Previous Issues Volume 1, Issue 2 - 2016
Investigation on Conventional and Cross-linked UHMWPE Artificial Joint Biomaterials Using a BiotriboPOD Method
Jiajia Chen1, Fei Tang1, Zikai Hua1
1School of Mechatronics Engineering and Automation, Shanghai University, China.
Corresponding Author: Zikai Hua, School of Mechatronics Engineering and Automation, Shanghai University, No. 149, Yanchang Rd, 200072 Shanghai, PRChina. Tel: +86 5633 1517; E-Mail: [email protected]
Received Date: 23 Mar 2016 Accepted Date: 27 Apr 2016 Published Date: 09 May 2016
Copyright © 2016 Hua Z
Citation: Chen J, Tang F and Hua Z. (2016). Investigation on Conventional and Cross-linked UHMWPE Artificial Joint Biomaterials Using a BiotriboPOD. Method. Mathews J Orthop. 1(2): 008.
ABSTRACT
Wear has been recognized as the key factor affecting the long-term performance of artificial joints such as hip, knee and spine prostheses. In this paper, the wear of ultra-high molecular weight polyethylene (UHMWPE) and cross-linked UHMWPE counter with cobalt chromium alloy was investigated using a new BiotriboPOD-Multi wear testing apparatus for orthopedic implant materials. Via this apparatus, an effective testing method for the wear evaluation of orthopedic implants can be achieved.
KEYWORDS
Orthopedic Implant; Artificial Joints; Wear; UHMWPE; BiotriboPOD.
INTRODUCTION
Osteolysis induced by ultra-high molecular weight polyeth- ylene (UHMWPE) wear particles is regarded as an important factor in the long-term durability of total joint arthroplasty [1- 3]. Up to 100,000 micron sized UHMWPE wear particles are released per footstep. Such wear particles can activate the immune system as well as be deposited into the surrounding tissue, which results in the lifetime of the most artificial joints limited to about 10-15 years [4-5]. Therefore, the investiga- tion on the wear performance of UHMWPE material is one of the important research areas in artificial joints. Since material selection and component design are two essential factors in the wear performance and durability of orthopedic implants, both biomaterial and product wear test are needed.
Because the tribological property of orthopedic biomaterials has significant influence on the wear performance of implants, the bench wear evaluation of orthopedic biomaterials is re- garded as another important testing methodology. A proper bench wear test can be used to develop an understanding of wear mechanisms and the influence of environmental, design, and material parameters on wear behavior [6-9]. Moreover, due to the low productivity, complex structure and high cost of joint simulator wear testing, a reliable and effective wear testing methodology for orthopedic biomaterials is expected and required.
In the tribological evaluation of orthopedic implants, POD testing methods have also been applied. However, unfortu- nately, the conventioncal POD test devices with unidirectional relative motions between a pin and a disk could not reproduce the wear factors obtained in clinic. For example, the wear fac- tors obtained for the most common combination, ultra high molecular weight polyethylene (UHMWPE) against polished CoCrMo alloy in bovine serum are of the order of 10-8 mm3/ Nm whereas the true clinical wear factors measured from re- trieved acetabular cups of artificial hip joints are of the order of 10-6 mm3 /Nm [10].
According to the study on the joint movements in human ac- tivities, researchers have observed a directional dependence on the wear rate of orthopedic biomaterials [11-12]. Thus, the multi-directional motion has been regarded as fundamental importance in laboratory wear tests for orthopedic biomateri- als. Following this rule, studies on the development of new POD devices for orthopedic biomaterials are being carried out. Saikko developed a high-capacity hip wear simulator of the circular translation pin-on-disk type, call super-CTPOD, which could provide 100 separated test stations and repro- duced the wear of artificial hip joints in vitro. The present au- thors designed a single-station POD apparatus, and validation test results show the wear factor agreed with those obtained in clinic [13, 14].
In this study, the wear performance of conventional ultra high molecular polyethylene (UHMWPE) and cross-linked UHM- WPE were investigated by a new BiotriboPOD-Multi method in a long-term testing duration. Wear factors were calculated and compared with clinical results.
MATERIAL AND METHODS
BiotriboPOD-Multi Description
This 36-station BiotriboPOD-Multi consisted of a frame mod- ule, a connecting screw, a loading module, a cylinder fixed plate, a motion module, a connecting flange and a driving module (Figure.1). The BiotriboPOD-Multi device has 36 sepa- rated test stations. The connecting block and the motion mod- ule are connected by the machine frame module. The loading module is fixed on the frame module.
Figure 1: BiotriboPOD-Multi device designed and built up for the wear test of orthopedic biomaterials.
Experimental Procedure
The pins were made of conventional ultra high molecular polyethylene (UHMWPE) and cross-linked UHMWPE. The di- ameter of the pins was 10 mm, but the initial diameter of the wear face was chambered to 3 mm with an average surface roughness of 0.1 μm.The disks were made of CoCrMo alloy, which were 40 mm in diameter and 10 mm in thickness. The wear surfaces of the disks were machined, ground and dia- mond-polished. The surface roughness was between 0.01 and 0.02 μm, measured by a diamond stylus profilemeter. All the tests were done at room temperature which was 20ºC on the average. All the samples were pre-cleaned with medical alcohol, and then ultrasonically cleaned for 30 min. The angular velocity of the disk was 6.28 rad/s (corresponding a frequency of 1 Hz), resulting as sliding distance of 31.4 mm per cycle and a con- stant sliding speed of 31.4 mm/s, which is between the mean and the maximum sliding speeds during a human walking cycle [15]. The nominal contact pressure was 1 MPa. The lu- bricant used in the experiments was Alpha Calf Fraction serum by Gibco Co. Ltd. And the lubricant was diluted 1:1 with Milli- Q-grade distilled water, and filtered through a 0.1 μm filter paper according to the requirements on the fluid test medium in ISO 1424X standards [16-18]. Two tests were performed by different wear couples and a to- tal 2 million cycles was run for each test. Every five hundred thousand cycles the specimens were weighed for gravimet- ric wear using a Satorius TG328B balance with a resolution of 0.01 mg. During a weigh stop, the specimen and lubricant chambers were carefully cleaned. After the weighing, the specimens were reassembled, and the test was continued with fresh lubricant.
RESULTS AND DISCUSSION
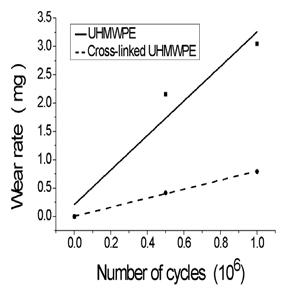
After 2 million cycles the BiotriboPOD-Multi could run smooth- ly. Wear rates observed in tests were linear fitted and sum- marized in Figure 2. The wear rate of cross-linked UHMWPE was lower than the conventional UHMWPE. The slopes of the linear regression were 0.817 mg/106 of cross-linked UHMPWE and 1.92 mg/106 of UHMWPE.
Figure 2: Mean gravimetric wear rate (weight loss) of pins with number of cycles.
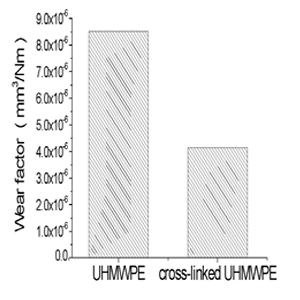
Figure 3: Wear factors of the UHMWPE and cross-linked UHMWPE speci- mens.
The apparatus remained in good operation condition after 2 million testing cycles. Since this testing method is developed and modified upon the standard POD wear tests, it still re- serves the advantages on quickly and reliably identifying those low-friction, low-wear materials for which the more expen- sive and time-consuming joint simulator testing is justified. In addition, this pin-on-disk test can be used to relate wear to material parameters such as polymer molecular weight or counterface surface finish, on a more practical basis than is possible in joint simulator tests. In retrieval analysis of artificial joints, the clinical wear factors are of the order of 10-6 mm3/Nm [19]. In this study, the wear factors of UHMWPE (8.53x10-6 mm3/Nm) and cross-linked UHMWPE (4.16x10-6 mm3/Nm (cross-linked UHMWPE) (Figure 3), which are within the range of clinical wear factors. This present results also confirmed the importance of multi-direc- tional motion in the wear simulation of orthopedic biomateri- als, whereas the wear factor of UHMWPE pins obtained in uni- directional wear tests were of the order of 10-8 mm3/Nm [20]. UHMWPE is the material of choice for total joint arthroplasty bearings. Radiation cross-linking is used to reduce the wear of UHMWPE, which has been associated with peri-prosthetic osteolysis in the long-term. Cross-linking is achieved by the recombination of free radicals induced by irradiation on the polyethylene chains in the amorphous phase [21]. The dependence of wear rates of all the tested specimens on the number of cycles is highly linear (Figure 2), which means that by using this testing methodology a well prediction of the wear can be achieved. In wear testing of orthopedic bioma- terials, an important requirement of a suitable method for a reliable investigation or validation has to be the ability to distinguish between design concepts and allow for a direct comparison of predicate biomaterials. The results obtained in the validation tests indicate that the difference in the tri- bological properties of different orthopedic biomaterials can be distinguished in BiotriboPOD-multi, even between conven- tional and modified biomaterials, i.e. UHMWPE. Moreover, the results show that the conventional UHMWPE pins pro- duce the higher wear than the cross-linked UHMWPE speci- mens. These results agree with the simulator testing results, which show the wear resistance of cross-linked UHMWPE will be enhanced and increased with decreasing radiation dosage, especially in artificial hip joints [22]. The results showed that the BiotriboPOD-multi may become a potential solution to the wear testing of orthopedic biomate- rials. More study should be carried out on the analysis of the wear particles produced in this apparatus.
CONCLUSION
A 36-station BiotriboPOD-Multi apparatus was developed to provide an efficient, reliable and economic for the wear test of UHMWPE. Through the validation tests, the apparatus re- mained in healthy operation condition. The wear factors of UHMWPE was more than twice as cross-linked UHMWPE, which are within the range of clinical wear factors. Wear rates and wear factors obtained in the tests agree with the clinical results. By using the BiotriboPOD-Multi testing methodology, highly linear wear rates can be obtained, and the difference in wear properties of orthopedic biomaterials can be distin- guished.
ACKNOWLEDGMENT
This Study was supported by Orthotek Laboratorty.
REFERENCES
- Amstutz HC, Campbell P, Kossovsky N and Clarke IC. (1992). Mechanism and clinical significance of wear debris-induced osteolysis. Clinical orthopaedics and related research. 276, 7-18.
- Harris WH. (1995). The problem is osteolysis. Clinical orthopaedics and related research. 311, 46-53.
- Wang A, Essner A and Schmidig G. (2004). The effects of lubricant composition on in vitro wear testing of polymeric acetabular components. Journal of Biomedical Materials Research Part B. 68(1), 45-52.
- Mcgee MA, Howie DW, Neale SD, Haynes DR et al. (1997). The role of polyethylene wear in joint replacement failure. Proceedings of the Institution of Mechanical Engineers, Part H: Journal of Engineering in Medicine. 211(1), 65-72.
- Lewis G. (1997). Polyethylene wear in total hip and knee arthroplasties. Journal of Biomedical Materials Research. 38(1), 55-75.
- Hua Z, Fan Y and Jin Z. (2014). A biotribo-acoustic testing method for ceramic orthopaedic biomaterials. Tribology International. 71, 1-6.
- Evans HP, Sindle RW, Sharif KJ, Shaw BA et al. (2012).Analysis of Micro-Elastohydrodynamic Lubrication and Prediction of Surface Fatigue Damage in Micropitting Tests on Helical Gears. Journal of Tribology-transactions of The Asme. 135(1).
- Zhang H, Luo JB, Zhou M, Zhang Y, et al. (2013). Biotribological properties at the stem-cement interface lubricatedwith different media. Journal of The Mechanical Behavior of Biomedical Materials. 20, 209-216.
- Hakala T, Laaksonen P, Saikko V, Ahlroos T, et al. (2012). Adhesion and tribological properties of hydrophobin proteins in aqueous lubrication on stainless steel surfaces. RSC Advances. 2(26), 9867-9872.
- Hua Z and Zhang J. (2010). Axiomatic Design of a Multi-Directional Motion Pin on Disk Apparatus for Biotribology Study
- Calonius O and Saikko V. (2003). Analysis of relative motion between femoral head and acetabular cup and advances in computation of the wear factor for the prosthetic hip joint. Acta Polytechnica. 43(4).
- Saikko V. (2014) Effect of shelf versus accelerated aging of UHMWPE on delamination in knee wear simulation. Tribology International. 73, 10-16.
- Saikko V. (2005). A hip wear simulator with 100 test stations. Proceedings of the Institution of Mechanical Engineers, Part H: Journal of Engineering in Medicine. 219(5), 309-318.
- Hua Z, Zhang H, Fan Y and Jin Z. (2014). Development of a BiotriboPOD testing methodology for the wear evaluation of orthopaedic biomaterials. RSC Advances. 4(38), 19987-19991.
- Kadaba MP, Ramakrishnan HK and Wootten ME. (1990). Measurement of lower extremity kinematics during level walking. Journal of Orthopaedic Research. 8(3), 383-392.
- ISO 18192-1, 2008
- ISO 14243-1, 2009.
- ISO 14242-1, 2009.
- Hall RM, Unsworth A, Siney P and Wroblewski BM. (1996). Wear in retrieved Charnley acetabular sockets. Proceedings of the Institution of Mechanical Engineers, Part H: Journal of Engineering in Medicine. 210(3), 197-207.V
- Korduba LA and Wang A. (2011). The effect of cross-shear on the wear of virgin and highly-crosslinked polyethylene. Wear. 271(9), 1220-1223.
- Jasty M and Smith E. (1992). Wear particles of total joint replacements and their role in periprosthetic osteolysis. Current opinion in rheumatology. 4(2), 204-209.
- Oonishi H and Kadoya Y. (2000). Wear of high-dose gamma- irradiated polyethylene in total hip replacements. Journal of Orthopaedic Science. 5(3), 223-8.