Previous Issues Volume 3, Issue 1 - 2018
Evaluation of p53, Ki-67 and c-erb-B2 Expression in Normal Oral Epithelium, Dysplastic Epithelium, and Oral Squamous Cell Carcinoma
Banu Ozveri Koyuncu1,Cemal Akay2, Banu Yaman3, Ali Veral4,Sevtap Gunbay5,Tayfun Gunbay5
1Associate Professor, Department of Oral and Maxillofacial Surgery, Faculty of Dentistry, Ege.
2Professor, Department of Oral and Maxillofacial Surgery, Faculty of Dentistry, Ege University, Bornova, Izmir, Turkey.
3Associate Professor, Department of Pathology, Faculty of Medicine, Ege University, Bornova, Izmir, Turkey.
4Professor, Department of Pathology, Faculty of Medicine, Ege University, Bornova, Izmir,Turkey.
5Professor, Department of Oral and Maxillofacial Surgery, Faculty of Dentistry, Ege University, Bornova, Izmir, Turkey.
Corresponding Author: Banu Ozveri Koyuncu, Associate Professor, Department of Oral and Maxillofacial Surgery, Faculty of Dentistry, Ege, Tel: 90-232-3881108; E-Mail: [email protected]
Received Date: 02 Oct 2018 Accepted Date: 09 Oct 2018 Published Date: 11 Oct 2018 Copyright © 2018 Koyuncu BO
Citation: Koyuncu BO, Akay C, Yaman B, Veral A, et al. (2018). Evaluation of p53, Ki-67 and c-erb-B2 expression in normal oral epithelium, dysplastic epithelium, and oral squamous cell carcinoma. Mathews J Dentistry. 3(1): 019.
ABSTRACT
The expression of p53, Ki-67, and c-erb-B2 in normal oral epithelium, dysplastic epithelium, and oral squamous cell carcinoma (OSCC) was investigated with immunohistochemical method and was determined the correlation between them. The study was performed on tissue sections of dysplasia (n= 25), OSCC (n=22). Twenty normal samples of oral epithelium were used as control. Sections were stained with immunohistochemically with antibodies to p53, Ki-67, and c-erb-B2 proteins. The percentage of positive cells was assessed in oral dysplastic, OSSC and normal epithelium. Statistical analysis was done using Kruskal-Wallis, Mann Whitney U, and the chi-square tests. The percentage of p53 and Ki-67 positive cells was evaluated and statistically significant differences were found between normal oral mucosa and dysplasia, and between dysplasia and invasive oral squamous cell carcinoma (p<0.05). C-erb-B2 positive cells increased from normal oral epithelium through dysplasia to OSCC, but it was not statistically important (p>0.05). A significant correlation between p53 and Ki-67 positive cells was observed (p<0.001) but no significant correlation was found between c-erb-B2, p53 and Ki-67 in OSCC (p>0.05). These findings suggested that the expression of p53 and increase of Ki-67 index may play roles in carcinogenesis.
KEYWORDS
Oral Squamous Cell Carcinoma; Dysplasia; Ki-67; p53; c-erb-B2; Immunohistochemical; Carcinogenesis.
INTRODUCTION
Head and neck cancer is the sixth most common type of cancer, representing about 6% of all cases and accounting for an estimated 650 000 new cancer cases and 350 000 cancer deaths worldwide every year [1]. They appear to have a relatively poor prognosis and survival, despite advances in treatment with surgery, radiation, and chemotherapy, and many recent advances in oncology and molecular biology over the last four decades [2].
Oral carcinogenesis is generally considered to be a molecular and histologic multistep process. The histologic features are predominantly caused by alteration of cell kinetics in the proliferative pool of the epithelium, expressed as increased growth fraction and cell division rate. This alteration determines the transformation of normal oral epithelium into a malignant tumor [3].
In recent decades, attention has been focused on handful of biomarkers frequently associated with cancer. Biomarkers of carcinogenesis are quantifiable molecules involved in physiological or pathological events which occur between exposure to exogenous or endogenous carcinogens and the subsequent development of cancer [4]. Biomarkers of early carcinogenesis provide a means of diagnosing very early changes associated with cancer development before actual tumours or even polyps are present. Among the most common biomarkers used to study is p53, a tumor suppressor gene, and the mutation of p53 has been considered to be the most common genetic alteration in head and neck cancers [5]. Another common immunohistochemical marker used to study cell proliferation is the Ki-67 antigen [6]. The c-erbB-2 is a kind of receptor for EGF (epidermal growth factor), a key molecule for growing of carcinoma [7]. Several studies were performed to demonstrate the expression of c-erb-B2 on breast, and endometrial cancer.[8,9] Hovewer, studies regarding expression of c-erb-B2 in head and neck cancers especially in oral mucosa are limited [10,11].
In this study, we aimed to investigate the expression of p53, Ki-67, and c-erb-B2 in normal oral epithelium, dysplasia, and invasive oral squamous cell carcinoma and to determine the correlation between them.
MATERIAL AND METHODS
This study was performed at the Department of Oral and Maxillofacial Surgery, School of Dentistry, and at the Department of Pathology, School of Medicine, in Ege University. The Ethics Committee of the university approved the study protocol. All selected samples had been routinely fixed in 10% neutral formalin (24 to 48 hours), dehydrated in graded alcohols, cleared in xylene, and embedded in paraffin. The hematoxylin-eosin slides were all reviewed, the diagnosis was confirmed by 2 pathologists (A.V. and B.Y.).
Samples of normal (20 cases), dysplastic (26 cases: 13 mild, 8 moderate and 5 severe dysplasia), squamous cell carcinomawell differentiated (G1) (4 cases), squamous cell carcinomamoderately differentiated (G2) (13 cases), and squamous cell carcinoma-poorly differentiated (G3) (5 cases) were included in the study. Normal samples of oral mucosa were obtained from resection margins of benign oral lesions and were used as control.
Epithelial dysplasia was diagnosed according to the criteria and the definition proposed by Pindborg et al [12]: mild dysplasia was considered mild expansion of the proliferative zone, with mitotic activity, hyperchromatism, and nuclear variability; moderate dysplasia indicated these same features were present in up to half of the thickness of the epithelium; and severe dysplasia indicated the atypical proliferative zone encompassed up to three fourths of the epithelium. Squamous cell carcinoma cases were classified into well-differentiated, moderately differentiated, and poorly differentiated using World Health Organization criteria.
Immunohistochemical analysis of p53, Ki-67, and c-erb-B2 p53 (clone SP5, Neomarkers, 1:200 dilution, catalog number: RM-9105-R7), Ki-67 (rabbit monoclonal, Neomarkers, 1:250 dilution, catalog number: RM-9106-R7) and c-erb-B2 (clone Ab-17, Neomarkers, 1:300 dilution, catalog number: MS-730-R7) primary antibodies were investigated in five micron sections from formalin-fixed, paraffin-embedded tissues. Immunohistochemical staining as non-biotinised painting system, HRP multimers based, hydrogen peroxide substrate and 3, 3 '- diaminobenzidin tetrahidroklorit (DAB) containing kromogen (ultraView ™ Universal DAB Detection Kit, Catalog number 760-500, Ventana Medical Systems, Tucson, AZ) with full automatic immunohistochemistry staining machine (Ventana Benchmark XT, Ventana Medical Systems, Tucson, AZ) were used.
Tissue sections taken on electrostatic slides (X-Trat, Surgipath Medical Industries, and Richmond, Illinois, USA) were dried at °C 60, at least two hours. All the mmunohistochemical staining, including deparaffinization and process of antigen retrieval was carried out in the Benchmark XT fully automatic immunohistochemistry staining device. Only the dropping process for the primary antibodies p53, Ki-67 and c-erbB-2 were manually done and were incubated at °C 37, 32 minutes. Previously known breast carcinoma sections were used as positive control for p53, Ki-67 and c-erb-B2, whereas the primary mAb was omitted in negative control sections.
After the sections were rinsed in the same buffer (TBS, three times, 2 min each), streptovidine peroxidase conjugate was added for another 15 min, followed by substrate/chromogen solution, 0.05% 3,3-diaminobenzidine tetrahydrochloride in 0.05M tris at pH 7.6, which forms a dark brown deposit in the presence of the enzyme that is complexed with the antigen. Counterstaining of the nuclei was done with Mayer's hematoxylin.
Nuclear staining for the p53 and Ki-67 and cytoplasmic membrane staining for the c-erb-B2 was considered positive immunohistochemically. It was calculated showing nuclear staining for p53 and Ki-67 cells to the total number of cells ratio. The percentage of p53-positive cells was evaluated from a minimum of 1000 cells in each case. We scored p53 expression as negative (-), meaning that staining was observed in <5% of the tumor cells, or positive (+), meaning that staining was observed in 5% or more of the tumor cells.
In the case of Ki-67, the quantitative labeling index was used. In areas having the highest nuclear labeling density, the percentage of tumor nuclei expressing Ki-67 was determined by counting 400 cells per slide, and the percentage was expressed on the available cells.
Subjective and semicantitative scoring system was used for cerb-B2 using the rate and intensity of staining of tumor cells. Scoring system was based on the ratio of positive staining cells to the other cells and the intensity of staining which were evaluated 1-3 according the optical magnification which was positivity can be selected [13].
STATISTICAL ANALYSIS
For statistical analysis SPSS (Statistical Package for Social Sciences) for Windows 15.0 program was used. Descriptive statistical analysis was performed for each group of lesions. Mann Whitney U, and the chi-square tests were used to evaluate the presence of statistically significant differences. Simple regression analysis was used to evaluate the correlation between features. Analysis showing a confidence interval above 95% (p<0.05) was considered significant
RESULTS
The results of immunohistochemical analysis of p53, Ki-67, and c-erb-B2 expression in normal oral epithelium, dysplastic epithelium, and OSCC are shown in Table 1.
Table 1: Comparison of p53, Ki-67 ve c-erb-B2 in normal, dysplastic and OSSC epithelium.
P0 : Comparison of all the groups
P1 : Comparison of the normal and dysplastic epithelium group
P2 : Comparison of the normal epithelium and OSSC group
P3 : Comparison of the dysplastic epithelium and OSSC group
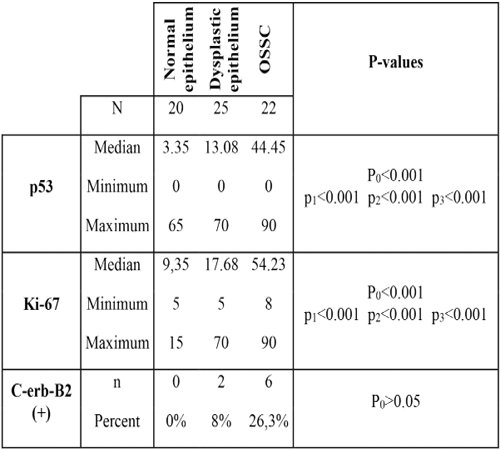
P53 p53 was present in the basal layer in normal oral epithelium; in the basal and parabasal layers in leukoplakia, dysplasia, and carcinoma in situ; and in central and peripheral regions in invasive carcinoma. In normal epithelium, the positivity of p53 was between 0% to 65% (mean 3.35%). In dysplastic group (25 cases), the positivity was between 0% to 70% (mean 13.08%) and in OSSC epithelium the positivity was between 0% to 90% (mean 44.45%) (Figure 1).
Figure 1: The p53 positivity in atypical cells of OSCC (anti-p53x200).
In normal oral epithelium p53 was positive in only 1 (5%) sample. In dysplastic group, 12 (48%) samples were p53 positive. In this group, 5 mild dysplasia samples, 3 moderate dysplasia samples, and 4 severe dysplasia samples had p53 positivity.
In carcinoma group, p53 was positive in 16 (72.73%) samples. In this group, 2 well-differentiated carcinoma samples and 9 moderately differentiated carcinoma samples had a p53 positivity. All poorly differentiated carcinoma samples were also p53 positive.
Statistically, it was found that the p53 positivity increased significantly from normal oral epithelium to dysplastic epithelium and OSSC (P<0.001).
In dysplastic group p53 positivity did not increased from mild dysplasia to moderate and severe dysplasia. In dysplastic group statistically significant differences were only found between moderate dysplasia and severe dysplasia (p<0.05). Statistically significant difference was also found between mild dysplasia and carcinoma group, between moderate dysplasia and carcinoma group.
No statistically significant difference was found between normal oral epithelium and mild dysplasia, between mild dysplasia and moderate dysplasia. No statistically significant difference was also found between severe dysplasia and carcinoma group. Although p53 positivity increased from well differentiated to poorly differentiated invasive carcinomas, the test showed nonstatistically significant differences among them. No statistically significant difference was found in aspect of differentiation in carcinoma group.
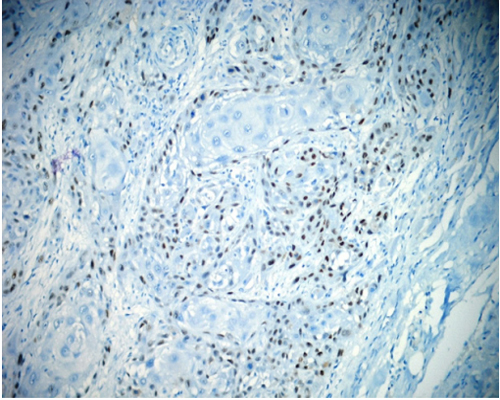
KI-67 When we evaluated the positivity of Ki-67, in normal epithelium, the positivity was between 5 % to 15% (mean 9.35%). In dysplastic epithelium, the positivity was between 5% to 70% (mean 17.68%) and in OSSC epithelium the positivity was between 8% to 90% (mean 54.23%) (Figure 2).
Figure 2: Dense Ki-67 positivity in OSCC (anti-Ki67x200).
Ki-67 positivity increased from normal oral epithelium to dysplasia and OSCC. Statistically significant differences were found between the three groups (P<0.001).
In dysplastic group Ki-67 positivity did not increased from mild dysplasia to moderate and severe dysplasia.
With the Mann-Whitney U test, statistically significant differences were found between moderately differentiated carcinoma and poorly differentiated carcinoma (p<0.01),between severe dysplasia and carcinoma group (p<0.01).
Statistically significant difference was also found between mild dysplasia and carcinoma group, between moderate dysplasia and carcinoma group, and between severe dysplasia and carcinoma group (p<0.01).
No statistically significant difference was found between normal oral epithelium and mild dysplasia, between mild dysplasia and moderate dysplasia, between moderate dysplasia and severe dysplasia, between severe dysplasia and well differentiated carcinoma. No statistically significant difference was found in aspect of differentiation in carcinoma group.
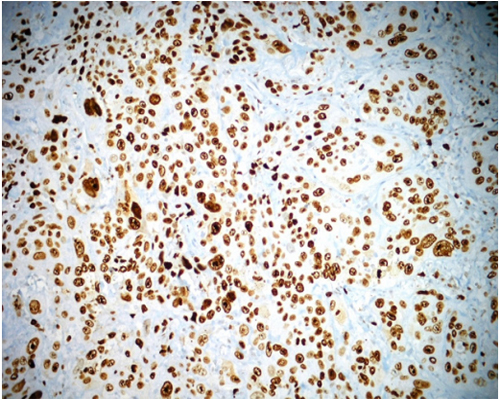
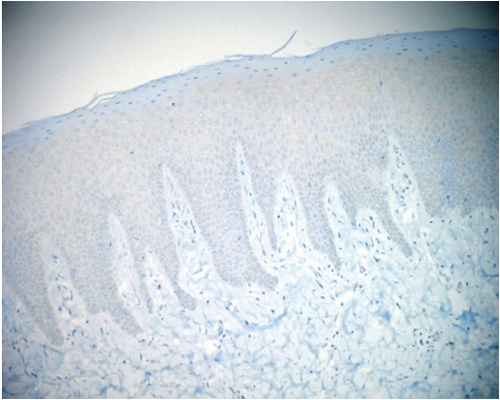
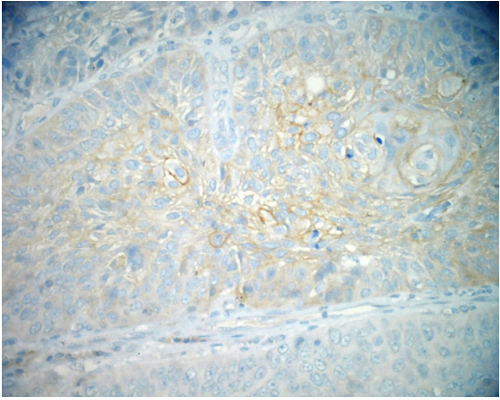
C-ERB-B2 The positivity of c-erb-B2 was 0% in normal epithelium, was 8% in dysplastic epithelium, and was 28% in OSSC epithelium (Figure 3, and Figure 4). C-erb-B2 positivity increased from normal oral epithelium to dysplasia and OSCC, but it was not statistically significant. (P>0.05) (Table 1).
Figure 3: The negativity of c-erb-B2 in normal epithelium. (anti c-erb-B2 x100).
Figure 4: The positivity of c-erb-B2 in neoplastic cells of OSCC (anti c-erbB2x200).
C-erb-B2 positivity did not increased from mild dysplasia to moderate dysplasia and severe dysplasia.
With the chi-square test, statistically significant differences were found between normal oral epithelium, and severe dysplasia (p<0.05), between normal oral epithelium and moderately differentiated carcinoma (p<0.01), between mild dysplasia and moderately differentiated carcinoma (p<0.05).
No statistically significant difference was found between normal oral epithelium and mild dysplasia, between mild dysplasia and moderate dysplasia, between moderate dysplasia and severe dysplasia, severe dysplasia and carcinoma group. No statistically significant difference was found in aspect of differentiation in carcinoma group.
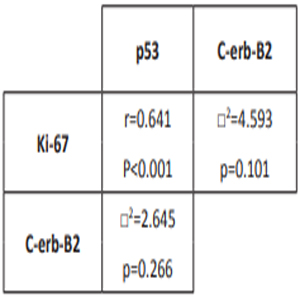
The correlation of p53, Ki-67 and c-erb-B2 A significant positive correlation between p53 and Ki-67 positivity was observed in OSCC (p<0.001) but no significant correlation was determined between c-erb-B2, p53 and Ki-67 (p>0.05) (Table 2).
Table 2: Correlations between p53, Ki-67 ve c-erb-B2 positivity.
DISCUSSION
The development of head and neck squamous cell carcinoma is generally accepted to be a multistep process in which alterations in oncogenes and tumor suppressor genes play an important role. [14] Because genetic changes accumulate during carcinogenesis and precede malignant transformation or invasion in OSCC, it is needed to define markers of cellular or molecular changes that are associated with specific stages of the disease. In recent years identification of such biomarkers has become an important field of research in head and neck cancer [15].
In this study, we aimed to demonstrate the expression of p53, Ki-67 and c-erb-B2 in normal oral epithelium, dysplasia, and oral squamous cell carcinoma and to investigate the relationship between them.
Overexpression of p53 protein is frequently detected in OSSC tissues, even in epithelial dysplasias, by immunohistochemical method. Alterations of the gene p53 have been reported to be an early event in the development of head and neck squamous cell carcinomas and its detection may be considered as a biomarker for these early stages of carcinogenesis [5].
The results of this study showed that the percentage of p53 in normal oral epithelium with a mean value of 3%, in dysplastic epithelium with a mean value of 13%, and in OSSC with a mean value of 45%. These data were in line with several investigators' findings which was reported high frequencies of p53 gene alteration in head and neck cancer and OSSC varying from 34 to 100% [16, 17]. In our study, the results revealed that p53 expression showed statistically significant differences between normal epithelium and dysplasia, and between dysplasia and OSSC. These results were in accordance with the observations of Abbas et al [18]. who showed that statistically significant differences between normal mucosa and severe dysplasia, between normal mucosa and invasive carcinoma, and mild and severe dysplasia. Girod et al [19]. showed that increased proliferative activity in benign, premalignant, and malignant lesions of the oral cavity presumably reflects a series of genetic and cellular alterations. They added that because proliferative activity alone does not necessarily indicate malignant transformation, assessment of p53 status may allow differentiation of reactive or potentially neoplastic proliferation in oral lesions.
Cuevas et al [20]. reported that p53 could be not specific enough to identify patients suffering OED with high risk to malignancy; however, the evaluation of the presence of p16 and in the tumoral invasive front of OSCC could contribute to establish the tumor progression.
Ki-67 shows the proliferation capacity of cancer cells, whose appearance correlates with the presence and severity of the epithelial dysplasia [6]. In our study, we found the percentage of Ki-67 positive cells in normal oral epithelium with a mean value of 9%, in dysplastic epithelium with a mean value of 18%, and in OSSC with a mean value of 54%. In Carlos de Vicente et al.'s study [21], the percentage of Ki-67 positive cells in all tumor specimens ranged between 5 and 85%, with a mean Ki-67 index value of 34%. In this study, the number of Ki-67 positive cells increased significantly in normal, dysplastic epithelium and OSSC. Our results are in accordance with the results reported by Piatelli et al [2]. 2 who found that Ki-67 expression increased with malignancy potential of the lesion in dysplastic epithelium and OSSC. Iamaroon et al [23]. found that p53 and Ki-67 showed a trend to increase with disease progression. In addition, Birajdar et al [24]. mentioned that Ki67 is a reliable proliferative marker which can be used for the diagnosis of oral epithelial dysplasias which have tendency to undergo malignant transformation in their study.
The c-erb-B2 oncoprotein is a transmembrane protein, the presence of which has been associated with poor prognosis in several human neoplasms such as breast, and endometrial cancer [8, 9]. In our study, we found the percentage of c-erbB2 positive cells in normal oral epithelium with a value of 0%, in dysplastic epithelium with a value of 8%, and in OSSC with a value of 28%. An immunohistochemical study on cancer of the head and neck revealed that expression of c-erb-B2 in malignant epithelium was 78% [11]. In another study made by Craven et al [25]. on the same subject, it has been stated that immunoreactivity of c-erb-B2 was 46%. The same authors added that, there was a definite association between immunohistochemical detection of c-erb-B2 and head and neck squamous cell carcinoma, since almost half of the tumour specimens manifested detectable c-erb-B2 protein. However, this association could not be extended to a predicted disease progression or outcome.
According to our findings, c-erb-B2 showed a trend to increase with disease progression but this was not statistically important. In a similar study conducted by Hou et al [26]. it was reported that as the cells acquire a more malignant phenotype, there was a progressive increase in neu/c-erb-B2 expression.
Iamaroon et al [23]. stated that a significant correlation between p53 and Ki-67 positive cells was observed in OSSC. In accordance with these authors, in the presented study the expression of both p53 and Ki-67 was clearly demonstrated in OSSC and was found a positive correlation between these two markers. This finding shows that alterations of p53 lead to increased cell proliferation in OSSC. Contrary to these findings, Kurokawa et al [27]. have reported that no significant correlation was found between p53-labeling index and Ki-67-labeling index, and clinical site or clinical types (normal epithelium and leukoplakia).
In conclusion, we reveal that expression of p53 and Ki-67 increased from normal through dysplastic epithelium to OSCC and suggesting that they may play a critical role in oral carcinogenesis. There was a significant positive correlation between both markers. On the other hand, it was found that c-erb-B2 positive cells tended to increase from normal through dysplastic epithelium to OSCC but this was not statistically important. No significant correlation was found between c-erb-B2, p53 and Ki-67. Further studies are recommended to highlight the significance of c-erb-B2 in oral squamous cell carcinoma.
REFERENCES
- Parkin DM, Bray F, Ferlay J and Pisani P. (2005). Global cancer statistics, 2002. CA Cancer J Clin. 55(2): 74-108.
- Greenlee RT, Murray T, Bolden S and Wingo PA. (2000). Cancer statistics 2000. CA Cancer J Clin. 50(1): 7-33.
- Papadimitrakopoulou VA, Hong WK, Lee JS, Martin JW, et al. (1997). Low dose isotretinoin versus beta carotene to prevent oral carcinogenesis: long term follow-up. J Natl Cancer Inst. 89(3): 257- 258.
- Kellof GJ, Boone CW, Steele VW, Crowell JA, et al. (1994). Progress in cancer chemoprevention: perspectives on agent selection and short-term clinical trials. Cancer Res. 54(7 Suppl): 2015s-2024s.
- Partridge M, Kiguwa S, Emilion G, Pateromichelakis S, et al. (1999). New insights into p53 protein stabilisation in oral squamous cell carcinoma. Oral Oncol. 35(1): 45-55.
- Tumuluri V, Thomas GA and Fraser IS. (2002). Analysis of the Ki-67 antigen at the invasive tumour front of human oral squamous cell carcinoma. J Oral Pathol Med. 31(10): 598-604.
- Mellon JK, Lunec J, Wright C, Horne CH, et al. (1996). CerbB2 in bladder cancer: molecular biology, clinical correlation with epidermal growth factor receptors and prognostic value. J Urol. 155(1): 321-326.
- Ross JS and Fletcher JA. (1999). HER-2/neu (c-erb-B2) gene and protein in breast cancer. Am J Clin Pathol. 112: S53-67.
- Han AC, Hovenden S, Rosenblum NG and Salazar H. (1998). Adenocarcinoma arising in extragonadal endometriosis: an immunohistochemical study. Cancer. 83(6): 1163-1169.
- Shnayder Y, Kuriakose MA, Yee H, Chen FA, et al. (2001). Adhesion molecules as prognostic factors in nasopharyngeal carcinoma. Laryngoscope. 111(10): 1842-1846.
- Silvaa SD, Agostinia M, Nishimotob IN, Colettaa RD, et al. (2004). Expression of fatty acid synthase, ErbB2 and Ki-67 in head and neck squamous cell carcinoma. A clinicopathological study. Oral Oncology. 40(7): 688-696.
- Pindborg JJ, Reichart PA, Smith CJ and van der Waal I. (1997). International typing of cancer and precancer of the oral mucosa. World Health Organization. 2nd ed Berlin Heidelberg: Springer-Verlag.
- Santini J, Formento JL, Francoual M, Milano G, et al. (1991). Characterization, quantification, and potential clinical value of the epidermal growth factor receptor in head and neck squamous cell carcinomas. Head Neck. 13(2): 132-139.
- Van Oijen MG, van de Craats JG and Slootweg PJ. (1999). p53 overexpression in oral mucosa in relation to smoking. J Pathol. 187(4): 469-474.
- Almangush A, Heikkinen I, Makitie AA, Coletta RD, et al. (2017). Prognostic biomarkers for oral tongue squamous cell carcinoma: a systematic review and meta-analysis. Br J Cancer. 117(6): 856-866.
- Langdon JD, Partridge M. (1992). Expression of the tumour suppressor gene p53 in oral cancer. Br J Oral Maxillofac Surg. 30(4): 214-220.
- Ogden GR, Kiddie RA, Lunny DP and Lane DP. (1992). Assessment of p53 protein expression in normal, benign, and malignant oral mucosa. J Pathol. 166(4): 389-394.
- Abbas NF, El-Sharkawy SL, Abbas EA and El-Shaer MA. (2007). Immunohistochemical study of p53 and angiogenesis in benign and preneoplastic oral lesions and oral squamous cell carcinoma. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 103(3): 385-390.
- Girod SC, Pfeiffer P, Ries J and Pape HD. (1998). Proliferative activity and loss of function of tumor suppressor genes as "biomarkers" in diagnosis and prognosis of benign and preneoplastic oral lesions and oral squamous cell carcinoma. Br J Oral Maxillofac Surg. 36(4): 252-360.
- Cuevas Gonzalez JC, Gaitan Cepeda LA, Borges Yanez SA, Cornejo AD, et al. (2016). p53 and p16 in oral epithelial dysplasia and oral squamous cell carcinoma: A study of 208. Indian J Pathol Microbiol. 59(2): 153-158.
- Carlos de Vicente, Herrero-Zapatero A, Fresno MF and Lopez-Arranz JS. (2002). Expression of cyclin D1 and Ki67 in squamous cell carcinoma of the oral cavity: clinicopathological and prognostic significance. Oral Oncol. 38(3): 301-308.
- Piatelli A, Rubini C, Fioroni M, Iezzi G, et al. (2002). Prevalence of p53, bcl-2, and Ki-67 immunoreactivity and of apoptosis in normal oral epithelium and in premalignant and malignant lesions of the oral cavity. J Oral Maxillofac Surg. 60(5): 532-540.
- Iamaroon A, Khemaleelakul U, Pongsiriwet S and Pintong J. (2004). Co-expression of p53 and Ki-67 and lack of EBV expression in oral squamous cell carcinoma. J Oral Pathol Med. 33(1): 30-36.
- Birajdar SS, Radhika M, Paremala K, Sudhakara M, et al. (2014). Expression of Ki-67 in normal oral epithelium, leukoplakic oral epithelium and oral squamous cell carcinoma. J Oral Maxillofac Pathol. 18(2): 169-176.
- Craven JM, Pavalic ZP, Stambrook PJ, Pavalic L, et al. (1992). Expression of c-erbB-2 gene in human head and neck carcinoma. Anticancer Res. 12(6B): 2273-2276.
- Hou L, Shi D, Tu S-M, Zhang H-Z, et al. (1992). Oral cancer progression and c-erbB-2/neu proto-oncogene expression. Cancer Lett. 65(3): 215-220.
- Kurokawa H, Matsumoto S, Murata T, Yamashita Y, et al. (2003). Immunohistochemical study of syndecan-1 down regulation and the expression of p53 protein or Ki-67 antigen in oral leukoplakia with or without epithelial dysplasia. J Oral Pathol Med. 32(9): 513-521.