. 66(6), 817-820.
- Rodriguez-Mora OG, Lahair MM, Evans MJ, Kovacs CJ, et al. (2006). Inhibition of the CaM-kinases augments cell death in response to oxygen radicals and oxygen radical inducing cancer therapies in MCF-7 human breast cancer cells. Cancer BiolTher. 5(8), 1022-1030.
- Salhab M, Singh-Ranger G, Mokbel R, Jouhra F, et al. (2007). Cyclooxygenase-2 mRNA expression correlates with aromatase expression in human breast cancer. J SurgOncol. 96(5), 424-428.
- Wakerley JB, Clarke G and Summerlee AJ. (1994). Milk Ejection and Its Control. The Physiology of Reproduction. New York: Raven Press. 1131-1177.
- Di S and Tasker JG. (2008). Rapid synapse-specific regulation of hypothalamic magnocellular neurons by glucocorticoids. Prog Brain Res. 170, 379-388.
- Wagner KU, Young WS 3rd, Liu X, Ginns EI, et al. (1997). Oxytocin and milk removal are required for post-partum mammary-gland development. Genes Funct. 1(4), 233-244.
- Boutet C, Vercueil L, Schelstraete C, BuffinA, et al. (2006). Oxytocin and maternal stress during the post-partum period. Ann Endocrinol (Paris). 67(3), 214-223.
- Wang P, Yang HP, Tian S, Wang L, et al. (2015). Oxytocinsecreting system: a major part of the neuroendocrine center regulating immunologic activity. Journal of Neurommunology. 289, 152-161.
- Abou-Saleh MT, Ghubash R, Karim L, Krymski M, et al. (1998). Hormonal aspects of postpartum depression. Psychoneuroendocrinology. 23(5), 465-475.
- Sopel M. (2010). The myoepithelial cell: its role in normal mammary glands and breast cancer. Folia Morphol (Warsz). 69(1), 1-14.
- Benson GK and Folley SJ. (1957). The effect of oxytocin on mammary gland involution in the rat. J Endocrinol. 16(2), 189-201.
- Hadsell DL, Bonnette S, George J, Torres D, et al. (2003). Diminished milk synthesis in upstream stimulatory factor 2 null mice is associated with decreased circulating oxytocin and decreased mammary gland expression of eukaryotic initiation factors 4E and 4G. MolEndocrinol. 17(11), 2251- 2267.
- Valdez SR, Penissi AB, Deis RP and Jahn GA. (2007). Hormonal profile and reproductive performance in lactation deficient (OFA hr/hr) and normal (Sprague-Dawley) female rats. Reproduction. 133(4), 827-840.
- Thyagarajan A, Jiang J, Hopf A, Adamec J, et al. (2006). Inhibition of oxidative stress-induced invasiveness of cancer cells by Ganodermalucidum is mediated through the suppression of interleukin-8 secretion. Int J Mol Med. 18(4), 657-664.
- Cassoni P, Sapino A, Fortunati N, Munaron L, et al. (1997). Oxytocin inhibits the proliferation of MDA-MB231 human breast-cancer cells via cyclic adenosine monophosphate and protein kinase A. Int J Cancer. 72(2), 340-344.
- Zaragoza R, Garcia-Trevijano ER, Lluch A, Ribas G, et al. (2015). Involvement of Different networks in mammary gland involution after the pregnancy/lactation cycle: Implications in breast cancer. IUBMB Life. 67(4), 227-238.
- Schuler F, Baumgartner F, Klepsch V, Chamson M, et al. (2016). The BH3-only protein BIM contributes to late-stage involution in the mouse mammary gland. Cell Death Differ. 23(1), 41-51.
- Pilar Carrera M, Ramirez-Exposito MJ, Duenas B, Dolores Mayas M, et al. (2006). Insulin-regulated aminopeptidase/ placental leucilAminopeptidase (IRAP/P-lAP) and angiotensin IV-forming activities are modified in serum of rats with breast cancer induced by N-methyl-nitrosourea. Anticancer Res. 26(2A), 1011-10114.