Previous Issues Volume 2, Issue 1 - 2017
A Review on Transdermal Spray: Formulation Aspect
Uttam Kumar Mandal1 , Bappaditya Chatterjee1 , Fatin Husna Binti Pauzi1
1National Institute of Water and Atmospheric Research, Private Bag 14-901, Wellington 6241, New Zealand.
2Department of Biological Chemistry, University of Michigan Medical School, Ann Arbor, MI 48109, USA.
3Institute for Drug Discovery, P.O. Box 45289, Jerusalem 91451, Israel.
Corresponding Author:Department of Pharmaceutical Technology, Kulliyyah of Pharmacy, International Islamic University Malaysia (IIUM), Kuantan, Malaysia, Tel: +60109062750; E-Mail: [email protected]
Received Date: 22 Feb 2016 Accepted Date: 24 Mar 2016 Published Date: 30 Mar 2016
Copyright © 2016 Mandal UK
Citation: Mandal UK, Chtterjee B and Pauzi FHB. (2016).A Review on Transdermal Spray: Formulation Aspect. Mathews J Pharm Sci. 2(1):006.
ABSTRACT:
Transdermal spray offers numerous advantages over the other conventional transdermal drug delivery forms such as gel, ointment and patches, in terms of its cosmeceutical appearance, ready availability for application, flexibility in dosage design, less occurrence of skin irritation and faster drying rate from the application site due to the use of volatile solvent. However, compared to other transdermal drug delivery dosage forms, transdermal spray has the least and limited number of products approved for marketing. Among the drugs are, Evamist ®, an estradiol formulation approved in 2007 by the FDA followed by Axiron ® a non-spray solution to treat low testosterone in men and Recuvyra ®, a pain reliever solution indicated for dogs. This review article focuses current status on the formulation and evaluation of transdermal spray in the background of the role and effects of its composition specially the selection of drugs, volatile solvents, penetration enhancers and film forming polymer, etc. The limitation of transdermal spray highlighted in this review is the concern of its use, especially, the third party exposure particularly for endocrinology indication. Moreover, transdermal spray is also restricted in drugs with large doses due to the limited diffusivity into the skin. The difficulty of exploiting hydrophilic drugs like peptides, macromolecules and new genetic treatments using DNA or small-interfering RNA (siRNA) into transdermal spray formulations is also a limitation that needs to be explored in depth.
KEYWORDS:
Transdermal Drug Delivery; Transdermal Spray; Stratum Corneum; Permeation Enhancer; Transdermal Flux.
INTRODUCTION:
Transdermal drug delivery (TDD) is one of the alternative modes of drug administration preferred over oral and injections. This is due to its distinct advantages such as avoidance of first pass metabolism relating to oral administration, provision of steady state drug-plasma concentration, improvement of patient adherence, prevention of potential gastrointestinal (GI) adverse effects and reduction of medical waste of hypodermic needles in low resource settings [1,2]. The skin provides a large surface area suitable for absorption and the noninvasive procedure for the transdermal drug system such as a patch that enables a continuous intervention with the applied medication [3]. The amount of drug delivered through the skin and the obtained therapeutic effect depends on the ability of the drug to permeate through the skin. The permeation of the drug into the skin is restricted by the stratum corneum (SC), the outermost layer of the skin, which is surrounded by a lipid region [4]. Numerous approaches like iontophoresis, sonophoresis, electroporation, use of chemical permeation enhancers (PE), microniddle, and the use of lipid vesicles have been studied for the last 30 years to break the barrier properties of SC and some of them have produced commercial success [5,6].
The development of the transdermal delivery system can be classified into three generations. The first generation consists of low-molecular weight, lipophilic, and low dose drugs. The second generation uses permeation enhancement methods such as conventional chemical PEs, iontophoresis, and non cavitational ultrasound in order to increase the drug permeability through the SC. The selection of PE was carried out carefully according to several criteria; i) enhanced permeation ability without causing permanent disruption in the structure of the stratum corneum, ii) ability to enhance transdermal flux in maximum amount iii) permeation ability without causing any injury to the deeper tissues. The third generation focuses more on giving the effects on the stratum corneum by incorporating microneedles, thermal ablation, microdermabrasion, electroporation and cavitational ultrasound [7]. Over the years, transdermal patch has emerged as the main form of TDD [8]. However, one of the main concerns in using a patch form is the high level of residual drug in the patch after wear time, which leads to unintended drug exposure and intentional drug abuse [8]. The patch is also associated with the skin hypersensitivity reaction, for example, redness and irritation, blistering and tattooing [9]. Large scale commercial production of the patches involves a certain degree of complexity, especially the scale-up procedures of multi-components patches. Occasionally, the problems of formulation stability and crystallization of drug molecules during long time storage have been evidenced to the marketed products [10]. Due to these particular reasons, gel system are being actively explored and developed as the alternative to patches. The gel system demonstrates almost equivalent clinical performance to the patch system, but with reduced skin irritation and improved patient compliance [11]. Semisolid dosage forms like gel, cream, ointment and paste could be easily rubbed off by the clothing and during routine daily activities. Gel needs to be applied over a large surface on the skin and the occasional transfer to a third party, especially for endocrinic indications is a major concern. In case of fungal infection of the skin, semisolid dosage forms should not be applied over the infected area with fingers. It might have a chance of cross-infection to other body parts.
Transdermal spray (TS) is believed to have more superiority over the conventional TDD systems in the light of its safety, and tolerability [8]. As compared to conventional TDD systems (patches, gel, and ointment), TS is readily available for application, provides flexible drug dosage delivery, and reduces the occurrence of skin irritation and the patients need not to clean their hands after the application [8, 12,13]. Because of volatile solvent content, TS system creates a fast drying and non-occlusive layer on the skin after its application and helps rapid drug permeation through the skin [11,14].
TS offers many benefits and has a wide potential to be developed as the preferred TDD over gels, patches and ointment in order to avoid skin irritations and improved patient's compliance. However, its efficacy is majorly dependent on the selection of appropriate excipients that will help improve the transdermal flux of the drug. So, the goal of this review article is to familiarize the readers about the formulation aspects of TS under the purview of latest developments in this field.
History of Transdermal Drug Delivery:
Human skin has been exploited as the route of drug delivery since ancient time. Medications applied on it can exert local or systemic action depending on their designations and their inherent properties coming out of their composition. Transdermal drug delivery system is defined as, "A drug loaded into any vehicle (e.g., patch, gel, or spray) applied to the skin for systemic action. In addition, the iontophoretic systems included in it irrespective of their systemic or local action" [2]. On the other hand, topical delivery is limited to the application of dosage form to the body surfaces such as the skin or mucous membranes for anti-infective of cosmetic purpose. In 1979, the first transdermal product approved by US-FDA was a scopolamine patch that delivers the drug systemically for three days to treat motion sickness. This was followed by the development of nicotine patch a decade later that emerged as the first blockbuster transdermal product. This achievement had raised the image of transdermal product in the health care field and in the eyes of the medical practitioners as well as the patient population [7].
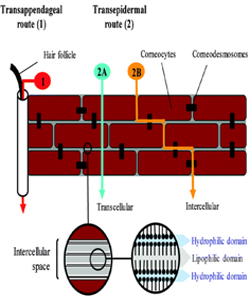
However, for the last three decades, the formulation scientists are searching for other ways and developing novel technologies to overcome the constrain of crossing the SC barrier. Passive diffusion is the most dominant mechanism of drug transport through the skin. Three possible pathways for transdermal delivery have been reported. A drug is transported through the transappendageal route such as hair follicles and sweat glands (1) (shunt pathway), transcellular or intracellular transport through the corneocytes (2A) and intercellular transport via the lipid matrix (2B) as shown in Figure 1 [15]. In transcellular or intracellular pathway the drug crosses the SC straight way through the different layers of the SC by repeated partition through the cornified cells, the extracellular lipid bilayers, viable epidermis and papillary layer of the dermis. In intercellular pathway the drug always remains within the lipid domains and transports through a lengthy and highly complex pathway attributed by repeated turns and bends as shown in Figure 1 (2B). Various PEs influence the drug transport via this route by temporarily modifying the structural features of the lipid bilayer. In Shunt pathway the drug permeates through the SC via hair follicles or sweat glands. However, considering its lower diffusional area (0.1% of the total skin area), the drug transport contribution through this route is considered very negligible. Ions and large polar molecules have been reported to permeate through this route [15]. The transcellular route is preferred by hydrophilic substances. In general, molecules with amphoteric chemical properties are thought to be able to penetrate the best. However, drugs must have certain criteria which are small molecular weight (below 500Da), moderate lipophilicity (logP ideally around 2-3) and lastly, good solubility both in water and oil so that they are able to permeate the lipophilic stratum corneum and hydrophilic compartment of the systemic circulation [6, 8]. With the aid of many novel approaches and technologies mentioned earlier, the drug delivery through the skin has been enhanced. These inventions have broadened the spectrum for the drug selection instead of being restricted to the criteria set due to the conventional passive diffusion limitations.
Figure 1: Drug transport pathways through the stratum corneum. Reprinted from [15].
Drug Delivery Mechanism of TS:
TS consists of a volatile solvent system which creates a rapidly drying film when sprayed onto the skin [8, 12]. Metered dose transdermal spray (MDTS) system ensures the required volumetric delivery of the dose from its primary packaging material. During application, the volatile solvent will carry the drug into the upper layers of the skin and then evaporates. This action leaves a high concentration of the drug in the skin which acts as the drug reservoir to release the drug continuously and slowly into the circulation [8]. Once the volatile solvent evaporates from the SC layers, it leaves a thin-uniform film of the drug that has a high thermodynamic activity and permeates into the skin rapidly [16]. The Equation below provides the explanation of drug permeation of a TS through the skin.
Msat = Csat × h/10...... (1)
Where, Msat = SC saturation dose, Csat = SC saturation concentration, h = thickness of the SC.
Msat depends on the evaporation rate of the volatile solvent and the depth of penetration of the solvent. If the dose is higher than Msat, the excess drug will remain deposited on the surface of the SC. It will maintain the saturation of the drug within the SC as the drug slowly permeates into the deeper skin layer and enters into systemic circulation. The drug delivered into the skin then act as a depot for the release of the drug into the deeper skin layer.
Transdermal Spray Formulation Components:
Focusing on the components of TS formulation is the main part of product development. The formulation components are, but limited to, drug, volatile solvents, PEs, and film forming polymers (FFP) [4, 14]. Drug molecule in a TS formulation Molecular weight and lipophilicity of the drug are two essential components for drug selection in a TS formulation [17]. Table 1 represents the required criteria for a drug molecule to be fit for any TS formulation [3].
Table 1: Considerations of drug molecules for TS formulationa.
|
Parameter |
Limit |
|---|---|
|
Aqueous solubility |
1mg ml-1 |
|
Lipophilicity |
10 < KO/W < 1000 |
|
Molecular weight |
< 500 Da |
|
Melting point |
< 200 ̊ C |
|
pH of saturated aqueous solution |
pH 5-9 |
|
Dose deliverable |
< 10 mg day-1 |
aReproduced from [3].
All of the above factors are considered to be suitable and ideal for a drug intended to overcome the SC barrier of the skin. Fick's law of diffusion describes the permeation of the drug into the stratum corneum as shown in equation 2. These variables are the important parameters in optimizing the transdermal bioavailability.
Jss = ((D.Ksc/veh.)/h).Cveh = Kp. Cveh..... (2)
Where, Jss = steady state flux (mg/cm2) across a membrane, h = membrane thickness in cm, D = drug diffusivity through the SC (cm2/hr), Ksc/veh = partition coefficient of the drug in the SC and vehicle, Cveh = drug concentration in the vehicle (mg/ ml), Kp = formulation-dependent permeability coefficient of the drug.
From the equation 2, it can be derived that transdermal flux can be modified mainly by three parameters: D, Ksc/veh and Cveh. The structure of lipoidal SC itself is supported by aqueous epidermal layer and this affects the diffusion process. Hence, Ksc/veh value of the drug must favour both transport into and out of the SC. Higher Cveh value may increase the drugs transfer into the skin. Therefore, the solvent must have a sufficiently high holding capacity and at the same time able to aid the transfer process of the drug across the SC [18].
Penetration Enhancer/s Penetration enhancers (PEs) are substances used in the TS formulation in order to increase the transdermal flux of the drugs [19-21]. PE reduces the binding of the drug to the skin and help to deliver the drug across the skin [22]. PE acts by disrupting the SC layer to provide the pathway for the drugs or by increasing the partitioning into the skin and the thermodynamic activity of the drug [7, 23]. Ethanol and propylene glycol are two examples of PEs that can be additionally used as solvent/vehicle for TS formulations [24, 25]. Water, hydrocarbons, acids, amines, amides, esters, surfactants, terpenes, terpenoids, essential oils, camphor, menthol, sulfoxides and lipids have been reported as PEs by many researchers [26, 27]. However, an ideal PE should have the following characteristics [28]:
-
It should be pharmacologically and chemically inert.
-
It should be chemically stable.
-
It should be transparent and generally not volatile in nature.
-
It should give reversible effect on the skin properties after its use.
-
It should be compatible with the components in the formulation and the skin.
-
It should not cause any irritation, sensitization, phototoxicity and comedogenicity.
-
It should be cosmetically acceptable, odorless, tasteless and colorless.
-
Its solubility parameter should be suitable for the formulation and skin.
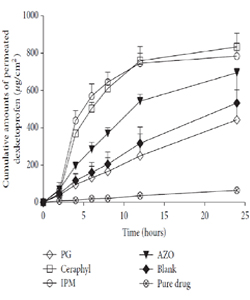
The solubility of a drug in PE has an important role in influencing the amount of transdermal flux. It is hypothesized that PE acts as a vehicle and help increase drug transport through the SC. In fact, the more the drug is solubilized in the vehicle, the higher transdermal flux will be reached [29]. Accordingly, during development of dexketoprofen TS formulation, Lu et al. [14] recorded the solubility of the drug in PE-cum-vehicle in the order of propylene glycol (PG) > isopropyl myristate (IPM) > lauryl lactate (LA) > azone (AZO). However, the ex vivo transdermal flux was found to be the maximum for LA and minimum for PG (Figure 2). The lowest transdermal flux in case of PG has been observed as its inability to form a drug reserThe solubility of a drug in PE has an important role in influencing the amount of transdermal flux. It is hypothesized that PE acts as a vehicle and help increase drug transport through the SC. In fact, the more the drug is solubilized in the vehicle, the higher transdermal flux will be reached [29]. Accordingly, during development of dexketoprofen TS formulation, Lu et al. [14] recorded the solubility of the drug in PE-cum-vehicle in the order of propylene glycol (PG) > isopropyl myristate (IPM) > lauryl lactate (LA) > azone (AZO). However, the ex vivo transdermal flux was found to be the maximum for LA and minimum for PG (Figure 2). The lowest transdermal flux in case of PG has been observed as its inability to form a drug reservoir as it carries the drug along with it during drug transport through the SC [14,30]. Interestingly, blank formulation (containing alcohol as volatile solvent, but no PE) provided much higher amount of transdermal flux of the drug as compared to the group containing pure drug. This might be due to the increased thermodynamic energy of the drug after the evaporation of alcohol. Additionally, ethanol might have provided its permeation enhancement activity [31].
Figure 2: Percutaneous permeation profiles of dexketoprofen MTDS containing different penetration enhancers (mean ± SD; ?? =3). Reprinted from [14].
Volatile Solvents
TS comprises volatile and non-volatile solvents. Volatile solvents are classified as the solvents with vapor pressure higher than 35mm Hg when the body temperature is 32 °C while nonvolatile solvents are those with vapor pressure below 10 mm Hg at the same body temperature [32]. Volatile solvent evaporates after a short period of time, causing an increase in the thermodynamic activity of the drug. Prior to that, the solvent carries the drug into the SC layers, an effect called the solventdrag [8]. To some extent solvent contributes to the permeation of the drug across the SC. That is why solvent selection is also important to optimize the drug delivery. The selected solvent should facilitate the transfer of the drug and have a high drug holding capacity [3].
In most of the TS formulations, alcohols such as alkenols, alkanols, polyglycols, glycols, and glycerols are used as volatile solvents. This group can also act as the penetration enhancers in improving transdermal flux of drugs [26]. Alcohols enhance the drug delivery into the skin by several mechanisms such as increasing the thermodynamic activity of the drug, solventdrag effect, extraction of lipids and proteins and swelling of the SC layers [33]. Bakshi et al. [12] reported that preformulation studies of solvent system for a TS formulation with oxybutynin. Ethanol: acetone: methylal with a ratio of 2:1:2 respectively, resulted desired spray patterns and high dispersibility of the polymers. According to Patel et al. [16], a combination of acetone with alcohol at 1:1 gives a faster film formation for the TS formulation. This finding was supported by Paradkar et al. [34] where the said combination of acetone and alcohol form a film layer in less than 5 minutes and the active ingredients and film forming polymer (FFP) have good solubility in the vehicle. Fast drying film is preferred as to avoid the loss of drug through draining or removal of the drug by transferring to other surfaces [16].
Film Forming Polymers: Film forming polymer (FFP) is associated with an occlusion effect on the skin. The increasing influx of drugs into the skin due to the occlusion effect could be attributed to the following factors [8]:
-
Higher fluidity in intercellular lipid of the SC
-
Changes in the polarity of the intercellular lipid
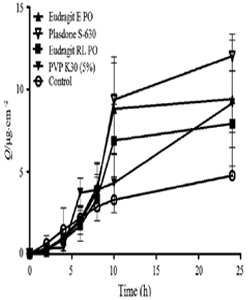
The dissolution of the excess drug on the surface of the skin Eudragit, acrylic polymer, polyvinyl pyrrolidone, ethyl cellulose, etc. are widely used as FFPs in TS formulations [27, 34]. There are few evidences where the incorporation of FFP can increase the transdermal flux of the drugs. Lu et al. [35] observed the above mentioned condition in five testosterone formulations, each containing different FFP. The results showed following trends in terms of transdermal flux: Plasdone S630>Eudragit EPO>PVP K30>Eudragit RL. The transdermal profiles of formulations containing each of these FFPs were higher compared to the control group which did not contain any FFP (Figure 3). This was attributed to the preventive role of the polymer in crystallization of the drugs.
Figure 3: Percutaneous permeation profiles of testosterone MTDS formulations containing different film forming polymers. Reprinted from [35].
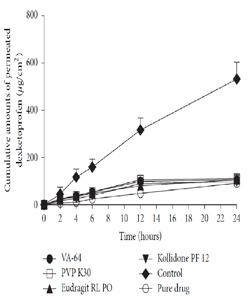
Interestingly, the same research group reported a declining level of transdermal flux compared to control group for dexketoprofen TS formulation as shown in Figure 4 [14]. It was hypothesized that FFP inhibits the penetration of the drug into the skin due to the different drug composition and the incompatibility of the drug with the type or concentration of FFPs in the formulations. That's why, there is a need for further discussion of the effect of FFP on occlusion to differentiate it from the role of solvent groups.
Figure 4:Percutaneous permeation profiles of MTDS of dexketoprofen containing different film forming polymers. Reprinted from [14].
Apart from the permeation enhancing effect, FFP has an important role in improving the uniformity of the spray pattern [16]. It is also responsible for reducing the drying time of the film layer [12, 14]. Other than that, FFPs are associated with the ability to inhibit the crystallization of the drugs as reported by many researchers [35, 36]. An ideal film forming polymer solution should have low viscosity, short drying time (less than 5 minutes) after application and cosmetically attractive. It should produce a flexible film without any sign of cracks or flakes and the film should be completely water washable after its use [37]. These FFPs should be well tolerated by the skin without any kind of compatibility issue.
Characterizations of the TS Formulations:
Developed TS formulations are characterized by various quantitative and qualitative parameters such as viscosity and pH of the formulations, drying time, stickiness, appearance and integrity of skin and water washability, drug content, in vitro drug release, spray pattern and spray angle, average weight per dose, effectiveness of pump seal, stability study etc. [12]. Evaporation time is measured to check the time required to form a dried film. It is determined by measuring the time required to dry the film when sprayed onto a white paper [12]. Spray pattern testing is done to have an idea about the droplet sizes of the sprays. It is checked by measuring the sizes of the colored droplets deposited onto a white paper sprayed from a distance of about 2.5 to 3.0 cm. Formulation is colored with 1% methyl orange for easy visualization of the droplets [34]. Spray angle is measured to check the distribution/spread of the formulation onto the application site. This is carried out by the previous experiment (spray pattern) with the same colored formulation where the white paper is placed horizontally at a distance of 15 cm from the nozzle. The radius of the circle formed on the paper is recorded and Spray angle is calculated from equation 3. Spray angle (?) = tan-1 (h/r) "................3 Where, h is the distance of paper from the nozzle, and r is the average radius of the circle [27]. To check the effectiveness of the pump seal, or otherwise said, to ensure the container free of any leak, the filled containers are positioned up right at 300 and allowed to keep for 3 days before checking any difference in the filled weight [12]. In vitro drug release and ex vivo skin permeation studies of the TS formulation are conducted by Franz diffusion cell with a synthetic membrane (e.g. nylon) and previously processed animal skin samples, respectively.
Recent Works on TS Formulations:
Lopinavir is used in the treatment for HIV infection. When taken orally, it has poor bioavailability (~20%) because of its low aqueous solubility (0.01 mg/ml), poor dissolution rate, and high first-pass metabolism by cytochrome P450 3A4 and P450 3A5 isoenzymes [38]. Oral administration of this anti HIV agent is also associated with several side effects such as headache, tiredness, weakness and gastrointestinal symptoms; diarrhea, mild stomach pain or upset, vomiting and nausea. Patel et al. [16] fabricated an user friendly MDTS of lopinavir with Kollidon ® VA 64 (FFP) and isopropyl myristate (PE). Ex vivo skin permeation and skin deposition studies performed on microporated pig ear skins resulted a significant increase in steady state transdermal flux compared to the plain drug solution and the formulation was found to be non-irritating to the skin. In vivo study in male Wistar rats gave higher bioavailability (almost three folds) compared to the oral route.
Nawaz et al. [39] fabricated a TS formulation Eezpain with a mixture of herbal components (gaultheria oil, eucalyptus oil, turpentine oil, clove oil, menthol and camphor) that posses clinically proven analgesic and anti-inflammatory activity. A combination of propylene glycol, polyethylene glycol (PEG) 400, IPA, tween 20, and benzyl alcohol was used to incorporate other formulation excipients. Applied on 20 human subjects within an age group of 30 to 65 under a prospective, open and non-comparative clinical trial set up, this spray formulation resulted significant improve to the patients suffering from various forms of pain.
Paradkar et al. [34] developed a TS formulation of clotrimazole for its antifungal efficacy up to 12 hours with reduced frequency This antifungal drug is poorly soluble in water and its absorption from the GIT is erratic. It also suffers from various adverse effects like abnormal liver function, nausea, vomiting, mild burning, and irritation after its oral administration. To avoid all these adverse effects, alternative TS formulation is quite rational approach for delivery of this drug [34]. The developed formulation had compositions of propylene glycol (PG) and polyethylene glycol 400 (PEG 400) as plasticizer and solubilizer, various grades of eudragit and ethyl cellulose as film forming polymer, different ratios of ethanol and acetone as volatile solvent. The developed formulations were characterized by viscosity, drying time, stickiness, appearance and integrity of skin and water washability, in vitro drug release, spray pattern and spray angle. Ethanol and acetone at 80:20 ratio together with camphor and menthol (1:1) as PE satisfied the desired criteria of TS formulation with a maximum skin transport of the drug and improved antifungal efficacy.
Helal and Lane [40] reviewed various published research works on the possibilities of transdermal formulation for the drugs of angiotensin converting enzyme (ACE) inhibitors (e.g captopril, enalapril maleate, lisinopril dihydrate, perindopril erbumine, and trandolapril) to avoid their first pass metabolisms, increase patient's compliance and maintenance of a constant plasma drug concentration for a prolong duration. This review revealed that captopril cannot be explored as transdermal formulation due to its large dose (80 mg) and skin irritation property. However, perindopril erbumine, and trandolapril were described as suitable molecules for this alternative dosage form compared to their available tablet and capsule forms.
Desai et al. [41] compared local anesthetic action of lidocaine during dressing change between two topical formulations: Nopayne ®, a lecithin and soybean oil based oil-in-water miniemulsion containing 3% lidocaine and Xylocaine ® (Astra Zeneca), a 4% lidocaine as a lidocaine aqueous solution. A double-blind randomized controlled, pilot trial conducted on 29 patients who were undergoing split thickness skin graft surgery. Both treatments were found equally effective in managing the procedural pain related to the dressing change. However, the emulsion formulation showed a tendency towards a better outcome for pain relief for a longer duration with smaller lidocaine dosage required leading to lower plasma concentrations. This study proved selection of an appropriate solvent system on the efficacy of TS formulations.
Brown et al. [42] reported comparable efficacy of terbinafine topical spray with added patient compliance against commercially available topical solution for Tinea pedis dermatophyte infection. This randomized, observer-blind, comparative study was conducted on 120 patients under Phase IIa clinical trial. The rate of mycological cure after one week of once daily treatment with equivalent to 20 mg of drug per foot was found to be statistically equivalent. However, TS formulation reduces the chance of contamination or transfer of organism as compared to the topical solution.
Modified transport fluconazole TS formulation was reported by Gohel and Nagori [27] for the treatment of tinea infection. Ethyl cellulose and Eudragit ® RS100 were used as FFPs for the fabrication of the spray formulation. The formulation had an eutectic mixture of camphor and menthol as PE cum volatile solvent system to dissolve the formulation composition. The optimized and stable formulation provided increased in vitro permeation of fluconazole through the shed snake skin with drug transport equivalent to its minimum inhibitory concentration (8.0 ± 0.4 μg/ml) in 1 hour study.
Bakshi et al. [12] developed TS formulation of oxybutynin, an antimuscarinic drug, used in the treatment of overactive bladder. The drug has significant anticholinergic side effect such as dry mouth [43]. The developed TS formulation was reported to minimize this side effect [12].
TS Formulation: Market Overview:
The total number of TDD approved by US-FDA from 2000 until 2014 is 39. The majority of them are patches, sprays and emulsion to overcome the hepatic first pass metabolism associated with oral formulations [44]. Around 49% of the approved TDD products are indicated for hormonal therapy followed by analgesic class with 23% occupancy [2]. Oral NSAIDs are associated with gastrointestinal adverse effects which sometimes can be life threatening with prolonged administration. NSAIDs topical formulations are being developed in order to avoid these side effects and reduce the health care cost, morbidity and mortality due to the limitation in using their oral dosage forms [45].
So far, a limited number of TS products are available in the market. A few more are in the late stage of the development [8]. One of the first transdermal products developed was the MDTS Evamist ®, which was approved by the FDA in 2007. It is indicated for the management of menopause-associated vasomotor symptoms [44] and has been proven to be able to achieve therapeutic levels of estradiol in the subjects studied [46]. The next product approved was Testagen ® to treat the low testosterone level in men, in spite of some concerns regarding the safety of this product were raised, especially regarding the third party exposure [11]. Recuvyra ® was approved at a later stage as a pain reliever indicated for dogs [8]. Other molecules that have been approved as TS formulations for commercial use are estrone, testosterone, hydrocortisone aceponate, and lidocaine [9,41]. MedSpray ®, a propellant based topic spray formulation of terbinafine for Tinea pedis dermatophyte infection is under Phase IIa clinical trial [42].
Limitation of TS Formulation:
The stratum corneum has been recognized as the main barrier in TDD. Due to this barrier property, the ranges of selections of drugs are limited; small molecular weight, moderate lipophilicity and good solubility both in water and oil compartment [7,8]. Despite of the high demand of TDD in numerous conditions such as angina, hypertension, motion sickness, smoking cessation and hormone deficiency, the number of drugs intended for TDD specifically in TS form is limited [47]. TS formulations are not suitable for drugs with large doses due to the limited diffusivity of the skin. The review of the transdermal activity of angiotensin converting enzyme (ACE) inhibitors by Helal and Lane [40]. Stated that the physicochemical properties of captopril is not appropriate for transdermal delivery compared to other drugs such as enalapril maleate and lisinopril dehydrate. This is due to the large dose of the drug as the skin can only accommodate 10-20mg dose only. Since captopril can cause irritation when taken orally, the reaction can be much worse when formulated as TDD system. The only solution for captopril to be formulated as TS is by adding antiirritant into the formulation. However, there must be an extensive study done to confirm this theory [40].
Apart from the advantages of TS formulation mentioned earlier from the patient's perspective, it is also a lucrative business from the manufacturer's point of view. This is because of their low cost of manufacturing and developing the formulations [7]. However, TS system faces a major problem due to its mechanism of passive diffusion for drug transport. The only assistance can be the use of chemical penetration enhancers to increase the driving force in delivering the drugs into the skin [48]. There must be an extensive stability and safety studies to be done and the optimization of the formulations should be ensured before the commercialization of the product. TS is a non-occlusive system which will reduce the skin irritation, thus the formulation should focus on reducing or improving the skin irritation attributes caused by the drugs and their excipients. Apart from the third party exposure, particularly in endocrinology indications, safety study should be targeted for the products, especially that are used for long term indications [49].
CONCLUSION:
Drug delivery in TS follows passive diffusion mechanism. There are certain criteria such as appropriate aqueous solubility, lipophilicity, molecular weight, melting point, pH and dose size that a drug must fulfill to exploit full advantage of this mechanism as these criteria are critical to ensure the delivery of the drug across the SC and to achieve the desired therapeutic effect. Due to this reason, there are only a limited number of drugs that can be developed as TS formulations. In order to enhance the transdermal flux, chemical penetration enhancers such as propylene glycol, isopropyl myristate, lauryl lactate and azone have been introduced into the formulations. They act by disrupting the SC layer to provide the pathway for the drugs or by increasing their partitioning into the skin. Volatile solvent present in the TS formulation also contributes to the permeation of drugs through evaporation mechanism. As the solvent evaporates, the thermodynamic activity of the drug is increased and the flux across the SC is enhanced. Last but not least, film-forming polymer (FFP) exerts its effect on the formulation through occlusion mechanism. The results from adding FFPs are varied depending on drugs, not all observe a rise in the drug permeation level. There is a need to do a further study whether the decline of the drug level is due to the incompatibility of FFPs with the drugs or skin or other excipients to optimize the TS formulations. In conclusion, the TS formulations need to be further developed and studied extensively to overcome the SC barrier. Although the path is challenging, but TS formulations have a wide scope to attain a high level of user compliance and therapeutic efficacy.
REFERENCES:
-
Thomas BJ and Finnin BC. (2004). The transdermal revolution. Drug Discovery Today. 9(16), 697-703.
-
Smith EW and Maibach HI. (1995). Percutaneous penetration enhancers. CRC Press.
-
Paradkar M, Thakkar V, Soni T, Gandhi T, et al. (2014). Formulation and evaluation of clotrimazole transdermal spray. Drug development and industrial pharmacy. (0), 1-8.
-
Oldfield V and Plosker GL. (2006). Lopinavir/Ritonavir. Drugs. 66(9), 1275-1299.