Previous Issues Volume 2, Issue 1 - 2017
Flow Cytometric Analysis of Hyperfractionated and Hypofractionated Radiotherapy for Breast Cancer Patients
Hossam Donya1, 2*, Mahmoud Moawad3
1Medical Physics Division, Physics Department, Faculty of Science, King Abdulaziz University, Jeddah 21589, Saudi Arabia.
2Physics Department, Faculty of Science, Menoufia University, Shebin El-Koom 32512, Egypt.
3Department of Surgical Pathology, National Cancer Institute (NCI), Cairo University, Egypt.
Corresponding Author:Hossam Donya, Medical Physics Division, Physics Department, King Abdulaziz University, Jeddah 21589, Saudi Arabia and Physics Department, Faculty of Science, Menoufia University, Shebin El-Koom 32512, Egypt, Tel: +201004164148; E-Mail: [email protected]
Received Date: 19 Dec 2016 Accepted Date: 26 Jan 2017 Published Date: 27 Jan 2017
Copyright © 2017 Donya H
Citation: Donya H and Moawad M. (2017). Flow Cytometric Analysis of Hyperfractionated and Hypofractionated Radiotherapy for Breast Cancer Patients. Mathews J Cancer Sci. 2(1): 010.
ABSTRACT
Our study is considered a continuing effort in radiation therapy treatment of breast cancer. It is concerned a comparison study between the effect of hyperfractionation radiotherapy (a short term course) and hypofractionation radiotherapy (a long term course) of treatment photon doses on breast cancer cells. The comparison is done with refereeing to normal breast tissue measurements using flow cytometric analysis of DNA ploidy studies. Our samples are extracted from normal benign tumors (non-irradiated) and cancer cells in patients pre-irradiated to VARIAN LINAC of 6 MV photon energy at Radiology Department, NCI. S-phase is extracted from flow cytometric analysis that presents a sophisticated comprehension on the rate of growth of breast tumor cells. It is showed a low S-phase fraction less than 7 % in hyperfractionation radiotherapy, however in hypofractionation radiotherapy showed a high S-phase fraction great than 10%. Accordingly, our study reveals the importance of hyperfractionation radiotherapy for breast cancer treatment, its capabilities to kill larger DNA of tumor cells and restrict its populations.
KEYWORDS
Hyperfractionation Radiotherapy; Hypofractionation Radiotherapy; Flow Cytometric Analysis; S-phase Fraction.
ABBREVIATIONS
HFRT: Hyperfractionation Radiotherapy; HRT: Hypofractionation Radiotherapy; RT: Radiation Therapy; IMRT: Intensity-Modulated RT; LINAC: Linear Accelerator; Flow cytometric: (FCM); SPF: S-phase Fraction; FFPE: Formalin Fixed Paraffin Embedded; PI: Propidium Iodide; DI: DNA index; NCI: National Cancer Institute; HDRBT: High Dose Rate Brachytherapy
INTRODUCTION
Breast cancer in some countries becomes a major public health issue because of its cause of mobility and mortality among women. It is the most prevalent cancer type in Arabian countries, as an example it represent at least 33% and 24% of total cancers in Egypt and Saudi Arabia, respectively. The characteristic of the DNA of tumor cells is an essential tool in cancer treating. The treatment by fractionated doses in radiotherapy showed a repopulation of some surviving malignant cells between the fractions [1-3]. FCM analysis is used for DNA cells analysis that count and sort cells and bookmark it for detection. It is based on real time fluorescence detection for DNA cells which exposed to different laser frequ cies. It al lows many instant physical and chemical parameters analysis for thousands of particles in a very short time. It is achieved by the analysis of fluorescence categorization of cell nuclei of tumor samples under study. Thus, there are some interesting phases which appears in the cell population, for example G1/G0 phase which has less DNA per cell in contrast to a Sphase (cells percentage that in phase with cycle of cell of DNA producing) or even in G2/M phase before cell division since the DNA got doubled during S-phase [4]. Photon treatment radiation effects on tumor cell cycle cell and distribution arresting is different than chemotherapy which has a variable effect depending on the administered drug, which may lead to partial synchronization (change cellular redistribution). Nowadays, modern technology of radiotherapy treatments are directed to high dose gradient delivery in a very short time for high conformal therapy implementation in cancer patients like IMRT and HDRBT. Our study is aimed to study the effect of radiotherapy treatment by introducing two different type of dose delivering a hyperfractionation and hypofractionation radiation therapies to breast cancer patients. The probabilities of killing tumor cells or restriction of its populations in the sensitive phases will be estimated by evaluated SPM. Our study is concerned the influences of a quantitative partial synchronization in tumor cells by different treatment modes that depend strongly on tumor type and kinetics such as length of the cell cycle.
MATERIALS AND METHODS
Materials
A fifty of blocks FFPE cases of highly aggressive carcinoma of cancer breast patients treated with conservative surgery and axillary dissection are obtained from department archives of the of surgical pathology, NCI. Their ages are in the range of 20-65 years old. From each block, three sections 50 μm thick each are obtained for DNA content determent by flow cytometric DNA analysis. Flow cytometric studies on patients are randomly allocated to one of the following groups:
- Ten Patients without radiation benign tumors (fibro adenoma cases).
- In the relative hyperfractionated groups twenty invasive breast cancer patients received short course of radiation cells are pre-irradiated to 6 MV LINAC photon energy with a total dose of 4160 cGy.
- Twenty invasive breast cancer patients received long curse of radiation, exposed to same irradiation facility of 6 MV LINAC photon energy with a total dose of 5000 cGy.
METHODS RT schedule
Three to four weeks after the 6th cycle of chemotherapy, patients started external beam irradiation using 6 MV photon linear accelerators, to the whole breast and ipsilateral lymph nodes (supraclavicular and axillary if it is indicated). The patients are randomized into two three groups: Group A: 10 patients control non recessive radiotherapy, Group B: 20 patients ; received short radiotherapy treatment course (HFRT) course with a total dose of 4160 cGy, 260 cGy/ fraction in 16 fractions in 3 weeks (5 days/week and the last {third} week 6 fractions/week are given to end the total over all time in 3 weeks ), Group C: 20 patients; received conventional long course with a total dose of 5000 cGy, 200 cGy/fraction with an overall time of 5 weeks; 5 days/weeks.
FCM studies
In this study, FCM is used to identify DNA (Ploidy) content in the 50 samples by using Dako-Cytomter System. Standardization of the FCM and initial alignment are performed according to the manufacturer's instructions.
Sample preparation
Solid tumor samples are mechanically dissociated to get a suspension of single cell nuclei and prepared for DNA analysis following the technique of Costa et al. [5]. The specimen, which should be represented, devoid of hemorrhage, necrosis, fat or fibrosis is minced in a clean sterile Petri-dish containing RPMI- 1640 medium using two scalpels or scissors. The cells released in the medium are withdrawn from the Petri dish using clean sterile Pasteur pipette and then filtered twice through a nylon mesh with pore size 53 micron. Later, it are collected in a fifteen milliliter sterile clean conical tube. Sample size of about 1.5-1 cm is considered optimum yielding about 0.5-x106 cells. Centrifugation of the filtered specimen is done at 1200 rpm, continued for 10 min, the supernatant is then discarded and the pellet is re-suspended by gentle tapping on the bottom of the tube. The cells are washed with a fresh medium followed by re-centrifugation is done twice. The pellet is then re-suspended in a citrate buffer solution. Total cell counting is performed prior to staining using a hemocytometer and adjusted to 2-3x10 6 cell/ml.
DNA staining
Staining is performed according to technique of Vindelov et al. [6]. Then, it is washed in phosphate - buffered saline (PBS), nuclear count is adjusted to 2.3x 106 cell/ml. Nuclear staining for DNA content is done using cold PI (Becton Dickinson, USA), which a Fluorochrome that binds to the DNA and emits visible light in the real-time it is stimulated by the laser beam of different frequencies. The tubes are light protected for 10 min, with shaking gently every minute. Sample is incubated in the dark in refrigerator till running in FCM which count your cells and calculated your amount of PI per cell number in both samples (untreated and treated)
Sample running and data acquisition
- At least ten thousands of cells is consistently attained by the FCM at a growth rate of one hundred up to two hundred cells per second for standard lymphocyte in addition to all cases under study.
- The normal lymphocyte suspension is run first to confirm that the diploid G0/G1 peak is at channel number (2N). The peak channel number and corresponding % CV is recorded for G0/G1 only since normal human lymphocytes.
- Sample is run in the FCM and proper gating of the desired population is done to exclude any debris that might be present in the specimen. DNA histogram shows the mean channel number of G0/G1, G2/M, % CV and the ratio of G2/M to G0/G1 which is the mean channel numbers for both diploid and aneuploid populations that is recorded and stored in the list mode.
Interpretation of the DNA histograms
The FL2 channel by a staining with PI shows DNA content since PI will intercalate with DNA. In G0/G1 phase have less DNA per cell in contrast to a cell in S-phase or G2/M phase before cell division since the DNA is got doubled during S phase. Therefore, more PI molecules can intercalate within DNA cells as follows:-
- If it represents a single peak (G0/G1) with a CV = 5%, so the DNA is diploid and whose position is confirmed by running of the lymphocyte sample at first.
- On the other hand, if a Sample is considered "DNA aneuploid" when an additional abnormal peak is presented and it represents at least 10% of the total population collected with an identifiable G2/M peak [6].
- The relative DNA content identified the DNA content of the tumor relative to a normal diploid control. It is obtained by calculating the ratio of the abnormal (aneuploid) G0/G1 peak. This ratio is defined as DI whereas diploid tumors with normality case of DNA content have DI is greater than or equal to 0.95 and less than or equal to1.03, aneuploidy tumors of abnormality case of DNA content have DI greater than 1.03 or less than 0.95.DI ranges followed in this study according to Flyger et al. [7].
RESULTS AND DISCUSSION
Flow cytometric DNA content analysis
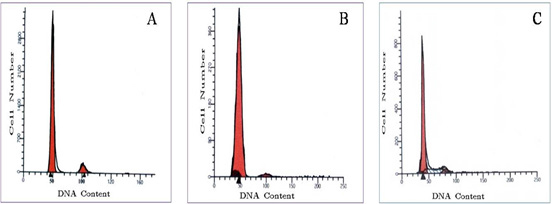
Results of FCM of normal breast tissues, hyper fractionated and hypo fractionated are represented in Fig.1 (a-c) which is histogram of total cell florescence (FL2-area) versus count of DNA cells.
- Normal breast tissue without radiation: All the 10 cases analysed are diploid. Fig. 1a shows a single peak (G0/G1) with a mean channel number at the (2N) position similar to the reference peak.
- Breast cancer patients received short course of radiation (fewer fractions, larger doses per fraction, shorter overall treatment time). All the 20 cases analysed are diploid. They showed diploid peaks at the 2N position. No aneuploidy peaks are detected (Figure. 1b).
- Breast cancer Patients received long curse of radiation (standard radiotherapy)
All the 20 cases analyzed are diploid. They showed diploid peaks at the 2N position. No aneuploid peaks are detected (Figure. 1c).
Figure 1: Histograms of DNA for:- (A) normal breast tissue and two tumors treated by (B) HFRT and (C) HRT recorded by FCM using red excitation optical fluorescence after staining of PI. Ratios of the first (normal tissue) and second (tumor tissue) of the fluorescence peaks.
Larger doses of radiation (hypofractionation) would probably kill larger cell populations in the sensitive phases.
- DNA frequency histogram of normal breast showing a diploid peak, at channel number 50 and with small G2/M cell population at channel number 100 and with low SPF.
- DNA frequency histogram of a case received short course of radiation (HFRT) showing a diploid peak at channel number 200 with a G2/M cell population at channel number 400 and low SPF (less than 7%). Cell cycle time would influence your outcome: breast tumour has a shorter cycle which in term has a lesser degree of synchronization (or 'pile up' in the S-phase) due to high cell turnover, than a tumour with longer cell cycle time.
- DNA frequency histogram of a case received long course of radiation (HRT) showing diploid population with high SPF (higher than 10%).
In hyperfractionation technique, small doses per fraction which in this case 200 cGy/fraction with an cause relatively short cell cycle delay or long cell cycle time, so during the intervals between fractions (e.g. 1 day), many cells can move back from resistant to sensitive cell cycle phases. This conclusion would be added to recent studies that insure the importance of HFRT in intratumoral dose escalation also it appears to be a safe and feasible strategy for patients with some tumors [8].
CONCLUSION
In this study, HFRT is more effective versus HRT for breast cancer patient. HFRT reduces cell cycle redistribution and repopulation. It offers a very short time of cell cycle that influences the outcome DNA Ploidy studies in tumor cells. Hence, it. So, it allows a lesser degree of synchronization that piling up the S-phase. FCM analysis showed low S-phase fraction less than 7 % in HFRT, however, it is showed high SPF great than 10% in HRT. According to this concluded results, our study revealed the importance of HFRT for breast cancer patient which give a positive results in killing larger DNA cell populations that leads to lower growth rate of tumor cells than HRT that may obtain aggressive cancer. It is matched with the recently advanced radiotherapy treatments like IMRT or high dose rate brachytherapy that decreases the total session numbers and high photon doses per fractions. Our concluding remark is that hyperfractionation is advantageous and highly recommended in breast cancer treatment in radiotherapy.
REFERENCES
- Fowler JF. (1986). Potential for increasing the differential response between tumors and normal tissues: can proliferation rate be used? Znt3 Rad Oncol Biol Phys. 12(4), 641-645.
- Trott KR and Kummermehr J. (1985). What is known about tumour proliferation rates to choose between accelerated fractionation and hyperfractionation? Radiother Oncol. 3(1), 1-9.
- Withers HR, Taylor JMG and Maciejewski B. (1988). The hazard of accelerated tumour clonogen repopulation during radiotherapy. Acta Oncl. 27(2), 131-146.
- Perez LA, Dombkowski D, Efird J, Preffer F, et al. (1995). Cell proliferation kinetics in human tumor xenografts measured with iododeoxyuridine labeling and flow cytometry: a study of heterogeneity and a comparison between different methods of calculation and other proliferation measurements. Cancer Res. 55(2), 392-398.
- Costa A, Silvestrini R, Delobino G and Motta R. (1987). Implication of desegregation procedures on biological. Representation of human solid tumors. Cell tissue Kinet. 20, 171-180.
- Flyger H, Larsen J, Nielsen H and Christensen L. (1999). DNA ploidy in colorectal cancer, Heterogeneity within and between tumors and relation to survival. Cytometry. 38(6), 293-300.
- Vindelov J, Christenson I and Nissen N. (1983). Standardization of high resolution FCM-DNA analysis by simultaneous use of chicken and trout RBCs as internal reference standards. Cytometry. 3(5) 328-333.
- Hingorani M, Colley WP, Dixit S and Beavis AM. (2012). Hypofractionated radiotherapy for glioblastoma: strategy for poor-risk patients or hope for the future?. The Brit. J. of Rad. 85(1017), e770-e781.