Previous Issues Volume 6, Issue 1 - 2023
A Curious Case of Drain Going Intraluminal
Keshavi Mehta1, Prathamesh Pathrikar1, Mohd Shaad Shaikh1, Sanjay Chatterjee2,3,*
1Resident Doctor, Department of General Surgery, Bombay Hospital, New Marine Lines, Mumbai, Maharashtra, India
2Assistant Professor, Maharashtra University of Health Sciences, Nashik, Maharashtra, India
3Postgraduate Teacher, Bombay Hospital Institute of Medical Sciences, Maharashtra, India
*Corresponding author: Sanjay Chatterjee, Postgraduate Teacher, Chamber no. 108, Bombay Hospital Institute of Medical Sciences, Bombay Hospital Avenue, 12-New Marine Lines, Mumbai, Maharashtra, India 400020; Tel: +919322243938; Email: [email protected]
Received Date: January 09, 2023
Publication Date: January 25, 2023
Citation: Mehta K, et al. (2023). A Curious Case of Drain Going Intraluminal. Mathews J Surg. 6(1):17.
Copyright: Mehta K, et al. © (2023)
ABSTRACT
Anastomotic leaks are among the most dreaded complications after intestinal surgeries. Some leaks presents in a dramatic fashion early in the postoperative period, leaving little doubt about the diagnosis. However, many others present in a far more subtle fashion, often relatively late in the postoperative period, and can be difficult to distinguish from other postoperative infectious complications. We report a case of a 66 year old male who presented with bleeding per rectum since 3-4 months. He had a history of being operated for hemorrhoidectomy 9 years ago. He underwent a colonoscopy which detected a large sessile polyp at the rectosigmoid junction for which polypectomy was done. The histopathology of the polyp revealed moderately differentiated adenocarcinoma arising within the villoglandular polyp (malignant polyp) with base involved. He was then operated for anterior resection followed by colorectal anastomosis. Until postoperative day 5, the patient had no symptoms and signs of having an anastomotic leak, after which he complained of extreme abdominal discomfort, sudden distension and the drain had collected 35ml of feculent matter. It is a rare and poorly understood case of anastomotic leak presenting without high fever, tachycardia, a rigid abdomen and hemodynamic instability.
Keywords: Anastomotic leak, intra-abdominal drain
INTRODUCTION
Surgeons are all too familiar with the potentially devastating consequences of an anastomotic leak. Patients classically develop agonizing abdominal pain, tachycardia, high fevers, and a rigid abdomen, often accompanied by hemodynamic instability. In these cases, urgent return to the operating room for peritoneal washout and fecal diversion is generally required; prolonged stays in the intensive care unit and death are not uncommon. The mortality rate for an anastomotic leak in the literature typically is in the 10% to 15% range. Further, anastomotic leakage has been associated with increased local recurrence and diminished survival after colorectal cancer surgery. However, a large number of patients ultimately found to have an anastomotic leak develop a more insidious presentation, often with low-grade fever, prolonged ileus, or failure to thrive. In these patients, making the diagnosis may be much more difficult as the clinical course is often similar to other postoperative infectious complications. Radiologic imaging is usually required; even then, the diagnosis may be elusive or at least uncertain.
Intestinal ischemia occurs when there is at least 75% reduction in intestinal blood flow for more than 12 hours. Abdominal pain is the most common symptom in patients with intestinal ischemia. Patients characteristics, such as age over 60 years, not appearing severely ill, mild abdominal pain, tenderness, rectal bleeding or bloody diarrhoea are the features that are more common in acute colonic ischemia.
CASE REPORT
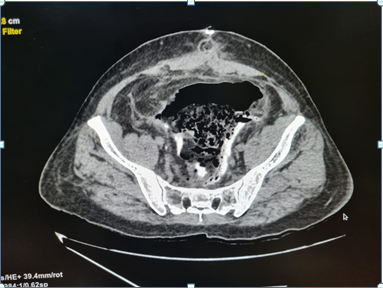
A 66 year old male, known case of hypertension and ischemic heart disease, presented to our clinic with complains of bleeding per rectum since 3-4 months. The bleeding was insidious in onset and occurred as streaks with the stools. There was no other positive history. The patient had been previously operated of hemorrhoidectomy 9 years ago, laparoscopic cholecystectomy 7 years ago and coronary angiography with stunting 3 years ago. On examination, there were no other positive findings. The patient was advised colonoscopy which revealed internal hemorrhoids and a large sessile polyp at the rectosigmoid junction for which polypectomy was done in the same sitting. The histopathology of the polyp revealed moderately differentiated adenocarcinoma (malignant polyp) with base involved. The patient was admitted and PET scan was done which revealed: smooth rectal wall thickening with focal increased metabolic activity noted. The patient was thus operated for anterior resection with descending colon with mid rectal anastomosis and the specimen was sent for histopathology. Pelvic drain was kept intraoperative. The patient passed flatus and motions and the foleys catheter was removed on post-operative day 3 and the patient was allowed liquid diet on day 4. On the postoperative day 5 when he complained of extreme discomfort in the abdomen, non-passage of flatus and stools, sudden abdominal distension, sluggish bowel sounds, feculent collection in the drain and soakage at lower suture line. The patient was kept nil by mouth, rules tube was inserted and aspiration was done continuously, iv fluids had been started, all the investigations had been done which mainly included an abdominal X-ray, complete blood count and serum electrolyte levels. All the investigated parameters were within normal limits except low serum potassium levels. X-ray (abdo+pelvis) showed signs of small bowel obstruction. Till postoperative day 7 electrolyte correction was done and the patient was managed conservatively. On post-operative day 8, ultrasonography of the abdomen and pelvis was done which was suggestive of small bowel obstruction without any localized collection. This was followed by a CECT scan (abdo+pelvis) on post-operative day 9 which revealed high suspicion for an ischemic bowel injury along with the drain being seen intraluminal. The patient was thus taken up for re-exploration and stoma creation. Intraoperatively, it was found that there was a collection of fecal matter inside the pelvic cavity and on further exploration sigmoid colon was discovered be gangrenous, And the drain was found intraluminal. This segment on histopathology revealed gangrenous necrosis of the sigmoid colon. The gangrenous bowel caused the drain to go intraluminal. Hartman’s procedure and Descending colostomy was done. The patient was shifted to the ICU postoperatively. After constant observation, monitoring of vitals, urine output, drain output and care, ryles tube aspiration, stoma health, correction of potassium and magnesium levels, starting of total parenteral nutrition, the patient was allowed liquid diet on post-operative day 5. Foleys catheter and ryles tube was removed on post-operative day 8. Patient was started on soft high protein diet on post-operative day 9 followed by full diet after the stoma became functional. The patient being vitally and clinically stable was discharged on post-operative day 11 [1-12] (Figures 1-3).
Figure 1
Figure 2
Figure 3
DISCUSSION
Abdominal drain use is a method that has been implemented for many years, its use after every surgical procedure has become an issue of controversy. In a meta-analysis published in 2004 on the use of prophylactic drain use in surgical procedures of the GI system, the authors argued that many GI procedures could be performed safely without prophylactic drains and there was a need for novel rules on prophylactic drain use for many centers. Many surgeons prefer to use drains because of the high risk of anastomotic leaks, in order to facilitate early diagnosis of leaks and to expedite their recovery, to decrease the frequency of postoperative collection and abscesses, and because of the fact that these leaks are potentially life-threatening.
Castellone JA, et al. in his study mentioned that the presentation of ischemic bowel disease is frequently subtle, especially in the elderly. Abdominal pain is the most common presenting chief complain in such patients. Early in the presentation, pain is nonspecific and out of proportion to the physical findings, which can confound the correct diagnosis. Similarly in our case, the patient presented with extreme pain while being vitally stable and all the investigating parameters being normal.
CONCLUSION
In conclusion, the intraluminal migration of the drain placed into the abdomen following GI surgery is rather rare. Ischemic bowel leading to gangrene is one of the rare causes for the drain to be seen intraluminal in the scans and intraoperatively. Endoscopy and contrast imaging can prove to be useful diagnostic tools. Overall, using an aggressive diagnostic approach will reduce mortality drastically. The most important prognostic factor in such cases relies on the time interval between onset of symptoms and definitive treatment.
LESSONS LEARNT
This case highlights the importance of prompt recognition and subsequent management of bowel gangrene causing . High index of suspicion is needed to avoid delay in diagnosis of this condition. Early CT scans to confirm the postoperative status of the drain and bowels is optimum to avoid such dire consequences.
CONFLICT OF INTEREST
None.
REFERENCES
- Golub R, Golub RW, Cantu R, Stein HD. (1997). A multivariate analysis of factors contributing to leakage of intestinal anastomosis. J Am Coll Surg. 184:364–372.
- Jex RK, Van Hcerden JA, Wolff BG, Ready RL, Ilstrup DM. (1992). Gastrointestinal anastomoses: factors affecting early complications. Ann Surg. 206:138–141.
- Walker KG, Bell SW, Rickard MJ, Mehanna D, Dent OF, et al. (2004). Anastomotic leakage is predictive of diminished survival after potentially curative resection for colorectal cancer. Ann Surg. 240:255–259.
- Pickleman J, Watson W, Cunningham J, Fisher SG, Gamelli R. (1999). The failed gastrointestinal anastomosis: an inevitable catastrophe? J Am Coll Surg. 188:473–482.
- Bruce J, Krukowski ZH, Al-Khairy G, Russell EM, Park KG. (2001). Systematic review of the definition and measurement of anastomotic leak after gastrointestinal surgery. Br J Surg. 88:1157–1168.
- Alves A, Panis Y, Pocard M, Regimbeau JM, Valleur P. (1999). Management of anastomotic leakage after nondiverted large bowel resection. J Am Coll Surg. 189:554–559.
- Alves A, Panis Y, Trancart D, Regimbeau JM, Pocard M, Valleur P. (2002). Factors associated with clinically significant anastomotic leakage after large bowel resection: multivariate analysis of 707 patients. World J Surg. 26:499–502.
- Eckmann C, Kujath P, Schiedeck TH, Shekarriz H, Bruch HP. (2004). Anastomotic leakage following low anterior resection: results of a standardized diagnostic and therapeutic approach. Int J Colorectal Dis. 19:128–133.
- Hosgood G. (1990). The history of surgical drainage. J Am Vet Med Assoc. 196:42–44.
- Petrowsky H, Demartines N, Rousson V, Clavien PA. (2004). Evidence based value of prophylactic drainage in gastroin- testinal surgery: systematic review and meta-analyses. Ann Surg. 240:1074–1084.
- Carlomagno N, Santangelo ML, Grassia S, La Tessa C, Renda A. (2013). Intraluminal migration of a surgical drain. Re- port of a very rare complication and literature review. Ann Ital Chir. 84:219–223.
- Subhash R, Ali IV, Natesh B. (2013). Intraluminal migration of drain tube: a short report. Ann Gastroenterol. 26:93–95.