Previous Issues Volume 2, Issue 1 - 2018
Does Immunohistochemical CD47 Staining Intensity Predict Prognozis of Renal Cell Carcinoma?
Eymen Gaze1,Engin Kaya2,Nesrin Turhan3,Metin Yığman4,Mehmet Emin Şirin5,Cavit Ceylan6,Öner Odabaş6
Corresponding Author: Engin Kaya, Gulhane Training and Research Hospital, Department of Urology, Ankara,Turkey, Tel: +00903123045607; Email: [email protected] Received Date: 18 May 2018 Accepted Date: 01 Jun 2018 Published Date: 06 Jun 2018 Copyright © 2018 Kaya E Citation: Gazel E, Kaya E, Turhan N, Yigman M, et all. (2018). Does Immunohistochemical CD47 Staining Intensity Predict Prognozis of Renal Cell Carcinoma. Mathews J Urol Nephrol 2(1): 007.
ABSTRACT
Introduction-Purpose : As for all cancer types, many biomarkers have been identified for renal cell carcinoma (RCC) as well. These biomarkers have been used to investigate the correlation between prognosis, tendency to metastasis and response given by advanced stage patients to treatment. In this retrospective study, in which the immunohistochemical staining intensity of CD47 was compared to the tumor stage and its histopathological features in patients underwent surgery due to renal mass, it was aimed to find whether CD47 could be used as a indicator in either one of diagnosis, follow-up and treatment processes. We believe that our study will contribute to the literature, since there have been no studies on the relationship between RCC and CD47 until today
Material-Method: In this study, pathology specimens of 110 patients who underwent radical nephrectomy and whose tumor pathologies were compatible with RCC were retrospectively examined. These pathology preparations were stained with CD47 using immunohistochemical technique and their staining intensities were examined.Their demographic data such as age and sex was found using clinical database. Parameters such as tumor diameter, capsule invasion, vascular invasion and lymph node involvement were found using pathology results reported after surgery. Patients were divided into groups as those who have clear cells and those who have non-clear cells (papillary and chromophobe); those with a tumor size smaller than 4cm, between 4-7 cm and bigger than 7cm; those with a fuhrman grade of 1-2 or 3-4; according to presence of capsular invasion, vascular invasion, lymph node involvement, and distant metastasis; those with local (T1-2-3) or advanced stage (T4 and/or N1,M1) cancer; weak stained (staining grade 0-1) and strong stained (2-3) according to CD47 staining intensity. In our study, Fischer's exact chi-square test was used in order to test the distribution of categorical variables among groups. Kruskal Wallis variance analysis was used in order to investigate differences of continuous variables between groups and the groups that creates the difference were identified using multiple comparison tests. In order to summarize the results of the study, median (minimum and maximum) and percentages were used for continuous variables. The limit of significance was p=0.05.
Findings:It has been found that 83 (75.5%) of the specimens were stained weak and 27 (24.5%) of the specimens were stained strong with CD47. A comparison between CD47 staining intensities of tumor tissue and non-tumor tissue taken from the same kidney has shown that although tumor tissue was stained stronger than non-tumor tissue, the statical analysis of this situation has shown that these two groups showed a 92.4% similarity in terms of staining patterns (confidence interval 95%). It has been found that non-clear cell group stained significantly stronger than clear cell group (p< 0.001). A significant relationship could not be found between CD47 staining intensity and large tumor diameter, presence of capsular invasion, relationship could not be found between CD47 staining intensity and large tumor diameter, presence of capsular invasion, vascular invasion or distant metastasis, which are known to be poor prognostic factors for RCC. Specimens belonged to patients with lymph node involvement stained strongly and this difference was statistically significant (p=0.02). Tumor specimens belonged to advanced stage patients stained significantly strong with CD47 compared to patients with local disease (p=0.04). A significant relationship was found between CD47 staining intensity and Fuhrman Nuclear Grade, which is thought to be the most important poor prognostic factor after tumor stage for RCC (p=0.009). Conclusion: In order to consider CD47 as a prognostic factor in renal cell cancer, there is a need for large scale prospective studies in which the survival of patients is presented.
KEYWORDS
Renal Cell Cancer; CD47; Biomarker; Prognosis.
INTRODUCTION
The most common solid lesion seen in kidneys is renal cell carcinoma (RCC).It constitutes 90% of all renal tumors.RCC, which constitutes approximately 3% of all cancers in adults, is one of the most aggressive tumors of urogenital system and the most common tumor in urogenital system following prostate and bladder cancer [1].
As for all cancer types, many biomarkers have been identified for RCC as well. These biomarkers have been used to investigate the correlation between prognosis, tendency to metastasis and response given by advanced stage patients to treatment.
CD47 is one of these markers and in recent years, it has been investigated for various cancer types to show its importance. CD47 is a glycoprotein which is a transmembrane receptor for immunoglobulins superfamily. In some studies, it has been found that CD47 sent a “don’t eat me”message to phagocytic cells and its expression increased in tumor cells [2]. It has been shown that increased CD47 expression caused the treatment to fail and the prognosis to worsen by preventing tumor phagocytosis [3]. Also, it has been seen in experimental studies that the effectiveness of treatments such as chemotherapy and radiotherapy has been increased in CD47+ tumors treated with CD47 antibody [4].
In this retrospective study, in which the immunohistochemical staining intensity of CD47 was compared to the tumor stage and its histopathological features in patients underwent surgery due to renal mass, it was aimed to find whether CD47 could be used as a indicator in either one of diagnosis, followup and treatment processes. We believe that our study will contribute to the literature, since there have been no studies on the relationship between RCC and CD47 until today. MATERIAL-METHOD
In this study, pathology specimens of 110 patients who underwent radical nephrectomy in our Urology Clinic and whose tumor pathologies were compatible with RCC were retrospectively examined. Their demographic data such as age and sex was found using clinical database. Parameters such as tumor diameter, capsule invasion, vascular invasion and lymph node involvement were found using pathology results reported after surgery. T staging was carried out according to criteria determined by “American Joint Committiee On Cancer in 2002. Fuhrman Nuclear Grading was used for nuclear grading. Immunohistochemical Study For immunohistochemical study on both tumor and non-tumor tissue, 3-micron thick sections were taken from paraffin blocks using rotary microtome and these were examined using Poly-L-lysine coated slides.
“The Avidin-biotin method” was used as the immunohistochemical staining system.Two drops of antibodies of these dyes were dropped on slides to be dyed with CD47 (Dako EnVision Catalog No: 1046; Dako, Denmark).
Slides taken into Mayer hematoxylin for contrast staining were kept in for 20 seconds and they were washed with distilled water. Entellan was dropped on these tissues passed from alcohol series and xylene, and they were mounted with lamella.
For each case, CD47 staining intensity was assessed using Olympus CX31 microscope and slides prepared with immunohistochemical method and graded according to method mentioned below.This grading was carried out by a pathologist who had no information on clinical and pathological properties of the patients.Brown staining in the cytoplasm and membrane of tumor cells was used as criteria for CD47 antigen positivity.
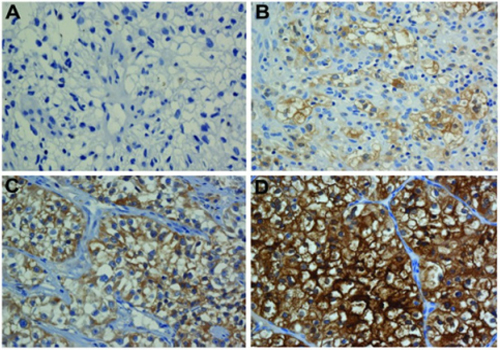
The staining assessment was performed in four categories, the pathology specimens were divided into the following categories (Figure 1);
Figure 1: CD47 staining intensity categories A: (0: No staining in tumor cells),B: (1+:staining can be seen in 20x10 mm enlargement),C: (2+: staining can be seen in 10x10 mm enlargement), D: (3+: staining can be seen in 4x10 mm enlargement).
0: No staining in tumor cells 1+: staining can be seen in 20x10 mm enlargement 2+: staining can be seen in 10x10 mm enlargement, 3+: staining can be seen in 4x10 mm enlargement.
Group Formation Patients were divided into groups as those who have clear cells and those who have non-clear cells (papillary and chromophobe); those with a tumor size smaller than 4cm, between 4-7 cm and bigger than 7cm; those with a fuhrman grade of 1-2 or 3-4; according to presence of capsular invasion, vascular invasion, lymph node involvement, and distant metastasis; those with local (T1-2-3) or advanced stage (T4 and/or N1,M1) cancer; weak stained (staining grade 0-1) and strong stained (2-3) according to CD47 staining intensity.
Statistics In our study, Fischer's exact chi-square test was used in order to test the distribution of categorical variables among groups. Kruskal Wallis variance analysis was used in order to investigate differences of continuous variables between groups and the groups that creates the difference were identified using multiple comparison tests. In order to summarize the results of the study, median (minimum and maximum) and percentages were used for continuous variables. The limit of significance was p=0.05. In order to investigate the concordance among calculated staining grades of the same patients, Intraclass Correlation Coefficient was used. FINDINGS
110 patients who underwent surgery due to renal mass and whose pathologies were compatible with RCC were included in this study.
Demographic findings Among the patients who were included in the study, 32 (29.1%) were female and 78 (70.9%) were male. The average age of patients was 63.8 (std deviation: 11.2). Histopathological findings According to tumor histologies of patients, 80 (72.7%) patients with clear cells and 30 (27.3%) patients with non-clear cells (papillary and chromophobe).23 (20,7%) of the patients in the non-clear cell group with papillary type RCC and 7 (6,6%) with chromophobe type RCC. The average tumor size was 6.9 cm and 27 (24.5%) patients with tumor size smaller than 4 cm, 38 (34.5%) patients with tumor size between 4-7 cm and 45 (40.9%) patients with tumor size bigger than 7 cm. It was found that 53 (48.2%) patients were Fuhrman nuclear grade 1 or 2 and 57 (51.8%) patients were Fuhrman nuclear grade 3 or 4. 59 (44.5%) patients with capsular invasion and 61 patients with no capsular invasion (55.5%). 32 (29.1%) patients with vascular invasion and 78 patients with no vascular invasion (70.9%). 16 (14.5%) patients with lymph node involvement and 94 patients with no lymph node involvement (85.5%). 8 (7.3%) patients with distant metastasis and 102 patients with no distant metastasis (92.7%).89 (80.9%) patients with local disease (T1-2-3) and 21 (19.1%) patients with advanced stage disease (T4 and/or N1, M1). Findings on CD47 staining intensity Our study showed that 83 (75.5%) of the specimens were stained weakly and 27 (24.5%) of the specimens were stained strongly with CD47. A comparison between CD47 staining intensities of tumor tissue and non-tumor tissue taken from the same kidney has shown that although tumor tissue was stained more severely compared to non-tumor tissue, the statical analysis of this situation has shown that these two groups showed a 92.4% similarity in terms of staining patterns (confidence interval 95%). Investigating the relationship between CD47 staining intensity and gender, 25 out of 32 (78.1%) female patients had weak staining, while 7 (21,9%) had strong CD47 staining.58 out of 78 (74.4%) male patients had weak staining, while 20 (25,6%) had strong CD47 staining.This difference was not statistically significant (p=0.80).It has been found that being male or female didn't affect CD47 staining of tumor on a statistically significant level (p=0.80) (Table 1).
Table 1 : Relationship with paitent's pathoclinic features and CD47 staining intensity.
| CD47 staining intensity | ||||
|---|---|---|---|---|
| Total (n=110) | Strong (%) | Weak(%) | P value | |
| Gender | 0.80 | |||
| Male | 78 | 20 (25.6) | 58 (74.4) | |
| Female | 32 | 7 (21.9) | 25 (78.1) | |
| Tumor type | <0.001 | |||
| Clear Cell | 80 | 5 (6.2) | 75 (93.8) | |
| Non-Clear Cell | 30 | 22 (73.3) | 8 (26.7) | 0.40 |
| <4cm | 27 | 5 (18.5) | 22 (81.5) | |
| 4-7cm | 38 | 8 (21.1) | 30 (78.9) | |
| >7cm | 45 | 14 (31.1) | 31 (68.9) | |
| Furhman grade | 0.009 | |||
| 1-2 | 53 | 7 (13,2) | 46 (86.8) | |
| 3-4 | 57 | 20 (35.1) | 37 (64.9) | |
| Capsular invasion | 0.50 | |||
| Positive | 49 | 14 (28.6) | 35 (71.4) | |
| Negative | 61 | 13 (21.3) | 48 (78.7) | |
| Lymph node involvement | 0.02 | |||
| Positive | 16 | 8 (50) | 8 (50) | |
| Negative | 94 | 19 (20.2) | 75 (78.8) | |
| Vascular invasion | 0.33 | |||
| Positive | 32 | 10 (31.2) | 22 (68.8) | |
| Negative | 78 | 17 (21.8) | 61 (78.2) | |
| Metastasis | 1.0 | |||
| Positive | 8 | 2 (25) | 6 (75) | |
| Negative | 102 | 25 (24.5) | 77 (75.5) | |
| Stage of disease | 0.04 | |||
| Local | 89 | 18 (20.2) | 71 (79.8) | |
| Advanced | 21 | 9 (42.9) | 12 (57.1) | |
Considering the relationship between CD47 staining intensity and histological subtype of tumor, 75 out of 80 (93.8%) patients with clear cells had weak staining, while 5 (6.2%) had strong CD47 staining.8 out of 30 (26.7%) patients with nonclear cells had weak staining, while 22 (73.3%) had strong CD47 staining. This difference was found to be statistically significant (p<0,001). It has been found that the group with non-clear cells stained with CD47 significantly stronger than the group with clear cells (p< 0.001). (Table1) Regarding the relationship between CD47 staining intensity and Fuhrman nuclear grading, 46 out of 53 (86.8%) grade 1-2 patients had weak staining, while 7 (13.2%) had strong CD47 staining. 37 out of 57 (64.9%) grade 3-4 patients had weak staining, while 20 (35.1%) had strong CD47 staining. This difference was found to be statistically significant (p=0,009). Thus, it has been found that patients with higher Fuhrman grades had a significantly higher level of CD47 staining intensity. (Table1) Considering the relationship between CD47 staining intensity and lymph node involvement, 8 out of 16 (50%) patients with lymph node involvement had weak staining, while 8 (50%) had strong CD47 staining.In other words, 8 out of 27 (29.6%) specimens stained strongly belonged to patients with lymph node involvement, only 8 out of 83 (9.6%) specimens stained weakly belonged to patients with no lymph node involvement. This difference was statistically significant (p=0,02).Thus, it has been found that patients with lymph node involvement had a significantly higher level of CD47 staining intensity. (Table1) The relationship between CD47 staining intensity and local or advanced stage is examined, 18 out of 89 (20.2%) patients with local tumor had strong staining, while 9 out of 21 (42.9%) advanced stage disease patients had strong CD47 staining. This difference was found to be statistically significant (p=0,04). Thus, it has been found that advanced stage disease patients had a significantly higher level of CD47 staining intensity. (Table1) DISCUSSION
Renal Cell Cancer is the most fatal type of cancer among urological cancers.Its universal incidence is 200.000 new cases annually and more than 100.000 of cases result in death each year [5].Local or distant recurrence is seen in 20-30% of RCC patients within 5 years after nephrectomy [6,7]. In spite of immunotherapy and targeted treatments, 5-year survival of this recurren group is below 20% [8,9].This is why it is very important to determine the prognosis of the disease as early as possible. Today, the prognosis of the RCC is tried to be determined using parameters such as tumor stage, size, grade and necrosis score, which are based on clinicopathology [10,11,12]. The most important of these parameters are pathologic tumorlymph node-metastasis (TNM), Fuhrman nuclear grade and patient performance status.Although it is possible to classify risks with these parameters, the heterogeneous structure of the disease makes it difficult to predict the prognosis of patients.For this reason, there have been studies on various molecular and genetic tissue markers to determine the prognosis of the disease.It has been understood that significant steps could be taken as a result of these studies to predict the prognosis of the disease and even treatment of it by developing antibodies [8,13,14]. A wide range of biochemical and genetic markers have been investigated for RCC as well, however a marker that might be ideal for all criteria of RCC could not be found yet. CD (Cluster of Differantiation) originated markers have been studied for RCC and have become popular in recent years. There is an increase in the expression of CD47 in many malignancies seen in humans. There are studies that indicates that strong CD47 expression is a diagnostic marker and a negative prognostic factor [15,16,17]. A study that clearly demonstrates the relationship between CD47 and RCC has not been reported in the literature yet.The only known study is that of Nishiyama Y. et al., which was performed on rats. In this study, rats were exposed to ferric nitrilotriacetate, which is a renal carcinogen, and it was observed that CD47 expression increased in rats that developed RCC. Also, CD47 expression was stronger in specimens taken from non-tumor tissue compared to specimens taken from tumor tissue and it has been noted as an unexpected finding of the study [18]. Although it was found in our study that tumor tissue was stained more intensity compared to non-tumor tissue, the statical analysis of this situation has shown that these two groups showed a 92.4% similarity in terms of staining patterns (confidence interval 95%). This could be interpreted as that the kidney tissue considered to be tumor-free might be oncologically unhealthy and can show a staining similar to that of tumor tissue. To clarify this uncertainty will only be possible by comparing the tumor tissue with the kidney tissue taken from another group, which have no oncological disease. We have investigated the relationship between parameters accepted as prognostic factors for RCC and CD47 staining intensity in our study. As we know, the histological subtype is important for the prognosis of the disease. Chromophobe and papillary RCCs are usually tend to be more localized and differentiate better, hence the prognosis is better [19]. In an earlier study, chromophobe and papillary RCCs have been compared to conventional RCCs and it has been found that conventional RCCs had significantly poor prognosis [20]. It has been found that the group with non-clear cells stained significantly more severely with CD47 compared to the group with clear cells (p< 0.001).This finding contradicts with literature information which indicates that increased CD47 staining intensity is a poor prognostic factor. Tumor diameter is another prognostic factor for RCC. Gulliani et al. have reported 5-year survival 84% in tumors smaller than 5 cm, %50 in tumors between 5-10 cm and 0% in tumors bigger than 10 cm [21]. Other studies have reported quite good prognosis in unilateral tumors smaller than 4 cm. 5-year survival in these patients is above 90% [22, 23]. On the other hand, it has been found in our study that there was no statistically significant relationship between the severity of CD47 staining and tumor size (p=0.4). It has been shown that microvascular invasion had prognostic significance in RCC cases. In a study, it has been found that microvascular invasion was associated with tumor size, grade, and stage and it was observed in T1 tumors [18]. In another study, it has been observed that it was associated with tumor diameter, grade and prognosis, but not an independent prognostic parameter [23]. According to results of our study, there was no statistically significant relationship between the severity of CD47 staining and vascular invasion or capsular invasion presence (p=0.33, p=0.5). Nuclear grade assessment method is a subjective and pathologist-dependent method. Therefore, there is no ideal grading classification [24]. Still, metastasis and local recurrence have been associated with pathological stage and most importantly life expectancy [25]. In Fuhrman's original article, 5-year survival have been reported to be 64%, 34%, 31% and 10% for grade 1 to 4 respectively and that nuclear grade was the most important prognostic factor in Stage 1 tumors [26,27,28]. In our study, it has been found that the group with higher Fuhrman grade [3,4] was stained with CD47 significantly stronger than the group with lower Fuhrman grade (p=0.009). The stage is the most important prognostic factor by itself in RCC. A lot of studies have shown that the stage was the most important factor that determines the course of the disease [29]. In these studies, it has been reported that 5-year survival was 70-90% in local disease, while this ratio decreased by 15- 20% with perirenal fat invasion presence. In our study, considering the relationship between CD47 staining intensity and local or advanced stage, 18 out of 89 (20.2%) patients with local tumor had strong staining, while 9 out of 21 (42.9%) advanced stage disease patients had strong CD47 staining. This difference was found to be statistically significant (p=0,04). Thus, it has been found that advanced stage disease patients had a significantly higher level of CD47 staining intensity. Lymph node involvement is a bad prognostic factor as well. 5 and 10-year surveys are 5-30% and 0-5%. In our study, it has been found that patients with lymph node involvement had a significantly higher level of CD47 staining intensity (p=0.02). Systemic metastasis is also a bad prognostic factor for RCC. Survival in these patients is less than 50% in one year, 5-30% in five years, and %0-5 in 10 years [30]. In our study, it has been found that there was no statistically significant relationship between the severity of CD47 staining and distant metastasis presence (p=1.0). CONCLUSION
As in many cancer types, CD47 is expressed on surface of renal cancer cells as well. In some studies, clear cell histologic subtype, which is considered to be poor prognostic factor for RCC, were stained less with CD47. A significant relationship could not be found between CD47 staining intensity and large tumor diameter, presence of capsular invasion, vascular invasion and distant metastasis, which are known to be poor prognostic factors for RCC. The cancer stage is the most important factor that affects prognosis and survival in RCC. Tumor specimens belonging to advanced stage patients stained significantly stronger with CD47 than patients with local disease. A significant relationship was found between CD47 staining intensity and Fuhrman Nuclear Grade, which is thought to be the most important poor prognostic factor after tumor stage for RCC.
In order to consider CD47 as a prognostic factor in renal cell cancer, there is a need for large scale prospective studies in which the survival of patients is present
REFERENCES
- Ljungberg B, Cowan NC, Hanbury DC, Hora M, et al. (2010) European Association of Urology Guideline Group. EAU guidelines on renal cell carcinoma: the 2010 update. Eur Urol. 58(3): 398-406.
- Majeti R, Chao MP, Alizadeh AA, Irving L, Weissman, Et al. (2009). CD47 is an adverse prognostic factor and therapeutic antibody target on human acute myeloid leukemia stem cells. Cell. 138(2): 286-299.
- Willingham SB, Volkmer JP, Gentles AJ, Sahoo D, et al. (2012). The CD47-signal regulatory protein alpha (SIRPa) interaction is a therapeutic target for human solid tumors. Proc Natl Acad Sci U S A. 109(17): 6662-6667
- Dotsikas MG, Konowalchuk T, Major PP, Kovac PE, et al. (1987). Cellular heterogeneity in normal and neoplastic human urothelium: a study using murine monoclonal antibodies. Br J Cancer. 56(4): 439-44.
- Parkin DM, Bray F, Ferlay J and Pisani P. (2005). Global cancer statistics. CA Cancer J Clin. 55(2): 74-108.
- Crispen PL, Boorjian SA, Lohse CM, Leibovich BC, et al. (2008). Predicting disease progression after nephrectomy for localized renal cell carcinoma: the utility of prognostic models and molecular biomarkers. Cancer. 113(3): 450- 460.
- Zisman A, Pantuck AJ, Dorey F, Wieder J, et al. (2002). Risk group assessment and clinical outcome algorithm to predict the natural history of patients with surgically resected renal cell carcinoma. J Clin Oncol. 20(23): 4559-4566.
- Klatte T, Seligson DB, Allan J.Pantuck, Leppert JT, et al. (2008). The chemokine receptor CXCR3 is an independent prognostic factor in patients with localized clear cell renal cell carcinoma. J Urol. 179(1): 61-66
- Motzer RJ, Hutson TE, Michaelson MD, Tomczak P, et al. (2009). Overall survival and updated results for sunitinib compared with interferon alfa in patients with metastatic renal cell carcinoma. J Clin Oncol. 27(22): 3584-3590.
- Frank I, Blute ML, Cheville JC, Lohse CM, et al. (2002). An outcome prediction model for patients with clear cell renal cell carcinoma treated with radical nephrectomy based on tumor stage, size, grade and necrosis: the SSIGN score. J Urol. 168(6): 2395-2400.
- Patard JJ, Kim HL, Lam JS, Belldegrun AS, et al. (2004). Use of the University of California Los Angeles integrated staging system to predict survival in renal cell carcinoma: an international multicenter study. J Clin Oncol. 22(16): 3316-3322.
- Zisman A, Pantuck AJ, Belldegrun AS, Dorey F, et al. (2001). Improved prognostication of renal cell carcinoma using an integrated staging system. J Clin Oncol. 19(6): 1649-1657.
- Lam JS, Klatte T, Figlin RA, Kim HL. Et al. (2008). Prognostic factors and selection for clinical studies of patients with kidney cancer. Crit Rev Oncol Hematol. 65(3): 235-262.
- Volpe A and Patard JJ. (2010). Prognostic factors in renal cell carcinoma. World J Urol. 28(3): 319-327.
- Majeti R, Chao MP, Weissman IL, Alizadeh AA, et al. (2009). CD47 is an adverse prognostic factor and therapeutic antibody target on human acute myeloid leukemia stem cells. Cell. 138: 286-299
- Van Niekerk CC, L. G. Poels, Ramaekers FC, Hanselaar AG, et al. (1993). Changes in expression of differentiation markers between normal ovarian cells and derived tumors. Am J Pathol. 142(1): 157-77.
- Buist MR, Kenemans P, Molthoff CF, Baak JP, et al. (1995). Tumor uptake of intravenously administered radiolabeled antibodies in ovarian carcinoma patients in relation to antigen expression and other tumor characteristics. Int J Cancer. 64(2): 92-98.
- Lerner SE, Hawkins CA, Blute ML, Zincke H, et al. (1996). Disease outcome in patients with low stage renal cell carcinoma treated with nephron sparing or radical surgery. J Urol. 155(6): 1868-1873.
- Naggar AK, Batsakis JG, Wang G and Lee MS. (1993). PCRbased RFLP screening of the commonly deleted 3p loci in renal cortical neoplasms. Diagn Mol Pathol. 2(4): 269-276.
- Naoe M, Ogawa Y, Morita J, Omori K, et al. (2007). Detection of circulating urothelial cancer cells in the blood using the cell search system. Cancer.109(7): 1439-1445.
- Goldstein NS. (1997). The current state of renal cell carcinoma grading. Union Internationale Contre le Cancer and the American Joint Commitee on Cancer. Cancer. 80(5): 977-980.
- Paharnik S, Ross F, Hampel C, Thüroff JW, et al. (2006). Nephron sparing surgery for renal cell carcinoma with normal contralateral kidney: 25 years of experience. J Urol. 175(6): 2027-2031.
- Van Poppel H, Vandendriessche H, Boel K, Mertens V, et al. (1997). Microscopic vascular invasion is the most relevant prognosticator after radical nephrectomy for clinically nonmetastatic renal cell carcinoma. J Urol. 158(1): 45-49.
- Sevinc M, Kirkali Z, Yörükoğlu K, Mungan U, et al. (2000). Prognostic Significance of Microvascular Invasion in Localized Renal Cell Carcinoma. Eur Urol. 38(6): 728-33.
- Fuhrman SA, Lasky LC and Limas C. (1982). Prognostic significance of morphologic parameters in renal cell carcinoma. Am J Surg Pathol. 6(7): 655-663.
- Green LK, Ayala AG, Ro JY, Swanson DA, et al. (1989). Role of nuclear grading in stage I renal cell carcinoma. Urology. 34(5): 310-15.
- Bonsib SM. (1999). Risk and prognosis in renal neoplasms. Urol Clin North Am. 26(3): 643-660.
- Thrasler JB and Paulson DF. (1993). Prognostik factors in renal cancer. Urol Clin North Am. 20(2): 247-262.
- Guiliani L, Gibrti C, Martorana G, Rovida S, et al. (1995). Radical extensive surgery for renal cell carcinoma: long term results and prognostic factors. J Urol. 153(): 901- 903
- Nishiyama Y, Tanaka T, Toyokuni S, Naitoh H, et al. (1997). Overexpression of integrin-associated protein (CD47) in rat kidney treated with a renal carcinogen, ferric nitrilotriacetate. Jpn J Cancer Res. 88(2): 120-128.