Previous Issues Volume 4, Issue 1 - 2020
Feline Alveolar Osteitis: Implant Protocol with Osseodensification and Early Crown Placement
Rocco E. Mele1, Gregori M. Kurtzman2*
1VCA Valley Animal Hospital and Emergency Center, Tucson, AZ, USA
2Silver Spring, MD, USA
*Corresponding author: Kurtzman GM, 3801 International Drive Suite 102 Silver Spring, Maryland, USA 20906, Tel: 301-598-3500; E-mail: [email protected] Received Date: November 11, 2020 Published Date: December 31, 2020 Copyrights: Mele RE, et al. © 2020. Citation: Mele RE, et al. (2020). Feline Alveolar Osteitis: Implant Protocol with Osseodensification and Early Crown Placement. Mathews J Vet Sci. (4)1:11.
ABSTRACT
Feline dental implants are becoming a predictable and viable treatment option for the replacement of lost canines due to maxillary Alveolar Osteitis (AO) a painful condition, commonly experienced by a growing number of cats. Surgical extraction and debridement remains the treatment of choice for this complex inflammatory process. However, future complications can be a common sequela of maxillary canine loss. This case will demonstrate the successful surgical extraction of a maxillary canine with implant placement following the osseodensification protocol and utilizing the sockets osteitis buttressing bone formation to promote a positive result with final crown restoration 13 weeks following implant placement.
KEYWORDS: Osseodensification; Alveolar osteitis; Implant; osteitis
INTRODUCTION
Alveolar Osteitis (AO) is a chronic inflammatory process more often diagnosed in maxillary canine sockets of the feline patient. Clinical presentation may include oral pain, bleeding, periodontitis, tooth resorption (ORL), and alveolar buccal bone changes [1-5].
Clinical Features
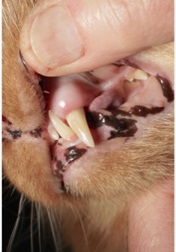
A presumptive diagnosis of (AO) is made on the awake patient, documenting clinical features such as; gingivitis with soft tissue swelling, gingival mucosal erythema, buccal bone expansion, and coronal extrusion (Figure 1).
Figure 1: Alveolar Osteitis (AO) associated with the upper left canine.
Radiographic Features
Radiographic changes are identified under general anesthesia. These bony changes and pathology may include; deep palatal probing (Figure2 red), alveolar bone expansion (Figure 2 green), buttressing condensing bone (Figure 2 blue) and a mottled osseous appearance mimicking rough, large trabeculae (Figure 2 yellow).
Figure 2: Deep probing (red) with osseous expansion of the socket wall (green) and condensing bone (blue) demonstrating a mottled trabeculae pattern in the radiograph (yellow).
Osseodensification (OD):
OD is a novel biomechanical bone preparation technique for dental implant placement to improve bone quality by increasing its density utilizing Densah burs. These burs are rotated in reverse mode (counterclockwise) at 800 to 1500 rpm. Standard osteotomy drills remove and excavate bone during implant site preparation. But the Densah burs allow bone preservation by condensation through compaction autografting during osteotomy preparation thereby increasing the peri-implant bone density (% BV) and the resulting implants mechanical stability [6].
Osseous densification has shown to increase the percentage of bone at the implant surface by increasing the bone mineral density in the peri-implant region. Bone compaction has been shown by numerous studies to improve early implant fixation and better load handling when restored [7-9].
CASE PRESENTATION
A nine year old neutered male DLH feline weighing 5 kg presented for evaluation of “swollen gums”. Initial oral examination noted: (1) alveolar bone expansion with tooth extrusion (#204), (2) gingival erythema and (3) mild tooth mobility (M1). (Figure 1) A preliminary diagnosis of Alveolar Osteitis (AO) was made. A final diagnosis and treatment plan would be presented to the owner at the time of general anesthesia following a complete oral exam.
Surgical Phase:
The morning of the scheduled oral surgical procedure, blood was drawn for a complete blood count (CBC) and a diagnostic profile to evaluate the patients general health. Results were all in the normal range.
Patient was premedicated with Atropine Sulphate (MWI Veterinary Supply, Boise, Idaho) 0.01mg/kg subcutaneously (SQ) and Acepromazine Maleate (MWI Veterinary Supply) 0.02mg/kg SQ. An intravenous catheter was placed and lactated ringers solution was started at a rate of 3ml/kg/h.
General anesthesia was induced by mask with Sevoflurane (Sevo) (MWI Veterinary Supply). The Sevo Vaporizer was set at #7 and O2 flow was set at 1L. Intubation with a cuffed endotracheal tube was completed, and the anesthetic agent was maintained at the vaporizer setting of 3%. O2 flow rate was set at 1L/M. A unilateral left maxillary infraorbital nerve block was administered with 0.5% Bupivacaine (Benco Dental, Tucson, AZ) at 0.1ml at the site and Buprenorphine (MWI Veterinary Supply) at 0.01 mg/kg IV following the General Anesthetic Protocol.
Complete oral examination and digital PA radiographs were obtained and a final diagnosis of Alveolar Osteitis was made. The owner was notified of the results and the treatment options available: 1) Surgical extraction (XSS) with socket debridement. 2) XSS with particulate allograft. 3) XSS with possible immediate implant placement. The felines owner chose to place an immediate implant, if possible and a future restoration following implant healing.
An envelope flap with a distal vertical releasing incision was designed to expose the underlying bone and to make removal of the affected canine (#204) less traumatic and maximize maintenance of the surrounding bone and tissue vascularity will make future implant/restoration more predictable (Figure 3).
Figure 3: Following extraction of the affected canine an envelop flap is elevated to expose the buccal aspect of the extraction socket.
Socket debridement was performed utilizing small curettes and copious lavage with sterile saline with removal of any connective tissue, bacterial contaminants and root/bone fragments ensuring a clean interface between the implant and alveolar bone.
Socket osteotomy was accomplished with an osseodensification drilling protocol known as compaction auto-grafting utilizing Densah Burs (Versah LLC, Jackson Michigan). This compresses the osteotomized alveolar bone increasing its density to yield better primary stability and superior bone-to-implant contact when the implant is inserted (Figure 4). The bur geometry rotating in reverse mode at a rotary speed of 800 to 1500 rpm with profuse saline solution irrigation to prevent bone over-heating, allows compaction of the bone along the inner surface of the implant osteotomy without osseous cutting. The bouncing motion (in and out movement) is helpful to create a rate-dependent stress to produce a rate-dependent strain and allowing saline solution pumping to gently pressurize the boney walls. This combination facilitates an increased bone plasticity and resulting bone expansion [10-13].
Figure 4: Densah bur utilized in the extraction socket to condense the osteotomy improving the bone density (quality) that will house the implant.
A 5.0mm x 8.0mm OCO Engage endosseuos implant (OCO Biomedical, Albuqerque, NM) was secured to a 4/5mm internal hex driver and attached to a Mont Blanc 20:1 implant handpiece. The implant motor (Aseptico, Woodinville, WA) was set at 45 Ncm with a speed of 20 rpm. The implant was introduced into the condensed site to a depth of 4/5mm subcrestal at which point the handpiece torqued out at 45 Ncm (Figure 5). The evolution of Implant Stability Quotient (ISQ) values to access implant secondary stability demonstrated statistically significant correlation with implant outcome. In fact, no implant with > 60 failed, while 19% of implants ISQ < 60 failed [14]. The implant was tested for Implant Stability Quotient (ISQ) using the Osstell IDX unit (Osstell USA, Baltimore, MD) read 65.
Figure 5: Implant is placement is performed utilizing the surgical handpiece into the prepared osteotomy.
A cover screw was inserted into the implant to prevent graft material from entering the implants connect at the platform. A condenser was utilized to pack an allograft (Veterinary Transplant Service (VTS), Kent, WA) into the gap between the implant and socket walls at the crest. The graft material not only fills the “jump gap” between the implant and bone but occupies the tissue zone to the height of the free gingival margin (FGM). The graft material is incorporated into the tissue zone, acting as a scaffold to support the ridge contour profile and peri-implant tissue [15] (Figure 6). The cover screw is carefully removed, so as not to disturb the graft and replaced with a 4/5mm flat healing abutment (OCO Biomedical) with a height slightly taller than the thickness of the soft tissue so that a single stage approach. This acts as a prosthetic seal and is tightened with finger pressure utlizing a hand held hex driver. (Figure 7). The flap margins were reapproximated around the healing abutment and fixated with Securocryl, a 5/0 synthetic suture material (Securos Surgical, Fiskdale, MA) in an interrupted manner (Figure 8). Post-Op radiographs were taken to evaluate the implant/abutment interface and graft placement (Figure 9). Recovery was uneventful and the patient was discharged with post-surgical instructions to the animals owner on the same day as surgery. Cefovecin Sodium was administered SQ at a dose of 0.045 ml/lb (Zoetis,Inc., Kalamazoo, MI) and Buprenorphine (MWI Veterinary Supply, Boise, ID) at a dose of 0.01mg/kg orally every 12 hours for five days.
Figure 6: Osseous graft material is placed into the gap between the placed implant and the wall of the extraction socket prior to flap closure.
Figure 7: A healing abutment is placed in to the implant prior to flap closure with a height equal to the soft tissue thickness to achieve a single stage surgical approach.
Figure 8: The soft tissue flap is approximated to achieve primary closure around the healing abutment and secured with interrupted sutures.
Figure 9: Radiograph at immediate placement (left) and following healing abutment placement with graft placement (right) at the surgical appointment.
Restorative Phase
Because of the owners busy schedule, re-evaluation was limited to e-mail photos of the surgical site at three weeks and eight weeks post-operatively (Figure 10 and 11). The implant site continued to display minimal inflammation with no evidence of mucositis and/or peri implantitis.
Figure 10: Clinical appearance at 3 weeks post-surgical at suture removal demonstrating marginal gingival inflammation.

Figure 11: Clinical appearance at 8 week post-surgical demonstrating non-inflamed gingival tissue.
At 10 weeks post implant placement, the patient returned for intra-oral dental radiographs and a final Implant Stability Quotient (ISQ) value (Osstell USA, Columbia, Maryland) to assess osseointegration for a possible early restoration phase. The same feline general anesthetic protocol (GAP) for the surgical phase was repeated for the restorative phase. Dental radiographs were taken demonstrating excellent stability and osseointegration with an ISQ value of 74 recorded. ISQ value is an objective world standard for measuring implants stability.
The healing abutment (HA) was removed and a 5mm diameter impression coping (OCO Biomedical) was attached to the implant (Figure 12). Rostral maxillary and mandibular impressions and bite registry were obtained with Vinyl Polysiloxane (VPS) (Benco Dental) (Figure 13). The impression coping was then detached intraorally and replaced with a new 5mm diameter HA repositioned into the implant to maintain the tissue emergence profile (Figure 14) and a final radiograph was taken (Figure15).
Figure 12: Impression coping placed into the implant at the restorative phase following removal of the healing abutment.
Figure 13: Vinyl polysiloxane (VPS) impressions of the arch being treated (bottom) and opposing arch (middle).
Figure 14: Clinical appearance at 10 weeks post-surgical demonstrating healthy non-inflamed gingival tissue around the healing abutment.
Figure 15: Radiograph at 10 weeks post-surgical demonstrating incorporation of the graft placed with the patients bone and lack of radiolucency between the implant and socket walls.
It is crucial to maintain healthy thick keratinized tissue (KT) around the implant for long term implant and bone stability. This KT tissue minimizes bone resorption and inflammation around the prosthetics under function [17,18].
Customized Digital Workflow for Veterinary Prosthetics
• Data Acquisitions (Stone Models / 3Shape Scanner)
• CAD (3 Shape Dental Design Software)
• CAM (Substractive Manufacturing-Milling / Sintering)
• Final Veneering (Stain / Glaze)
The impressions are sent to the dental lab (Dental Prosthetics, Tucson, AZ) and a stone model was created with an implant analog within the model and scanned utilizing a 3 Shape D2000 scanner (3 Shape Inc., Warren, NJ) to create a virtual model (Figures 16 and 17) Final components were designed incorporating 3 Shape software and CAD CAM milled (Figures 18 and 19).
Figure 16: Virtual models with the restorations prosthetic design with emergence of the screw that will retain the restoration to the implant (yellow).
Figure 17: Virtual models buccal view with the restorations prosthetic design with emergence of the screw that will retain the restoration to the implant (yellow).
Figure 18: Cad/Cam milled wax crown on the mounted models to verify relationship with the opposing arch.
Figure 19: Cad/Cam milled zirconia crown on the mounted models demonstrating the screw access hole in the crown.
Three weeks after dental impressions were taken, the patient returned for delivery of the final prosthetic components, a screw-retained zirconia crown on a custom titanium abutment. The restoration arrived from the laboratory as a one piece restoration with the zirconia crown luted to the titanium abutment that would be screw retained to the implant intraorally (Figure 20). The patient was again anesthetized following the General Anesthetic protocol followed in the two previous procedures. An oral exam and intra oral radiographs were obtained to evaluate the peri-implant soft tissue and the quality of the bone surrounding the implant. (Figure 21). The crown was soaked in a 0.12 % Chlorhexidine Gluconate rinse (Delta Hex Oral Rinse w ZN, MWI Veterinary Products) for two minutes to aid in elimination of oral bacteria into the implant connection during insertion. The restorations external hex was lined up with the implant platforms internal hex to get the correct rotational position and seated into the implant. The restorative fixation screw was inserted into the screw access channel and tightened to 25 Ncm following the manufactures recommendations and verified with a calibrated torque wrench (Figure 22). A piece of Teflon tape was placed into the crowns screw access channel and then filled with a light-cured radiopaque, methacrylate-based flowable composite, PermaFlo (Ultradent Products, Inc. South Jordan, UT) (Figures 23 and 24).
Figure 20: The Cad/Cam screw retained crown off the model demonstrating the screw access hole and connector that will engage the connector in the implants platform to prevent rotation of the restoration on the implant under function.
Figure 21: Radiograph at 13 weeks post-surgery with the screw retained restoration fixated to the implant.
Figure 22: The screw retained restoration intraorally following torque of the fixation screw to secure the restoration to the implant.
Figure 23: Composite has been placed to seal the restoration screw access and complete the esthetic appearance of the restoration.
Figure 24: Finalized implant restoration providing a natural appearance and returning the patient to function.
The nine day and six week re-call appointments display exceptional emergence profile of the restoration with minimal inflammation and good implant stability. The axial contour of the natural tooth or prosthetic crown as it relates to the adjacent soft tissue, as a good emergence profile has been shown improve the effectiveness of oral hygiene near the gingival sulcus limiting potential for recession and marginal inflammation related to food impaction during mastication [19] (Figures 25 - 26). Design of the restoration allows long-term maintenance of the gingival health without inflammation (Figure 27).
Figure 25: Clinical appearance 9 days post insertion of the implant restoration demonstrating non-inflamed gingival tissue.
Figure 26: Restored implant at 9 days post-insertion of the restoration restoring the patient to a natural dentition and occlusion.
Figure 27: Gingival appearance at 5 months post restoration of the maxillary canine demonstrating a continuing lack of inflammation of the soft tissues.
At a four-year routine appointment, the maxillary canine implant/restoration demonstrates excellent tissue and bone stability (Figure 28). Minimal gingival inflammation was noted with bone loss even though the feline patient does not maintain the daily oral hygiene that the human patient would practice.
Figure 28: Four year routine follow-up demonstrating gingival health surrounding the implant and its restoration and an absence of gingival inflammation.
DISCUSSION
Dental implants in the view of many veterinarians, under no circumstances, should ever be placed in dogs and felines for many reasons including lack of formal training programs [20]. The authors would agree that formal and rigorous training is necessary to develop acceptable and predictable outcomes. In contrast other disciplines that are universally accepted, such as, endodontics, orthodontics, and periodontics, are continually utilized to save, not extract, functional teeth in our pets. Ironically, the favored argument is that pets do very well without their teeth.
Many years ago there was very little evidence that endodontic and orthodontic treatment was a viable and predictable option for companion animals. Yet, over the years and with a number of published case reports, it has been accepted as a predictable valuable service that can be offered to pet owners in confidence [21-25]. Until recently there has been no evidence in the literature or any published reports that dental implants have ever been applied in specialized treatment planning in canine and feline dentistry. Because clinical case documentation and short and long term follow up is lacking, many would consider this discipline experimental. However, the principal author has placed 60 implants in 40 feline patients with more than half being restored. All cases have complete documentation with many appearing in multiple publications in Veterinary and Human dental journals [26-28].
Feline Alveolar Osteitis (AO) is a common presenting problem that the veterinary dental clinician has to deal with routinely. Treatment of this complex condition involves a comprehensive oral exam with dental radiographs under general anesthesia. Most cases require surgical extraction of the affected tooth, buccal bone osteoplasty, and extensive debridement before a tension free gingival flap is closed. In this case presentation we explore a paradigm shift in the treatment of AO with surgical extraction, utilizing the alveolar inflammatory changes (bone buttressing) and a osteotomy drilling protocol (osseodensification) to develop an ideal site for immediate implant placement, excellent primary stability and a prosthetic crown restoration 13 weeks following implant placement. This new treatment protocol has proven effective and predictable in over 30 plus cases. Restoration not only provides a normal functioning maxillary canine yet prevents a common complication, lip entrapment, especially in the feline patient.
Customized digital planning is now being utilized in all our implant crown restoration cases compared to older techniques utlizing direct wax-ups. With digital planning, the dental laboratory creates a soft tissue model and subsequent castings employing advanced computer-aided design. The computer aided process produces more precise restorative results than the traditional methods of the past.
Detailed post-op instructions for the long-term survival of the implant/crown restoration are discussed and sent home with the owner in the departing instructions. Softer food only (no dry food), no toys and gentle cleaning of the prosthesis with soft moist swabs daily for 2/4 weeks. Pain medication and antibiotics are dispensed on case by case evaluation.
CONCLUSION
Utilizing the AO inflammatory socket modifications combined with an innovative osteotomy technique (osseodensification) we can optimize the surgical site by maintenance of the surrounding bone, soft tissue, and implant stability thus preventing future collapse within the buccal plate. Immediate implant /restoration can be a manageable approach to restore the feline patient to full function and preclude the serious common complication of felinemaxillary canine extraction, lip-entrapment. Extraction and immediate implant placement with early loading utilizing the new osteotomy protocol (Densah Drilling) provides a predictable treatment for this common feline dental condition.
REFERENCES
- Lewis JR, Okuda A, Shofer FS, Pachtinger G, Harvey CE, et al. (2008). Significant association between tooth extrusion and tooth resorption in domestic cats. J Vet Dent. 25(2):86-95.
- Newman MG, Takei HH, Klokkevold PR. (2015). Carranza’s Clinical Periodontology, 12 th ed. St. Louis Missouri: Sanders. 875.
- Beebe DE, Gengler WR. (2007). Osseous surgery to augment treatment of chronic periodontitis of canine teeth in a cat. J Vet Dent. 24:30-38.
- D’Astous J. (2015). Periodontology: An overview of alveolar bone expansion. Can Vet J. 56(3):295-300.
- Bell CM, Soukup JW. (2015). Histological, Clinical, and Radiographic Findings of Alveolar bone expansion and osteomyelitis in the jaws of cats. Veterinary pathology. 52(5):910-8.
- Huwais S, Meyer EG. (2016). A Novel Osseous Densification approach in Implant Osteotomy Preparation to increase Biomechanical Primary Stability, Bone Mineral Density, and Bone-to- Implant contact. J Oral Maxillofac Implants. 32(1):27-36. doi: 10. 11607/JOMI. 4817.
- Summers KB. (1994). A New concept in Maxillary implant surgery: The osteotome technique. Compendium. 15:152,154-162.
- Green JR, Nemzek JA, Arnoczky SP, Johnson LL, Balas MS. (1994). The effect of bone composition on early fixation of porous-coated implants. J Arthroplasty. 14:91-97.
- Kold S, Rihbek O, Vester M, Overgaard S, Soballe K. (2005). Bone compaction enhances fixation of weight bearing titanium implants. Clin orthop Relat Res. (431):138-144.
- Trisi P, Bernardini M, Falco A, Podaliri M. (2016). New Osseodensification implant site preparation method to increase bone density in low-density bone. In Vitro evaluation in sheep. 25(1).
- Perren SM, Huggler A, Russenberger M. (1969). The reaction of cortical bone to compression. ACTA OrthopScand Suppl. 125:19-29.
- Trisi P, Todisco M, Comsolo U, Travaglini D. (2011). High versus low implant torque: A histological, histomorphometric, and bio medical study in sheep mandible. Int J Oral Maxillofac Implants. 26(4):837-849.
- Huwais S, Meyer EG. (2017). A Novel osseous densification approaching implant osteotomy preparation to increase bio mechanical primary stability, bone mineral density and bone-to- implant contact. Int J Oral Maxillofac Implants. 32(1):27-36.
- Rodrigo D, Aracil L, Martin C, Sans M. (2010). Diagnosis of implant stability and its impact on implant survival: A prospective case series study. Clin. Oral Impl Res. 21:255-261.
- Chu SJ, Salama MA, Salama H, Garber D, Saito H, et al. (2012). The dual-zone therapeutic concept of managing immediate implant placement and provisional restoration in anterior extractionug. 33(7).
- Baltayan S, Anfruns JP, Aghaloo T, Moy P. (2016). The predictive value of resonance frequency analysis in surgical placement And loading endosseous implants. J Oral Maxillofac Surgery. 74:1145-1152.
- Rieder C. (1996). Customized implant abutment copings to achieve biologic mechanical and esthetic objectives. Int J. Periodont Restorative Dent. 16(1):20-29.
- Chiche FA, Leriche MA. (1998). Multi-diciplinary implant dentistry for the improved aesthetics and function. Pract Periodontics Aesthetics Dent. 10(2):177-186.
- Croll BM. (1990). Emergence profile in natural tooth contour. Part II: Clinical Considerations. J Prosthet Dent. 63(4):374-9.
- Tannenbaum J, Arzi B, Reiter AM. (2013). The case against the use of dental Implants in dogs and cats. J AM Vet Med Assoc. 243(12):1680-1685.
- Visser C. (1990). Endodontology: Chronic Maxillary Sinus abcessation in the Canine. J Vet Dent. 7(2):12-13.
- Eisner E. (1990). Transcoronal approach for endodontic access to the fourth maxillary premolar in dogs. J Vet Dent. 7(4):22-23.
- Willams C. (1986). Endodontics. Vet Clin North Am Small Anim Pract. 16(5):875-893.
- Visser C. (1991). Coronal access of the canine dentition. J Vet Dent. 8(4):12-16.
- Kuntsi-Vaattovaara H, Verstraete F, Kass P. (2002). Results of root canal treatment in dogs; 127 cases. (1995-2000). J Am Vet Med Assoc. 220(6):775-780.
- Mele RE, Kurtzman GM. (2019). New paradigm shift in Feline Dental Implants in Maxillary Alveolar Osteitis treatment with osseodensification. Spectrum Implants – Spring. 10(1).
- Mele RE, Caiafa A, Kurtzman GM.(2017). Feline Dental Implants: Long term follow - up of two cases. J Vet Dent. 34(4):249-258.
- Mele RE, Caiafa A, Kurtzman GM. (2016). Dental Implants and Incisor Bridge Placement in a Dog. J Vet Dent. 33(4):249-258.
.jpg)


























.jpg)