Previous Issues Volume 5, Issue 1 - 2022
Antioxidant Bio-Constituents from Agro-Industrial Waste and their Importance in Functional Foods
Gargi Tikoo1, Bhagat Singh3, Girish Sharma1, Charu Gupta2*
1Amity Institute of Biotechnology, Amity University-UP, Sector-125, NOIDA-201313, India
2Amity Institute of Herbal Research & Studies, Amity University-UP, Sector-125, NOIDA-201313, India
3Department of Medical lab, Technology, Faculty of Paramedical Science, Uttar Pradesh University of Medical Sciences, Saifai, Etawah, UP-206130, India
Received Date: December 9, 2022
Publication Date: December 22, 2022
*Corresponding author: Charu Gupta, Amity Institute for Herbal Research & Studies, Amity University, Noida, 201313, Uttar Pradesh, India; Email: [email protected]
Citation: Gargi Tikoo, et al. (2022). Antioxidant Bio-Constituents from Agro-Industrial Waste and their Importance in Functional Foods. Mathews J Nutr Diet. 5(1):20.
Copyright: Gargi Tikoo, et al. © (2022)
ABSTRACT
The Food processing industries and post-harvest produce large volumes of waste that pose severe pollution problems and loss of valuable nutrients. Such wastes might have a potential for conversion into useful products of high value as food or feed after proper treatment. These fruit and vegetable processing wastes may be good sources of bioactive phytochemicals. Phenols, a major group of antioxidant phytochemicals, have profound importance due to their biological and free radical scavenging activities (FRSA). Free radicals induce cellular and tissue pathogenesis leading to several diseases like cancer, inflammation, and aging processes. Antioxidants provide protection from such damage caused by reactive oxygen species (ROS) in lipid peroxidation, protein damage, DNA strand breakage, and associated disorders. Therefore, the prime focus of the present studies was to manage agri-waste to develop useful products and combat environmental pollution. The wastes like peels, leaves, twigs, flowers, pod pericarp, seed meal, seed coat, and residues from kitchen waste were studied for total phenolic content (TPC) and antioxidant activity (AOA) to explore their potential for use in nutraceuticals or functional foods.
Keywords: Agri-horticultural wastes, Antioxidant activity, Total Phenolic contents (TPC), Free Radical Scavenging Activity (FRSA), Functional foods
INTRODUCTION
Agro-industrial wastes represent the organic as well as the inorganic material which is generated as residues from various sectors such as agriculture, different industries like food and beverage, diary production, livestock, etc. From the food industry, various sources of waste generated are fruits and vegetables, fish, milk and meat, etc. Due to these large amounts of waste that is being generated worldwide, researchers are recovering the antioxidant bio-constituents from these agro-industrial wastes. The agro-industries yield a very huge amount of waste such as the seeds, peels, flowers, pericarp, pulp, and husks of coffee, etc. The majority of these wastes comprise of enriched constituents such as phenol compounds. These phenolic compounds are essential for antioxidant activity. Phenolic compounds are aromatic having one or more than one substituent of hydroxyl and also have small to high molecular weights, all of these phenol compounds are usually mentioned as “Polyphenols”. Polyphenols are usually divided into two main categories. The first category is the phenols that generally have a low molecular weight that is the Low Molecular Weight Phenols (LMWP) or the Extractable Polyphenols (EP). These LWMPs or EPs include flavonoids, benzoic acid, stilbenes, xanthones, etc. The second category is the High Molecular Weight Polyphenols (HMWP) or the Non-Extractable Phenols (NEP). These HWMPs or NEPs include tannins, lignans, etc. These Extractable Polyphenols and Non-Extractable Polyphenols from the residues of agro-industries characterize the sources of value-added compounds with their uses as constituents in functional foods or in supplements of diet due to their various benefits in health which include antioxidant activity also. Antioxidants, immunostimulants, and microbiota modulators are just a few of the biological properties of these phenolic compounds. Furthermore, they have anticancer, anti-inflammatory, and antibacterial properties (Hernández-Carlos et al., 2019) [1].
Antioxidants are recognizable as reducing oxidative damage which is linked with many diseases which include cancer, diabetes, ageing, cardiovascular diseases, and many more. The oxygen radicals provoke oxidative stress which is accepted to be prime factor in several diseases, also in the standard process of ageing. Reactive Oxygen Species (ROS) are the highly proactive compounds that are produced from oxygen and which include peroxide, superoxide, etc. Reactive Oxygen Species or ROS are free radical species, and they have very high tendency to react with the available biomolecules and affect their function as well as their structure. They majorly affect the structures of Deoxyribonucleic Acid (DNA), Ribonucleic Acid (RNA), and Proteins. The Reactive Oxygen Species (ROS) are majorly caused either by exogenous sources also known as the extracellular sources or from endogenous sources also known as the intracellular sources. The extrinsic factors include Xenobiotics, infection from microbes, exposure to UV Radiation, Stress, or Growth Factors or Cytokines and Chemokines. While the intrinsic ROS can be generated via the Oxidation Reactions of metabolic pathways in the Mitochondria, Stress of Endoplasmic Reticulum (ER), metabolizing enzymes, drug metabolism, and inflammation. ROS majorly cause Oxidative Stress which in turn causes number of disorders. Accumulation of these cellular Reactive Oxygen Species can disturb or oxidize the contents of cell such as the phospholipid cell membrane layer, proteins, and DNA. This thus endorses the release of pro-inflammatory mediators. This oxidative damage of cells can thus help in the formation of oxidative stress, which sequentially causes various age-related disorders especially Cancer, Ageing, Diabetes, Arthritis, etc. Excess Reactive Oxygen Species causes oxidative stress, which further causes the oxidation of lipid, DNA Damage, and injuries to cellular components (Fierascu et al., 2019) [2].
To overcome this increasing level of free radicals from both exogenous as well as from endogenous sources, our body has a natural oxidative stress defense mechanism to protect the human body against cellular damage. The ROS defense mechanism comprises the antioxidant machinery which helps to reduce the damage induced by oxidative stress as mentioned above. This defense mechanism includes the Repair Mechanism which basically repairs the DNA enzymes, the Physical Defense Mechanism which provides the protective barriers around the skin and cells so that there is no entry of Reactive Oxygen Species into the human body and the body also tried the Prevention Mechanism to overcome the production of these ROS by chelating metals.
Another machinery working against this oxidative stress is the antioxidant defense which includes Non-Enzymatic and Enzymatic Antioxidants. The enzymatic antioxidants include Catalase (CAT), Glutathione Reductase (GRx), Superoxide Dismutase (SOD), Ascorbate Peroxidase (APX) and Glutathione Peroxidase (GPx) while the Non-Enzymatic Antioxidants which forms the other half part of the antioxidant machinery generally comprises of the Low Molecular Weight Antioxidant (LMWA) compounds and Plant Antioxidants (Valenzuela et al., 2003) [3]. These compounds are efficient of inhibiting oxidative injury interacting directly or indirectly. These can be further divided into various subtypes. These are the dietary sources which include Tocopherols, Vitamins A, E and C and Carotenes; Phytochemicals which include Bioflavonoid and Polyphenols; Direct Acting Low Molecular Weight Antioxidant that includes Scavenging Free Radicals; and Indirect Acting Low Molecular Weight Antioxidant which includes Transition Metal Chelators. Both the arms of this machinery, that is, the antioxidant enzymes and the non-antioxidant enzymes, work together to lower the damaging effects of the Reactive Oxygen Species (ROS) and develop tolerance to a variety of environmental stressors (Mohamed, 2015; Engwa et al., 2018) [4,5].
Recent research indicates that the natural antioxidants synthesis from agro-industrial waste or from fruits and vegetable waste has received a lot of interest recently. Waste materials like flowers, peels, seeds, etc. are generated in large quantities during vegetables, fruits, and other agro-industrial waste processing. Dumping of these wastes generally represents a problem (Jimenez-Lopez et al., 2020) [6]. Furthermore, essential nutrients from agricultural as well as industrial wastes are lost. Thus, new features involving the usage of these agro-industrial wastes as by-products for some further utilization in the creation of high-value food supplements or high-additive food additives have aroused interest, because these are the high-value goods and their recapture may be economically beneficial and appealing as well (Arun et al., 2022) [7]. Natural antioxidants can be found in these Agro-industrial residues or leftovers. These natural antioxidants present in agro-industrial leftovers have a significant role in the food industry. These are then recovered through various advanced extraction methods. Functional foods, Nutra or pharmaceuticals, beauty products, and food additives can all benefit from the substances that have been recovered. Both natural as well as synthetic antioxidants have a broad range of applications in the food industry since they are used as an additive in food and also used in functional foods (Leyva-López et al., 2020) [8]. Functional foods are nutritional food items that in addition to delivering energy and nutrients, benefit two or even more targeted functions within the human body by improving a certain physiological response or decreasing the possibility of the diseases such as cancer, Alzheimer’s, etc. (Fierascu et al., 2019) [2].
Figure 1: Natural Antioxidants, Functional Foods, Stress Prevention, and Benefits of Well-Being.
METHODOLOGY
Raw Materials
Samples of different agro-industrial wastes such as peels of Citrus nobilis x Citrus deliciosa also known as Kinnow; Petals, leaves, stem, and flowers of Calendula Plant; Peels of Citrus x sinensis, also known as Orange; Flower petals of Tagetes, also known as Marigold; the peel or the Exocarp of papaya; peel of Banana; Exocarp or Epicarp of Pomegranate, Potato, Cucumber, Bottle Gourd; the peels of Allium cepa that are also known as Onion and the Seed Coat of Arachis hypogaea were collected from different sites.
Figure 2: Calendula Sample.
Figure 3: Kinnow Sample.
Figure 4: Pomegranate Sample.
Figure 5: Tagetes Sample.
Isolation or Extraction of Essential Oils
The stems, leaves, and flower of the plant Calendula, leaves, and stem of the plant Cymbopogon also known as Sweet Lime, Peels of Citrus limetta, and Peels of Citrus nobilis x Citrus deliciosa also known as Kinnow were taken. All of these samples were dried for 1 to 2 days at room temperature one by one or one at a time. The dried aerial material was crumpled. The crumpled material was placed in a round bottom glass flask of 1 Liter and it was filled with about 500 ml of tap water. After that, about 2 to 3 ml of hexane was added to the glass flask. The Extraction of essential oils was done via the Hydro-distillation Clevenger method. The experimental setup was positioned in a heating mantle that was attached to a refrigerator which will certify the condensation of the essential oils. This experiment was carried out at a distillation temperature between 110°C-150°C and the distillation time of approximately 4 to 5 hours, which are the optimal conditions for this experiment. By the end of hydro-distillation, there were two phases that were witnessed. The first one was an aqueous phase i.e., aromatic water and the second one was an organic phase i.e., an Essential Oils, which was less dense than the H2O. The essential oils were collected and were stored in the vials which were sealed. Those vials were then stored in the refrigerator at a temperature of about 4° C, until it will be further used.
Phenol Test
The Phenol-Ciocalteu (F-C) reaction is used to determine an antioxidant’s capacity of reduction by an electron transfer-based antioxidant assay. It is commonly used to determine the total phenol or polyphenol content of plant-based foods and biological samples. It is the reduction of folin-ciocalteu reagent. When the phenols are present, molybdenum-tungsten blue is produced, which can be measured spectrophotometrically at 720 nm.
Reagents Used:
- Folin-Ciocalteu reagent (1N)
- Gallic Acid (as reference) 1mg/ml
- 20% Na2CO3
- Blank which is used for auto-zero
Preparation of Blank: Take 1 ml of distilled H2O in a test tube. Then add 0.5 ml reagent (folin-ciocalteu) in the test tube and add 1 ml of 20% Na2CO3 and left for 20 minutes at room temperature.
Preparation of Sample: Take the sample and weigh 250 mg on a filter paper and transfer it into a small conical flask or a small beaker. Then add 10 ml 50% methanol (CH3OH), then left it overnight. Filtrate it via filter paper. Throw the residue as it will be not used and keep the filtrate as it will be used further in the test.
Procedure: Take aliquots of the sample in 0.1 and 0.2 ml in two test tubes. Mark them as test tube ‘A’ and test tube ‘B’ and make up to 1 ml in each of the test tubes via distilled water. Then add on 0.5 ml reagent (folin-ciocalteu) in each test tube. Then add 0.1 ml of 20% Na2CO3 in ‘A’ and ‘B’ test tubes. Then left both the test tubes for 20 minutes at room temperature. Then after 20 minutes add distilled water equal to the total volume in each of the glass test tubes i.e., 2.5 ml of distilled water in each of the test tubes. And then take the absorbance, O.D. that is the optical density at λmax = 720 nm.
Preparation of Standard (GallicAcid): Varying concentrations of gallic acid were used to make the standard curve. Different dilutions of the gallic acid were made. For example, for 20 μg/ml of standard, we added 20 ml gallic acid from the Eppendorf to the test tube + 0.5 ml of folin-ciocalteu reagent. Then add 1 ml of Na2CO3 to the test tube. And left it for 20 mins at room temp. Its O.D. was recorded at λmax = 720 nm.
To make 25 μg/ml standard, we added 25 ml of Gallic Acid from the Eppendorf to another test tube + 0.5 ml of the reagent i.e., folin-ciocalteu. Then add 1 ml of Na2CO3 to the test tube. And left it for 20 mins at room temp. Its O. D. was recorded at λmax = 720 nm.
These various concentrations or different dilutions of Gallic Acid were made like 30 μg/ml, 40 μg/ml, 60 μg/ml, 80 μg/ml, and many more. These concentrations or dilutions were used to plot the standard curve.
All Total Phenolic Content (TPC) was exhibited in Gallic Acid Equivalence (GAE).
Figure 6: Different dilutions or concentrations of Gallic Acid.
Figure 7: Different samples used in Folin-Ciocalteu method.
Antioxidant Test (DPPH Method)
DPPH (2,2-diphenyl-1-picryl-hydrazyl-hydrate), is a free radical approach which is an antioxidant test that uses the transfer of electrons to generate a solution which is violet in colour in the presence of ethanol. The free radical which is stable at room temp., is reduced in the existence of an antioxidant molecule, yielding a colourless solution of ethanol. 2,2-diphenyl-1-picryl-hydrazyl-hydrate i.e., DPPH is a free radical scavenging method, which is the first method for determining the antioxidant potential of an extract, a compound or some other biological sources. In this experiment, the discoloration of the solution was used to assess the ability to scavenge the 2-diphenyl-1-picryl-hydrazyl-hydrate (DDPH) radicals. The results were measured on the photo-spectrometer at the wavelength of 517 nm.
Reagents Used
- 2-diphenyl-1-picryl-hydrazyl-hydrate (DDPH)
- Ethanol
- Methyl Alcohol or Methanol (MeOH)
Preparation of Sample: Take the sample and weigh 10 mg on a filter paper and transfer it into a small conical flask or a small beaker. Then add 10 ml 100% methanol (CH3OH), then left it overnight. Filtrate it via filter paper. Throw the residue as it will be not used and keep the filtrate as it will be used further in the test.
Procedure: Take aliquots of the sample in 0.1 and 0.2 ml in two test tubes. Mark them as test tube ‘A’ and test tube ‘B’. In 0.1 ml test tube i.e. ‘A’ test tube add 2.9 ml of 6 * 10-5 Molar DPPH. Whereas in 0.2 ml test tube i.e., ‘B’ test tube add 2.8 ml of 6 * 10-5 Molar DPPH. Then left both the test tubes for 20 minutes at room temperature. Then after 20 minutes add distilled water equal to the total volume in each test tube that is 3 ml of distilled H2O in each of the test tube. After that then take the absorbance, O.D. that is the optical density at λmax = 517 nm.
Preparation of Standard [2-diphenyl-1-picryl-hydrazyl-hydrate (DDPH)]: Varying concentrations of 2-diphenyl-1-picryl-hydrazyl-hydrate (DDPH) were used to make the standard curve. Different dilutions of the DPPH were made. For example, for 0.01 Molar of standard, we added 0.01 Mol gallic acid from the Eppendorf to the test tube + 1000 ml or 1 Litre of distilled water. And left it for 20 mins at room temperature. Its O. D. was recorded at λmax = 517 nm.
To make 0.02 Molar standard, we added 0.02 Mol of DPPH from the Eppendorf to another test tube + 1000 ml or 1 L distilled water. And left it for 20 mins at room temperature. Its O. D. was recorded at λmax = 517 nm.
To make 0.03 Molar standard, we added 0.03 Mol of DPPH from the Eppendorf to another test tube + 1000 ml or 1 Litre of distilled water. And then left it for 20 minutes at room temperature. Its optical density was recorded at λmax = 517 nm.
Like these various concentrations or different dilutions of 2-diphenyl-1-picryl-hydrazyl-hydrate (DDPH) were made like 0.03 Mol/ L, 0.06 Mol/ L, 0.10 Mol/ L, 0.20 Mol/ L and many more. These concentration or dilutions were used to plot the standard curve.
Antimicrobial Activity
Antimicrobial activity can be interpreted as the collaborative term for all active or bioactive agents or constituents which inhibit or kill bacterial growth, prevent microbial colony formation, and may kill the microorganisms.
For this, we first prepared 1000 ml or we can say 1 Litres of Nutrient Agar (NA) media. For the preparation of Nutrient Agar media, firstly we collected the materials such as Agar-Agar, Peptone, Beef Extract, NaCl, and distilled Water. We weighed these components on a weighing machine.
Components Total Amount
NaCl 5 g
Beef Extract 3 g
Peptone 5 g
Agar-Agar 15 g
Distilled H2O 1000 ml or 1 Litre
We can add an extra 1 gram of Agar for the solidifying purpose.
Firstly, fill the Glass Reagent Bottle with half of the distilled water i.e., 500 ml. After weighing put all these components in the Glass Reagent Bottle with 500 ml of distilled water. After adding all of the components fill the glass reagent bottle with another 500 ml of the distilled water to make it 1000 ml or 1 Litre.
Now, we will uniformly mix all of these components in the distilled water with the help of Magnetic Stirrer. The use of magnetic stirrer is to direct the biological experiments or the chemical experiments by mixing two or more components. So, we will put the magnet inside the Glass Reagent Bottle and we will put the bottle onto the magnetic stirrer. We will set the speed of the stirring between 600 to 800 and we will switch on the temperature button so that the beef extract, agar agar, and peptone melt down quickly with the help of heat or temperature, and by constant stirring, all the components will mix down uniformly in the Glass Reagent Bottle. We will keep the bottle to stir and mix for at least 10 to 15 minutes or more depending if the components have mixed uniformly or not. After 10 to 15 minutes, we will turn off the stirrer and take out the magnet piece from the glass bottle. And will centrifuge the Glass Reagent bottle with other experimental materials such as petri plates, forceps, etc. for 15 minutes at 115 psi and 121° C. After 15 minutes of centrifugation, we will directly put all these materials into the Lamina Air Flow and will wait for 5 to 10 minutes to let the media cool so that we can pour it into the Glass Petri Plates or Petri Dishes. After 5 to 10 minutes, we will start pouring the Nutrient Agar Media into the Petri Plates and we will fill half of the Petri Plates with Nutrient Agar Media. We will wait for the Media to solidify for about 30 minutes to 1 hour.
Now, we will do the Agar-diffusion procedure which is a commonly used procedure for discovering the antimicrobial activity of microbial and extracts of the plant. In this procedure, the entire surface of the agar petri plate is inoculated or infected by spreading a volume of microbial inoculum. After that, a hole with a diameter of approximately 6 to 8 mm is aseptically punched with a sterile tip or with a sterile cork borer, and the volume of approx. 20-100 μL of the extract solution or Essential oils solution at the appropriate concentration is put into the punched wells with the help of a micropipette. We took different microbial inoculum like gram negative and gram positive bacteria such as the Staphylococcus aureus. Escherichia coli, Enterobacter aerogenes, Bacillus subtilis, and Pseudomonas fluorescens and inoculated on the entire agar plate surface. And the Essential oils we put into the aseptically punched holes were Citrus Limetta, Citrus nobilis x Citrus deliciosa (also known as Kinnow), and Lemon Grass. After inoculation, we incubated all the Petri plates in the BOD Incubator at 37∘ C for about a day or two and will check the results.
RESULTS AND DISCUSSION
Peel Portion and Essential Oils Yield
The results for the Citrus nobilis x Citrus deliciosa (also known as Kinnow); Calendula Plant; Citrus x sinensis (also known as an Orange); Tagetes (also known as Marigold) and Citrus limetta (also known as sweet lemon) in terms of % of the peel portion and the % of the oil yield of the essential oils are shown in Table 1.
Table 1: % of Peel Portion and % of Oil Yield.
|
Cultivator Common Name |
% of Peel Portion |
% of Oil Yield |
|
Citrus nobilis x Citrus deliciosa (Kinnow) |
34.50 ± 1.15 |
8.61 ± 0.05 |
|
Citrus x sinensis (Orange) |
31.67 ± 0.50 |
1.76 ± 0.10 |
|
Calendula |
35.98 ± 0.05 |
2.08 ± 0.10 |
|
Tagetes (Marigold) |
29.77 ± 0.10 |
5.38 ± 0.50 |
The percentage of peel portion from Calendula plant was higher (35.9%), followed by the fruit Kinnow (34.5%), then Orange (31.6%) and then Tagetes (29.7%). Kinnow (Citrus nobilis x Citrus deliciosa) peels yielded the most oil (8.6%) among all the other fruits and flower species extracted using the Clevenger apparatus hydro distillation method, followed by the Marigold (Tagetes) which yielded oil (5.3%), Calendula yielded oil (2.0%) and then Orange (Citrus x sinensis) which yielded oil (1.7%).
The Chemical Composition of extracted Essential Oils
The percentage concentration of the main chemical components determined in the essential oils extracted from the peels of Kinnow, Orange, and flowers of Calendula and Tagetes is shown below in table 2.
The GC/MS study exhibited a significant descriptive and analytical variance in the chemical composition of the above samples.
Table 2: GC/MS Chemical Composition of extracted Essential Oils.
|
Volatile Compounds |
Kinnow |
Orange |
Calendula |
Tagetes |
|
Limonene |
54.57 |
46.30 |
3.3 |
40.8 |
|
β-Myrcene |
4.16 |
6.27 |
0.2 |
- |
|
α-Pinene |
2.79 |
1.49 |
6.2 |
- |
|
γ-Terpinene |
- |
3.34 |
1.01 |
- |
|
β-Ocimene |
- |
- |
- |
39.4 |
|
Caryophyllene |
1.41 |
- |
0.5 |
39.7 |
|
(Z)-Ocimenone |
- |
- |
- |
9.15 |
|
(Z)-Tagetone |
- |
- |
- |
8.78 |
α-Pinene, Caryophyllene, β-Myrcene, and Limonene were the major constituents that is >1% found in Kinnow. Similarly, Limonene, β-Myrcene, α-Pinene, and γ-Terpinene were major constituents (>1%) found in Orange. γ-Terpinene, Limonene, Caryophyllene, β-Myrcene, and α-Pinene were major constituents (>1%) found in Calendula. Likewise, Limonene, β-Ocimene, Caryophyllene, (Z)-Ocimenone, and (Z)-Tagetone were major constituents (>1%) found in Tagetes.
Total Phenolic Content (TPC) determination
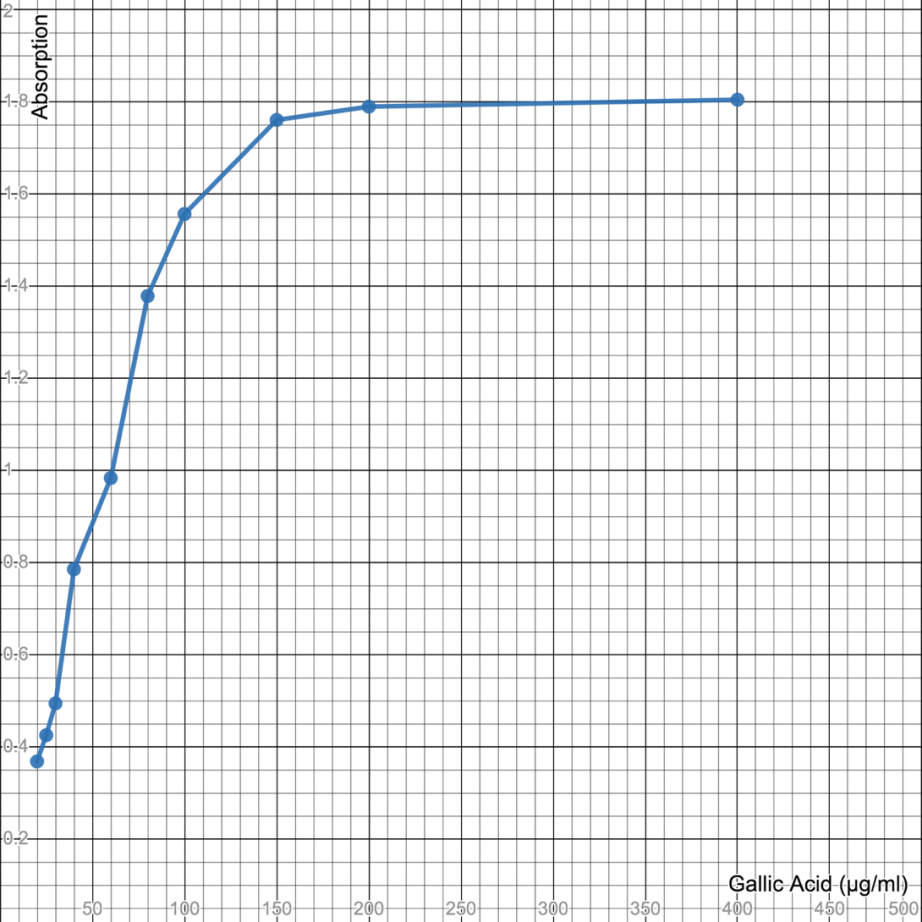
With applying the Folin-Ciocalteu procedure, Total Phenolic compounds were measured. Gallic Acid (GA) as a standard was used to create the calibration curve with values ranging from 20 μg/ml to 400 μg/ml. Gallic Acid is commonly employed as a reference (standard) since it is a chemical that is inexpensively or cheaply available in its highly pure form and has moderate activity. Total phenolic content is usually expressed in terms of the Gallic Acid Equivalence (GAE). The blank was measured spectrophotometrically which came out to be 0.005 and the standard measured spectrophotometrically came out to be 1.969. To measure the absorbance of different dilutions of Gallic Acid different dilutions were first put into the glass cuvette. We have to fill ¾ of the glass cuvette with the different dilutions of gallic acid, one at a time, and then put it in the spectrophotometer whose wavelength is already set at 720 nm. Different Concentrations or dilutions of Gallic Acid (in μg/ml) have different Absorbance that was recorded by the spectrophotometry and are given below in the table and the Standard Gallic Acid curve has been plotted on the Graph.
Table 3: Different dilutions of Gallic Acid and their absorbance measured by UV-Vis Spectrophotometry.
|
S. No. |
Gallic Acid (μg/ml) |
Absorbance (720 nm) |
|
1. |
20 |
0.369 |
|
2. |
25 |
0.426 |
|
3. |
30 |
0.495 |
|
4. |
40 |
0.786 |
|
5. |
60 |
0.984 |
|
6. |
80 |
1.379 |
|
7. |
100 |
1.557 |
|
8. |
150 |
1.761 |
|
9. |
200 |
1.790 |
|
10. |
400 |
1.805 |
Figure 8: Calibration curve (Standard) made with Gallic Acid for Folin-Ciocalteu Total Phenolic Content.
Calculations
By calculating the Gallic acid equivalence of Orange, Tagetes, Calendula, Pomegranate, Potato, and Kinnow, the results we obtained after calculating are given below and are segregated in the table.
|
Gallic Acid Equivalence (GAE) = 20 * Absorbance of sample Absorbance of Standard
|
Table 4: Calculation of Gallic Acid Equivalence (GAE) using UV-Vis Spectrophotometry.
|
Sample |
Concentration (N1 and N2) |
Calculations |
Gallic Acid Equivalence (GAE) |
|
Orange |
0.1 |
20 * 0.101/ 1.969 |
1.025 |
|
0.2 |
20 * 0.098/ 1.969 |
0.995 |
|
|
Tagetes |
0.1 |
20 *.0.010/ 1.969 |
0.101 |
|
0.2 |
20 * 0.020/ 1.969 |
0.203 |
|
|
Calendula |
0.1 |
20* 0.023/ 1.969 |
0.233 |
|
0.2 |
20* 0.024/ 1.969 |
0.243 |
|
|
Pomegranate |
0.1 |
20 * 0.507/ 1.969 |
5.149 |
|
0.2 |
20 * 1.013/ 1.969 |
10.28 |
|
|
Potato |
0.1 |
20 * 0.012/ 1.969 |
0.121 |
|
0.2 |
20 * 0.104/ 1.969 |
1.056 |
|
|
Kinnow |
0.1 |
20 * 0.181/ 1.969 |
1.838 |
|
0.2 |
20 * 0.280/ 1.969 |
2.844 |
Antioxidant Test (DPPH) Determination
With Applying the 2-diphenyl-1-picryl-hydrazyl-hydrate (DDPH) procedure the antioxidant activities of the wastes were measured. DPPH as a standard was used to create the calibration curve with the values ranging from 0.01 μg/ml to 0.60 μg/ml. DPPH is used because it is simple, inexpensive, rapid, and most commonly used which calculates the potential of the compound to serve as a free radical scavenger or H+ donors, as also to evaluate the antioxidant activity of the eatables. Antioxidant activity is usually expressed in terms of the percentage. The blank was measured spectrophotometrically which came 0.064 and the standard measured spectrophotometrically came out to be 0.193. To measure the absorbance of different dilutions of DPPH different dilutions were first put into the glass cuvette. We have to fill ¾ of the glass cuvette with different dilutions of DPPH, one at a time, and then put it in the spectrophotometer whose wavelength is already set at 517 nm. Different Concentrations or dilutions of DPPH (in μg/ml) have their different Absorbance which was recorded by the spectrophotometry and are given below in the table and the Standard DPPH graph for determining radical scavenging activity has been plotted on the Graph.
Table 5: Different dilutions of DPPH and their absorbance measured by UV-Vis Spectrophotometry.
|
S. No. |
DPPH (μg/ml) |
Absorbance (517 nm) |
|
1. |
0.01 |
0.085 |
|
2. |
0.02 |
0.136 |
|
3. |
0.03 |
0.176 |
|
4. |
0.06 |
0.363 |
|
5. |
0.10 |
0.559 |
|
6. |
0.20 |
1.170 |
|
7. |
0.30 |
1.673 |
|
8. |
0.60 |
3.000 |
Figure 9: Standard DPPH graph used for determining the DPPH radical scavenging activity.
Calculations
|
DPPH Scavenging effect = Control-Sample * 100 % Control
|
By calculating the Antioxidant Activity of Orange, Tagetes, Bottle Gourd, Pomegranate, Potato and Kinnow, the results we obtained after calculating are given below and are segregated in the table.
Table 6: Calculation of DPPH Scavenging Effect (in %) using UV-Vis Spectrophotometry.
|
Sample |
Concentration (N1 and N2) |
Calculation |
DPPH Scavenging Effect (in %) |
|
Orange |
0.1 |
0.193-0.208/ 0.193* 100 |
7.77 % |
|
0.2 |
0.193-0.137/ 0.193* 100 |
29.01 % |
|
|
Tagetes |
0.1 |
0.193-0.145/ 0.193* 100 |
24.87 % |
|
0.2 |
0.193-0.065/ 0.193* 100 |
66.32 % |
|
|
Bottle Gourd |
0.1 |
0.193-0.164/ 0.193* 100 |
15.0 % |
|
0.2 |
0.193-0.091/ 0.193* 100 |
52.84 % |
|
|
Pomegranate |
0.1 |
0.193-0.062/ 0.193* 100 |
67.87 % |
|
0.2 |
0.193-0.056/ 0.193* 100 |
70.98 % |
|
|
Potato |
0.1 |
0.193-0.233/ 0.193* 100 |
20.72 % |
|
0.2 |
0.193-0.173/ 0.193* 100 |
10.36 % |
|
|
Kinnow |
0.1 |
0.193-0.212/ 0.193* 100 |
9.80 % |
|
0.2 |
0.193-0.150/ 0.193* 100 |
22.2 % |
Antimicrobial Activity
Outcome of the antibacterial efficacy of these essential oils from the peels of Kinnow, Orange, and the flowers of Calendula and Tagetes against Gram negative bacteria such as the E. aerogenes, P. fluorescens and E. coli and Gram positive bacteria such as the S. aureus and the B. subtilis is shown in Table given below.
Table 7: Zone of Inhibition in Gram negative Bacteria.
|
Essential Oils |
Inhibition Zone Diameter (in Cm) |
||
|
Gram-ve Bactria |
E. aerogenes |
P. fluorescens |
E.coli |
|
Kinnow |
1.9 |
- |
3.7 |
|
Orange |
1.1 |
- |
1.4 |
|
Calendula |
3.3 |
2 |
3 |
|
Tagetes |
1.2 |
0.7 |
1.3 |
|
Essential Oils |
Inhibition Zone Diameter (in Cm) |
|
|
Gram + ve Bacteria |
B. subtilis |
S. aureus |
|
Kinnow |
- |
1.5 |
|
Orange |
- |
1.4 |
|
Calendula |
3 |
2.4 |
|
Tagetes |
1.5 |
- |
In the current study, the essential oils extracted from the flower of Calendula disclosed maximum zone of inhibition in opposition to microbial strains such as the E. aerogenes (3.3 cm) and B. subtilis (3 cm) in comparison to the essential oils of the peel of Kinnow (1.9 cm, 3.7 cm,1.5 cm), the peels of Orange (1.1cm, 1.4 cm, and 1.4 cm) and the flowers of Tagetes (1.2 cm, 0.7 mc, 1.3 cm, and 1.5 cm).
These results demonstrated that the essential oils extracted from the flower of Calendula is more potent or effective against all of the bacterial strains in comparison to Orange, Kinnow, and Tagetes.
Figure 10: Antimicrobial Plates Showing Zone of Inhibition in Gram positive Bacteria and Gram-negative Bacteria.
Figure 11: Antimicrobial Plates Showing Zone of Inhibition.
DISCUSSION
Fruits and vegetable waste as well as wastes obtained from plants are generally made up of peels, flowers, stem, trim residues which are viable sources of useful substances because of their nutritional qualities. The agro-industries yield a very huge amount of waste such as seeds, peels, flowers, pericarp, pulp, and husks of coffee, etc. The majority of these wastes comprise of enriched constituents such as phenol compounds. Fibers, Vitamin E, C, Phenolic Compounds, Carotenoids, and other antioxidants are the most important bioactive components present in various types of wastes, all of which have been shown to have positive impact on human health. These phenolic compounds are essential for the antioxidant activity. These antioxidants are recognizable to reduce oxidative damage which is linked with many diseases which include cancer, diabetes, ageing, cardiovascular diseases, and many more. These antioxidants are getting lost by dumping of the agro-industrial waste. The bioactive compounds that are present in agro industrial waste such as phenols, carotenoids, etc. contain all the magnificent molecules for the production of functional foods, nutraceuticals as well as food additives [9]. Because fruits and vegetables contain high bioactive components, they are the most basic kind of functional foods. The agro-industrial waste can be utilized for the creation of high food additives which can be used as functional food. Functional foods can be defined as nutritional food, which apart from providing nutrients and energy to our body can improve a specific physiological response in our body and can lower the chances or possibility of occurring a disease such as Cancer, Alzheimer’s, ageing, etc. These positive benefits have been related to the radical scavenging properties and antioxidant properties, which can slow or stop protein, DNA, and lipid oxidation. Organic wastes that are generated by industries are dangerous to the environment and can be exploited as a bioresource for extracting bioactive compounds. The availability of extremely sensitive measurement methods and bioactive substances present in particular food wastes or residues provide a tremendous chance to measure, detect or assess these metabolites in a wide range of agro-industrial food waste materials.
Our studies have depicted the presence of phenolic compounds that are present in the agro-industrial waste that is getting lost by dumping these agricultural and industrial waste, and these phenolic compounds are essential for the function of antioxidant activity which can be used by our body to reduce the chances of getting any disease.
CONCLUSION
The presence of phenolic compounds and antioxidants in the agro-industrial waste can help increase or improve both the functional food’s nutritional content as well as its therapeutic benefits. It is essential to utilize only natural antioxidants in functional foods and other nutritional supplements.
REFERENCES
- Carlos BH, Sánchez NFS, Coronado RS, Cañongo CV, Mendoza PCG. (2019). Antioxidant Compounds from Agro-Industrial Residue". Antioxidants. Shalaby E (Ed): IntechOpen.
- Fierascu RC, Fierascu I, Avramescu SM, Sieniawska E. (2019). Recovery of Natural Antioxidants from Agro-Industrial Side Streams through Advanced Extraction Techniques. Molecules 24(23):4212.
- Valenzuela BA, Sanhueza J, Nieto S. (2003). Natural antioxidants in functional foods: From food safety to health benefits. Grasas y Aceites. 54(3):9.
- Mohamed S. (2015). Antioxidants as Functional Foods in Health and Diseases. Austin J Nutri Food Sci. 3(3):1067.
- Engwa GA. (2018). Free Radicals and the Role of Plant Phytochemicals as Antioxidants Against Oxidative Stress-Related Diseases. In: Phytochemicals Source of Antioxidants and Role in Disease Prevention, Asao T, Asaduzzaman Md (Ed), Intech Open.
- Jimenez-Lopez C, Fraga-Corral M, Carpena M, García-Oliveira P, Echave J, Pereira AG, ET AL. (2020). Agriculture Waste Valorisation as a Source of Antioxidant Phenolic Compounds within a Circular and Sustainable Bioeconomy. Food Funct. 11(6):4853-4877.
- Madhavan A, Sindhu R, Binod P, Pandey A, Reshmy R. et al. (2022). Remodeling Agro-Industrial And Food Wastes Into Value-Added Bioactives And Biopolymers. Industrial Crops and Products. 154:112621.
- Leyva-López N, Lizárraga-Velázquez CE, Hernández C, Sánchez-Gutiérrez EY. (2020). Exploitation of Agro-Industrial Waste as Potential Source of Bioactive Compounds for Aquaculture. Foods (Basel, Switzerland). 9(7):843.
- González A, Cruz M, Losoya C, Nobre C, Loredo A, Rodríguez R, et al. (2020). Edible Mushrooms as a Novel Protein Source for Functional Foods. Food Funct. 11(9):7400-7414.