Previous Issues Volume 1, Issue 1 - 2016
Chemistry, Origin, Antitumor and Other Activities of Fungal Homo-Dimeric Alkaloids
Dmitry V Kuklev1 and Valery M Dembitsky2
1Department of Biological Chemistry, University of Michigan Medical School, Ann Arbor, MI 48109, USA. 2Institute for Drug Discovery, P.O. Box 45289, Jerusalem 91451, Israel. Corresponding Author:Valery M Dembitsky, Institute for Drug Discovery, 8 Ha-Marpe Str, P.O. Box 45289, Jerusalem 91451, Israel, Tel: +972-526877444; E-Mail: [email protected] Received Date: 27 Feb 2016 Accepted Date: 17 Mar 2016 Published Date: 23 Mar 2016 Copyright © 2016 Dembitsky VM Citation: Kuklev DV and Dembitsky VM. (2016). Chemistry, Origin, Antitumor and Other Activities of Fungal Homo-Dimeric Alkaloids.Mathews J Pharm Sci. 1(1): 004.
ABSTRACT:
Homo-dimeric alkaloids produced by fungi, lichenized fungi and fungal endophytes are a structurally unique class of natural products with extensive biological activities that are presented in this article. More than 100 selected fungal metabolites have been confirmed to exhibit antitumor, antimicrobial, antibacterial, and other activities KEYWORDS: Homo-dimeric; Alkaloids; Fungi; Fungal Endophytes; Lichens Activities. INTRODUCTION: Alkaloids are nitrogen-containing metabolites found mainly in plant species; they are also found in fungi, microorganisms and marine invertebrates [1-3]. Studies on bioactive alkaloids, done with respect to both their biological activity and the role of alkaloids in the introduction of new drugs, have gained considerable importance during the past fifty years, and have been received a prominent position in the field of organic and medicinal chemistry [4-7]. Homo-dimeric alkaloids produced by fungi are a structurally unique class of natural products with extensive biological activities that are presented in this article. This mini-review article is intended to provide an overview of the properties of homo-dimeric alkaloids produced by fungi, fungal endophytes, and lichenized fungi. Homo-Dimeric Alkaloids: All dimeric alkaloids are complex molecules consisting of two structurally similar monomers linked by bonds that can be weak or strong, covalent or intermolecular. The term homodimer is used when the two monomer molecules are identical [1-4]. The alkaloidal pigment necatarone, its dehydrodimer (1), which prevails in aged fruiting bodies, and its 10-deoxydehydrodimer (6) are major pigments of the green-brown flesh and cap skin of Lactarius turpis [8,9].
The alkaloids 4,4'-binecatorone (1) and 10-deoxy-4,4'-binecatorone (2) were isolated from fruiting bodies of Lactarius necator, while the American species L. atroviridis contains 10,10'-dideoxy-4,4'-binecatorone (3) as the major alkaloid [9].
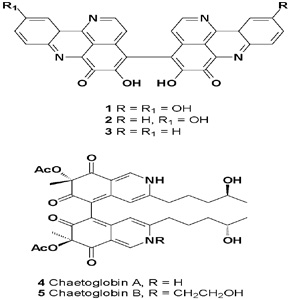
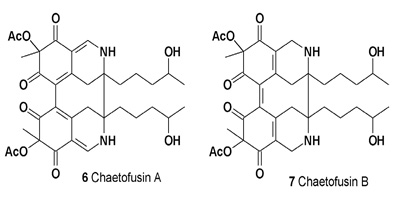
Chaetoglobins A (4) and B (5), antitumor azaphilone alkaloid dimers with an unprecedented skeleton, were detected in the endophytic fungus C. globosum [10]. The culture broth of Chaetomium fusiforme, from liverwort Scapania verrucosa, afforded two azaphilone alkaloids called chaetofusins A (6) and B (7). Both compounds have shown antifungal activity [11].
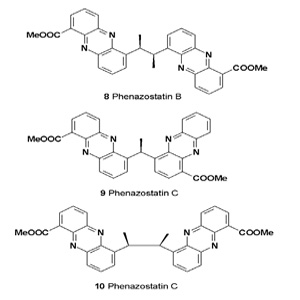
The diphenazine alkaloids designated phenazostatins A-D were isolated from the culture broth of Streptomyces sp. 833. In a cell assay, phenazostatin B (8) inhibited glutamate toxicity in N18-RE-105 cells with an EC50 value of 0.3 μM [12,13].
Phenazostatin B has also shown free radical scavenging activity. Phenazostatin C (9) protected neuronal N18-RE-105 cells from glutamate toxicity in a dose-dependent fashion (EC50 = 0.37 μM), and had antioxidant activity against free radicalinduced lipid peroxidation in liver microsomes [14].
An actinomycete from the littoral sediment of Mauritius (Indian Ocean), Pseudonocardia sp., was the source of the phenazine derivative phenazostatin D (10), and the antibiotic phenazostatin B (8).
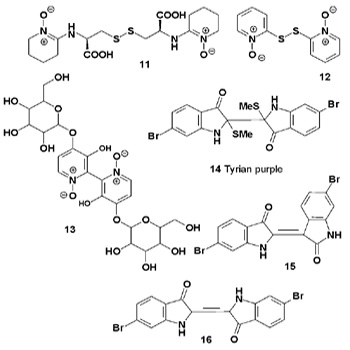
Three unusual disulfide metabolites (11-13) have been isolated from the fruiting bodies of the basidiomycete Cortinarius sp., collected in the Catlins (New Zealand). Both compounds (11) and (12) have shown significant cytotoxicity and antimicrobial activity [15]. The well-known dimeric brominated indole alkaloid Tyrian purple (14) and two other compounds (15 and 16) have been isolated from the deep-sea actinomycete Serinicoccus profundi [16]. Compound (14) was also found in the lichenized fungi Roccella tincteria and in the other lichen species Umbilicaria sp. and Parmelia perlata [17,18]. It was found that several endophytic fungi, including Camptotheca acuminata and Nothapodytes foetida, produced the anticancer alkaloid camptothecin (17a).
Three camptothecin-producing endophytic fungi, Aspergillus sp. LY341, Aspergillus sp. LY355, and Trichoderma atroviride LY357, were isolated and identified from Camptotheca acuminate [19].
The endophytic fungus Trichoderma atroviride LY357 was obtained from the tree Camptotheca acuminate, and its fermentation broth extract contained camptothecin [19-22].
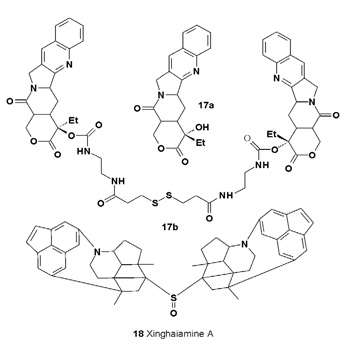
Compound (17b) has shown strong activity against tumor cell lines A-431 (IC50 = 0.15 μM), Hela (IC50 = 0.7 μM) and HL-60 (IC50 = 0.92 μM). The antibiotic xinghaiamine A (18) has been isolated from the marine-derived actinomycete Streptomyces xinghaiensis NRRL B24674T.
Xinghaiamine A has exhibited broad-spectrum antibacterial activities against both Gram-negative pathogens (e.g., Acinetobacter baumannii, Pseudomonas aeruginosa, and E. coli), and Gram-positive ones, which include Staphylococcus aureus and Bacillus subtilis. Furthermore, xinghaiamine A has shown potent cytotoxic activity against human cancer cell lines MCF- 7 and U-937 with IC50 values of 0.6 and 0.5 μM, respectively [23].
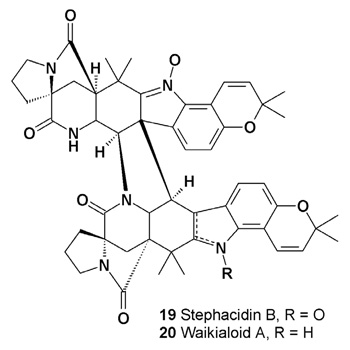
Stephacidin B (19), an antitumor dimeric N-hydroxyindole antibiotic, was isolated from the fungus Aspergillus ochraceus [24]. Waikialoid A (20), a dimeric prenylated indole alkaloid, isolated from an Aspergillus species (Waikiki Beach, Honolulu, Hawaii), has inhibited biofilm formation with an IC50 value of 1.4 μM [25].
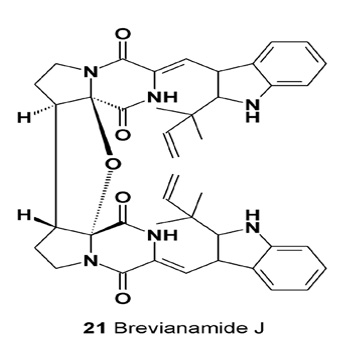
Dimer brevianamide J (21), together with diketopiperazine alkaloids, brevianamides K-N, were isolated from Aspergillus versicolor [26].
Ditryptophenaline (22) and known mycotoxins have been isolated from the marine-derived fungus Aspergillus flavus, and have shown cytotoxicity against HL-60, MOLT-4, A-549, and BEL-7402 human cell lines [27].
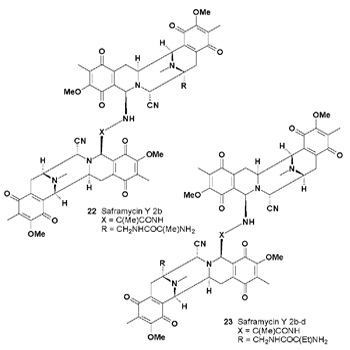
Ditryptophenaline was also isolated from another strain of Aspergillus flavus [28]. A series of saframycins: Y3, Yd-1, Yd- 2, Ad-1, Y2b-d and the dimer of Y2b (23), have been directly biosynthesized by the saframycin producer, Streptomyces lavendulae [29]. Both compounds (22 and 23) have shown anticancer activities [29].
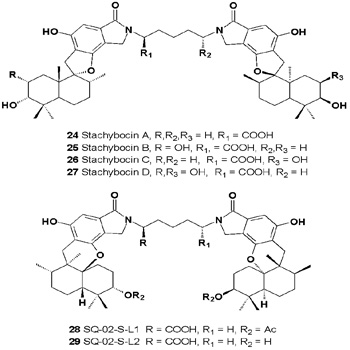
More than 20 years ago, Nakamura and co-workers discovered a series of metabolites, the stachybocins (24-27), from cultures of Stachybotrys sp. M6222 [30]. The isolated metabolites consisted of two spirobenzofuran units, each fused to one of two substituted drimane sesquiterpenoids, which were connected by a lysine residue [31].
These compounds inhibited the binding of radiolabeled endothelin- 1 to human endothelin receptors type A with IC50 values of 13, 12, and 15 μM, respectively, for isomers A, B, and C, and with IC50 values for inhibiting binding to the type B receptor of 7.9, 9.5, and 9.4 μM, respectively. Stachyflins (SQ-02-S-L2 28 and SQ-02-S-L1 29), two anti-influenza virus agents, were found in an extract of Stachybotrys sp. RF-7260 (genus Memnoniella) [32].
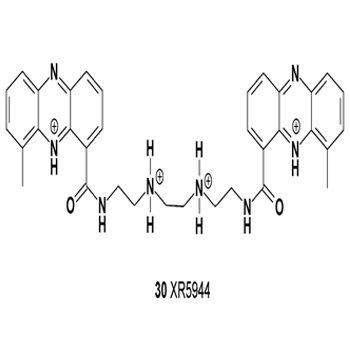
The anticancer agent XR5944 (30) has shown antitumor activity both in vitro and in vivo. Against a panel of human cell lines in vitro, the IC50 values of XR5944 ranged from 0.04 to 0.4 nM/L [33]. XR5944 also induced cleavage complex formation for topoisomerases I, IIα, and IIβ in human leukemic K562 cells visualized using the trapped in agarose DNA immunostaining (TARDIS) assay [34].
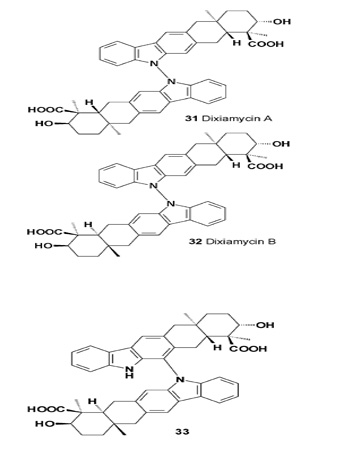
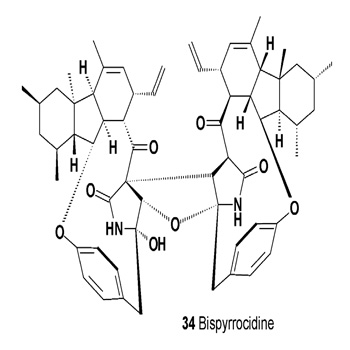
The indo-sesquiterpene alkaloids dixiamycin A (31), dixiamycin B (32), and compound (33), which are antibacterial and antitumor agents, have been obtained from the fermentation broth of Streptomyces SCSIO 02999 and were extracted with butanone [35]. The endophytic fungus Neonectria ramulariae KS-246 has produced a homo-dimer bispyrrocidine (34) with specific non-competitive inhibitory activity against a prolyl oligopeptidase (IC50 = 2.6 μM) [36,37].
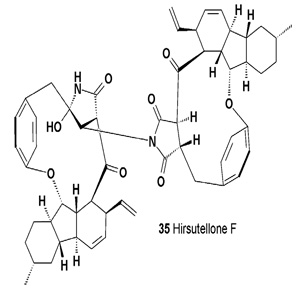
The alkaloid dimer hirsutellone F (35), as well as the monomers hirsutellones A, B, and C were isolated from the seed fungus Trichoderma sp. BCC 7579 [38].
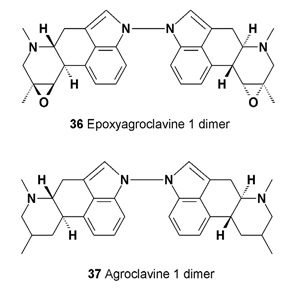
Compound (36), an epoxyagroclavine 1 dimer, and compound (37) have been isolated from the culture fluid of Penicillium sizovae [39,40].
Cultures of Furcatum sp., Penicillium citrinum, P. corylophilum, P.fellutanum, and P. waksmanii have produced the ergot alkaloids agroclavine-I, and epoxyagroclavine-I; their N-N dimers (36 and 37) (e.g. the dimer of epoxy-agroclavine-I) and the mixed dimer of epoxyagroclavine-I and agroclavine-I [41].
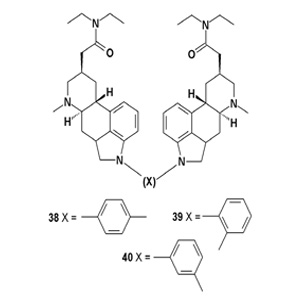
Several dimers of clavine alkaloids were synthesized from natural agroclavine. Dimers linked via an aromatic spacer (38-40) have shown a high toxicity (1 μM) to lymphoma cells, which was not detected in the other derivatives.
In vitro and in vivo experiments demonstrated an immunesuppressive effect of dimers with an aromatic spacer and an NK cell stimulatory effect of the terguride hexamer and trimer with an aliphatic spacer. There is considerable evidence that the indolic part of the molecules contributes to the immunosuppressive action of tergurides, which dimers carrying an aromatic linker also potentially possess [42].
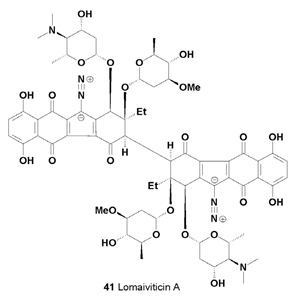
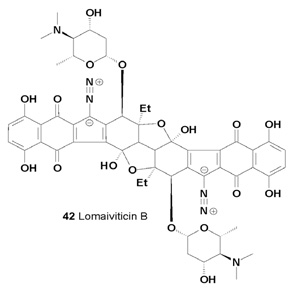
The secondary metabolites of Salinispora spp. that have been reported to date are unusual and very interesting. These compounds are lomaiviticins A (41) and B (42) [43].
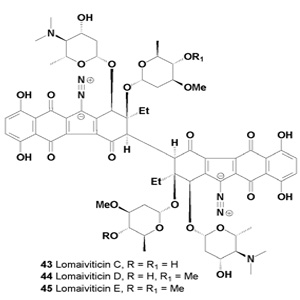
A series of potent antitumor antibiotics named lomaiviticins A (41), B (42), C (43), D (44) and E (45) have been isolated from the fermentation broth of Micromonospora lomaivitiensis [44,45]. The metabolite (-)-lomaiviticin A (41) was isolated as a potent cytotoxic agent with half-maximal inhibitory concentrations (IC50) in the 0.007-72 nM range against twenty five human cancer cell lines [46].
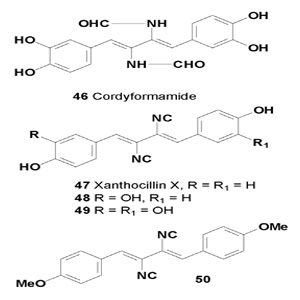
The plausible biogenetic precursor of xanthocillin Y2, called cordyformamide (46) was isolated from a culture broth of the insect pathogenic fungus Cordyceps brunnearubra BCC 1395. This compound was found to exhibit activity against the malarial parasite Plasmodium falciparum K1 (IC50 = 18 µM), whereas its cytotoxic activity is too weak to be significant [47].
Several metabolites containing an isocyanide group (47-50) have been isolated from Aspergilus species. Xanthocillin X (47) has been isolated from Penicillium notatum by Rothe in 1950, and was also isolated from Dichotomomyces albus, D. cejpii, and P. chrysogenum [48,49]. Compound (47) was found to be active against an Ehrlrich ascites carcinomabearing mouse strain with a median lethal dose (LD50) of 40 mg/kg, and it was also shown that this metabolite inhibited chronic myeloid leukemia K-562, human cervical cancer HeLa, breast cancer MCF-7 and MDA-MB-231, liver cancer HepG2, lung cancer NCI-H460, and prostate cancer DU-145 cell lines, with IC50 values between 0.4 and 12 µg/mL [48,50,51].
Compound BU-4704 (49) inhibited human colon HCT-116 and murine melanoma B16-F10 cell lines with IC50 values of 0.6 and 4.3 µg/mL, respectively [52], in addition compound (50) also inhibited HepG2, MCF-7 and KB cancer cell lines with IC50 values of 0.2, 0.4 and 0.4 µg/mL, respectively [53].
The antitumor compound SD118 (51), from a deep-sea sediment sample of Penicillium commune, has been shown to inhibit the up-regulation of the class III PI3K/Beclin 1 signaling pathway, and HepG2 cells [54].
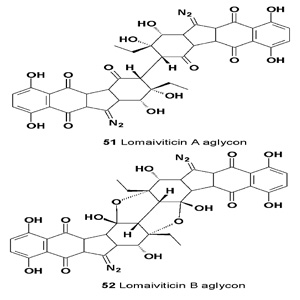
The lomaiviticins are potent anti-proliferative anti-microbial alkaloids isolated from various strains of Streptomyces and Salinispora [55]. All lomaiviticins contain a diazotetrahydrobenzo[ b]fluorene (diazofluorene) functional group, which is unique among known natural products.
The lomaiviticins A (51) and B (52) were isolated from the fermentation broth of an actinomycetes strain of Micromonospora LL-371366, which was initially isolated from the core of a host ascidian.
The fermentation broth exhibited potent DNA-damaging activity and was cytotoxic against a number of cancer cell lines. Lomaiviticin A (51) exhibited activity against a variety of cancer cell lines (IC50 values of 0.01-9.8 ng/mL) [56]. (-)- Lomaiviticin A glycoside residues and the monomeric lomaiviticin aglycon (51) are cytotoxic agents that induce double-strand breaks (DSBs) in DNA [56,57].
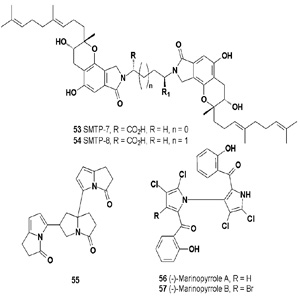
SMTP-7 (53) and SMTP-8 (54) are prenyl phenol compounds and staplabin analogs that have been isolated from cultures of Stachybotrys microspora IFO 30018. Both metabolites have shown anti-thrombotic activity [58]. Pyrazoline-3-one trimer (55) has been isolated from the endophytic fungus Aspergillus fumigatus sp. R7 [59], and from the marine-derived Streptomyces sp. QD518 [60].
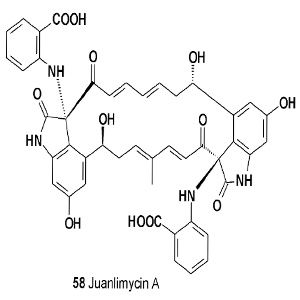
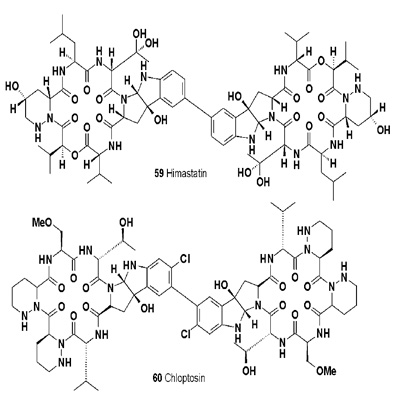
Two chlorinated alkaloids, named marinopyrroles A (56) and B (57), were isolated from marine Streptomyces. Both pyrroles possess potent antibiotic activities against methicillinresistant Staphylococcus aureus [61]. An ansamycin macrodilactam with the unprecedented feature of juanlimycin A (58) was reported in Streptomyces sp. LC6 [62]. The isolated compound has shown antibacterial activity. Himastatin (59) is a depsipeptide antitumor antibiotic produced by cultured broth of Streptomyces hygroscopicus (ATCC53653) [63-65].
Chloptosin (60), adimeric cyclohexa-peptide and a rare apoptosis- inducing agent, was isolated from the culture broth of Streptomyces sp. Chloptosin was found to induce apoptotic activity in the apoptosis-resistant human pancreatic adenocarcinoma cell line AsPC-1 and shown strong antimicrobial activity against Gram-positive bacteria, including methicillinresistant Staphylococcus aureus [66].
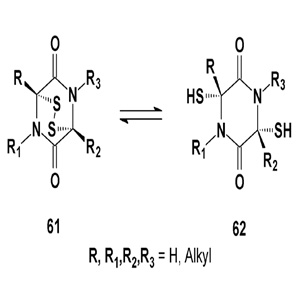
Epipolythiodiketopiperazines are bioactive alkaloids produced by a number of filamentous fungi including those from the Aspergillus, Chaetomium, Leptosphaeria, Penicillium, Pithomyces and Verticillium genera; these alkaloids have been shown to have anticancer and other activities [67-69]. Dimeric metabolites comprise the core bioactive constituents of the highly cytotoxic extracts from fungi of the genus Chaetomium.
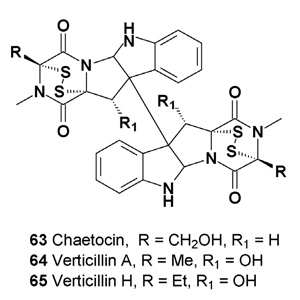
This unique class of fungal alkaloids is characterized by a sulfur- bridged dioxopiperazine (61), a feature generally requisite for the potent biological activity prevalent among members of this class. At present, two simple biologically active epidithiodioxo- piperazines, (61) and (62), and their derivatives have been described [70,71]. Chaetocin (63) is a fungal metabolite with antimicrobial and cytostatic activity [72-74]. Chaetocin has also shown strong cytotoxicity against HeLa cells (IC50 = 0.05 µg/mL) [73-75].
The cytotoxic dioxopiperazine dimer verticillin A (64) was isolated from the fungus Verticillium sp. [76]. The same compound (64) has also been isolated from the mycelium of a marine-derived fungus Penicillium sp.
The isolated metabolite has exhibited potent in vitro cytotoxicity against HCT-116 human colon carcinoma cells (IC50 = 30 ng/mL) [77,78]. Verticillin A (64) and H (65) were isolated from two filamentous fungi of the Bionectriaceae strains MSX 64546 and MSX 59553.
Compounds (64 and 65) have shown cytotoxicity against a panel of human cancer cell lines, displaying IC50 values ranging from 1.2 µM to 10 nM [79]. Verticillin D (66) was isolated from solid-substrate fermentation cultures of Gliocladium catenulatum and Bionectra byssicola F120 [80].
Verticillin G inhibited the growth of Staphylococcus aureus, including methicillin-resistant and quinolone-resistant S. aureus, with MICs ranging from 3 to 10 µg/mL [81]. A diketopiperazine dimer, ditryptophenaline (67), was isolated from the fungus Aspergillus flavus [82].
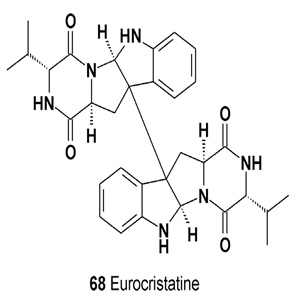
Eurocristatine (68) was isolated from a culture of the spongeassociated fungus Eurotium cristatum KUFC 7356 [83].
Brevianamide S (69) isolated from Aspergillus versicolor exhibited selective antibacterial activity against Bacille Calmette- Guerin, which is suggestive of a new mechanism of action that could inform the development of next-generation antitubercular drugs [84].
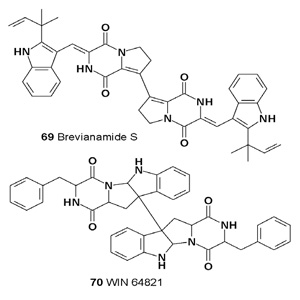 Homo-Dimeric Symmetrical Alkaloids:
Homo-Dimeric Symmetrical Alkaloids:
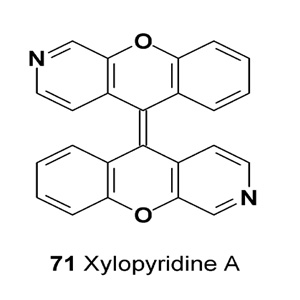
The fungal metabolite xylopyridine A (71) was isolated from Xylaria sp. #2508 (South China Sea). This metabolite has shown a strong DNA-binding affinity [86].
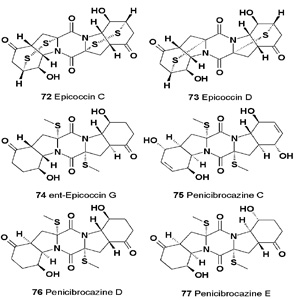
Epicoccins C (72) and D (73), two unique epipolythio-dioxopiperazines possessing unusual sulfur bridges, have been isolated from cultures of a Cordyceps-colonizing isolate of the fungus Epicoccum nigrum. Both compounds have shown modest antimicrobial activity [87].
Ent-epicoccin G (74) has been isolated from the Epicoccum nigrum. It has shown potent activity in vitro against the platelet- activating-factor-induced release of β-glucuronidase in rat polymorphonuclear leukocytes, with an IC50 value of 3.07 µM, and exhibited detectable cytotoxic activities toward six tumor cell lines (A549, Be-l7402, BGC-823, HCT-8, HCT-116, and A2780) in the MTT assay [88].
The sulfide diketopiperazine derivatives penicibrocazines C (75), D (76) and E (77), were isolated and identified from the Penicillium brocae MA-231, an endophytic fungus obtained from the fresh tissue of the marine mangrove plant Avicennia marina. All compounds (75-77) have exhibited antimicrobial activity against some of the tested strains, with MIC values ranging from 0.25 to 64 μg/mL [89].
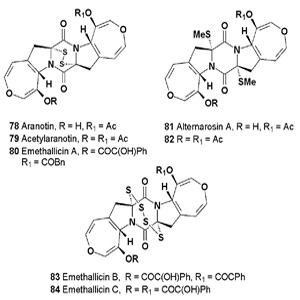
Two alkaloids, aranotin (78) and acetylaranotin (79) have been isolated from Arachniotus aureus and Aspergillus terreus, and exhibit antiviral activity that is apparently selective for RNA viruses such as the polio, Coxsackie (A21), rhino-, and parainfluenza viruses through inhibition of viral RNA synthesis [90-96]. The alkaloid emethallicin A (80) from the fungus Emericella heterothallica has shown potent inhibitory activity against histamine release from mast cells [97].
A halotolerant fungus Alternaria raphani has produced a diketopiperazine, alternarosin A (81), which displayed very weak antimicrobial activity against E. coli, Bacillus subtilis and Candida albicans, and also shown cytotoxicity against human HL60 cells, was also reported [98]. The Aspergillus terreus strain C-520, which was isolated from a soil sample collected in the city Takarazuka, produced several plant growth regulators, including the dimer (82) [99,100].
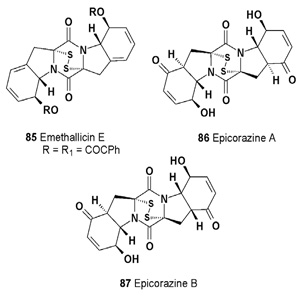
The emethallicins B (83), C (84), and E (85), along with emethallicin A (80), were isolated from Emericella heterothallica. Compounds (83 and 84) are epitetrathiodioxopiperazines and have the same basic carbon skeletons as apoaranotin and acetylaranotin, respectively [89]. The isolated emethallicins (83-85) have potent inhibitory activities against compound 48/80-induced histamine release from mast cells, as does emethallicin A [101]. Epicoccum nigrum was found to exhibit activity against Staphylococcus aureus, and from its fermentation broth, two antibacterial compounds, epicorazines A (86) and B (87), were isolated [102].
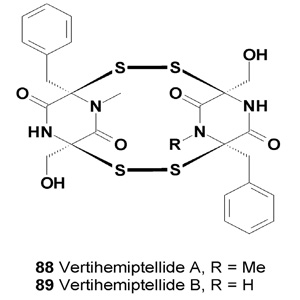
Epicorazines A-C display marginal activity against Gram-positive bacteria, S. aureus and E. faecalis. These compounds exhibit anti-proliferative effects against L929 mouse fibroblast cells and K562 human leukemia cells as well as cytotoxicity toward the HeLa human cervical carcinoma cell line [103]. Two diketopiperazine dimeric derivatives linked by dithio bridges, the vertihemiptellides A (88) and B (89), were isolated from the entomopatho-genic fungus Verticillium hemipterigenum BCC 1449 and found to be moderately cytotoxic and anti-mycobacterial compounds [104].
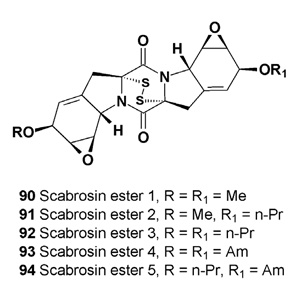
The scabrosin esters (90-94) were isolated from the lichenized fungus Xanthoparmelia scabrosa, which was collected in Australia [105]. These scabrosins were found to exhibit potent cytotoxic activity against the murine P815 mastocytomia cell line (IC50 = 0.5 μM) and the human breast MCF7 carcinoma cell line (IC50 < 1 nM). These scabrosin esters from the lichen X. scabrosa belong to the epipolythiodioxopiperazine class of secondary metabolites, which are characterized by possession of a reactive disulfide bond and have been studied regarding anticancer and other activities [106,107].
Enantiomeric Fungal Alkaloids:
The occurrence of enantiomeric alkaloids in nature is known, however, they are generally produced and isolated as racemic or scalemic mixtures. Natural chiral alkaloids are usually produced in the optically pure form, but occasionally both enantiomers are formed by different fungal species [1,2,4,7,109,110].
One of the largest groups within the fungal indole alkaloids is the group which includes the anticancerous stephacidins (95, 96), notoamides A (97, 98), notoamides B (99, 100), and citrinadins, the insecticidal brevianamides (101, 102, Figure 1) and sclerotiamides, the anthelmintic paraherquamides, the calmodulin-inhibitory malbranche-amides, and the mycotoxin fumitremorgin, among other compounds [111,112].
Figure1:Enantiomeric reverse prenylated indole alkaloids produced by Aspergillus species.
CONCLUSION:
Dimeric alkaloids comprise a rare group of natural products. These alkaloids are mainly isolated from microorganisms, fungi and/or plants. Little information is known about biological activities of these metabolites. Nevertheless, reported activities for isolated compounds have shown strong antibacterial, antimicrobial, and others activities. The widest spectra of pharmacological activities are exhibited by isolated alkaloids and/or their synthesized derivatives. These alkaloids have shown to be promising candidates for the development of new drugs toward several diseases. CONFLICT OF INTEREST: The author confirms that this article content has no conflict of interest.
REFERENCES:
-
Dembitsky VM and Tolstikov GA. (2003). Natural halogenated alkaloids. Chem. Sustainable Develop. 11, 451-466.
-
Kok A. (1966). A short history of orchil dyes. Lichenologist. 3, 248-272.
-
Lindsay WL. (1854). Dyeing properties of lichens. Edinburgh New Philosoph. J. 42, 228-250.
-
Luo Y, Pu X, Qu X, Chen F, et al. (2013). Fermentative camptothecin production by endophytic Trichoderma viride strain, Faming Zhuanli Shenqing, Chengdu Institute of Biology, Chinese Academy of Sciences, Peop. Rep. China.
-
Wang Y and Chen H. (2011). Isolation and identification of a camptothecin-producing endophytic fungus from Camptotheca acuminate. Weishengwuxue Tongbao. 38(6), 884-888.
-
Zhang C, Zhang Q, Li S, Tian X, et al. (2012). Preparation of antibacterial and antitumor indosesquiterpene derivatives from Streptomyces, Chinese Patent: CN 102757908 A, Faming Zhuanli Shenqing, 19.
-
Rothe W. (1950). Vorlaufige Mitteilung uber eine neues Antibiotikum, Pharmazie. 5, 190-198.
-
Scheuer PJ. (1992). Isocyanides and cyanides as natural products. Acc. Chem. Res. 25(10), 433-439.
-
Greiner D, Bonaldi T, Eskeland R, Roemer R, et al. (2005). Identification of a specific inhibitor of the histone methyltransferase SU(VAR)3-9. Nat. Chem. Biol. 1, 143-145.
-
Gomesa NM, Dethoup T, Singburaudom N and Gales L. (2012). Eurocristatine, a new diketopiperazine dimer from the marine sponge-associated fungus Eurotium cristatum. Phytochemistry Lett. 5(4), 717-720.
-
Ernst-Russell MA, Elix JA, Chai CLL, Hockless DCR, et al. (1999). Structure revision and cytotoxic activity of the scabrosin esters, epidithiopiperazinediones from the lichen Xanthoparmelia scabrosa. Aust. J. Chem. 52(4), 279-283.
-
Cordell GA. (1997). The Alkaloids: Chemistry and Pharmacology, 49, 1-405
.jpg)