Previous Issues Volume 2, Issue 1 - 2017
L-carnitine Partially Improves Metabolic Syndrome Symptoms but does not Reverse Perturbed Sperm Function or Infertility in High Fat Diet-Induced Obese Mice
Mayumi Nagata1, Toshikazu Suzuki1,2
1Department of Health and Nutrition and 2Graduate School of Human Ecology, Wayo Women's University, 2-3-1 Konodai, Ichikawa, Chiba 272-8533, Japan.
Corresponding Author: Toshikazu Suzuki, Department of Health and Nutrition, Graduate School of Human Ecology, Wayo Womens University, 2-3-1 Konodai, Ichikawa, Chiba 272-8533, Japan, Tel: +81-43-371-1547; E-Mail: [email protected]
Received Date: 08 Mar 2017 Accepted Date: 20 Mar 2017 Published Date: 27 Mar 2017
Copyright © 2017 Suzuki T
Citation: Nagata M and Suzuki T. (2017). L-carnitine Partially Improves Metabolic Syndrome Symptoms but does not Reverse Perturbed Sperm Function or Infertility in High Fat DietInduced Obese Mice. Mathews J Nutr Diet. 2(1): 013.
ABSTRACT
In recent years, adverse effects of obesity on female reproduction and male fertility have received significant attention. In this study, we investigated infertility improvements following L-Carnitine treatment using a high-fat diet (HFD)-induced obesity mouse model.
Five-week-old male mice were divided into a control diet (CTD) group, a HFD group, and a HFD with L-Carnitine supplementation (HFD+C) group that received L-Carnitine-supplemented water and a HFD. At 30 weeks of age, mating tests with female mice were conducted to investigate reproductive ability. Dissection was then performed to analyse organ weights, blood glucose and lipid levels, and sperm motility.
These results suggest that L-Carnitine administration does not reverse male infertility in diet induced obesity but partially improves metabolic syndrome symptoms.
KEYWORDS
Diet-Induced Obesity; Male Infertility; Sperm Motility; L-Carnitine.
INTRODUCTION
It is currently estimated that one out of every six married couples in Japan have undergone infertility treatment and testing [1], indicating that this is a social problem. Infertility is defined by the World Health Organization as "inability to achieve pregnancy after 12 months of unprotected sexual intercourse" [2]. According to the National Fertility Survey of birth trends conducted by the National Association of Population and Social Security Research in 2002, 12.1% of Japanese married couples had experienced infertility testing and treatment. This figure increased to 18.2% in the 2015 Survey [1]. Research has demonstrated that 41% of infertility cases can be traced to problems with the woman, 24% to problems with the man, 24% to problems with both partners, and 11% unknown [3]. This indicates that nearly half of infertility cases can be traced to problems with the man. Biological disorders that cause infertility in women include fallopian tube disorders, anovulation, and cervical disorders and in men include hypo spermatogenesis, seminal duct obstruction, accessory organ disorders, and sexual dysfunction [4, 5]. Other factors related to infertility include lifestyle-related causes, such as alcoholic beverage consumption, smoking, stress, obesity, and excessive dieting [6-8].
In Japan, obesity is defined as a body mass index (BMI) of 25 kg/m2 and above. According to the international standard established by the World Health Organization, a BMI of 25 kg/m2 and above is considered 'overweight' while a BMI of 30 kg/m2 and above is considered "obese" [9]. Obesity is caused by energy intake habitually exceeding energy con- sumption. Currently, 2.1 billion people, or 29% of the worlds population, are overweight or obese [10], indicating that this is a global problem. In Japan, although the percentage of obese adult women is on the decline, the percentage of obese adult men increased to 29.5% in 2015 from 20.4% in 1987 [11, 12]. When the data for people of reproductive age were examined by age group, 26.6% of men in their twenties, 30.3% of men in their thirties, and 36.5% of men in their forties were either overweight are obese [12]. Studies investigating the correlation between obesity in women and infertility reported that obesity caused declines in fertility rate and made women more susceptible to menstrual disorders. Furthermore, these studies revealed that overweight women had lower ongoing pregnancy rates than women at their ideal weight [7, 13, 14]. Investigation of the correlation between obesity in men and infertility revealed that obese men have fewer sperm, lower sperm motility, and a higher percentage of sperm with DNA fragmentation [15-17]. The majority of these studies were clinical studies conducted in humans.
Research using animals has revealed that in ob/ob mice that have mutations in the gene encoding leptin, obesity was accompanied by male infertility. Furthermore, although dietary restrictions in these mice did not lead to weight reduction, leptin supplementation led to recovery of fertility because of weight loss [18]. Heterozygosity in male mice with lethal yellow (Ay) mutation was associated with reduced fertility with aging and increased leptin tolerance [19]. Ghanayem et al. reported sperm quality deterioration and male infertility in mice with diet induced obesity [20]. Recently, Fan et al. reported that the integrity of the blood-testis barrier was interrupted with reduced expression of tight junction-related proteins, occludin, ZO1, and androgen receptor, in addition to decreased fertility in high-fat diet (HFD)-fed obese mice [21]. However, many aspects regarding these issues remain unclear.
Studies investigating supplements with the objective of improving male infertility include those that have examined clomiphene citrate, vitamin E, and coenzyme Q10, among other substances [22, 23]. Of these, many studies have examined the efficacy of L-carnitine, which activates fatty acid metabolism and is commonly sold as a dietary supplement. Previous research revealed that it improved sperm motility and forward progression and led to improved fertility rates [24].
Research that aimed to improve infertility associated with obesity reported that weight loss and exercise therapy were effective in improving obesity [25]. However, few studies have analyzed the effect of supplements and nutritional components on infertility associated with obesity. Thus, we investigated the use of supplements as an anti-obesity and infertility treatment and investigated the efficacy of oral ingestion of potable water com bined with L-carnitine in the improvement of male infertility.
MATERIALS AND METHODS
Materials
Animals
C57BL/6J male mice (4 weeks old) and B6C3F1 female mice (8 weeks old) were purchased from CLEA Japan, Inc. (Tokyo, Japan).
Diets
The standard control diet (CTD, 10% of energy derived from fat; D12450B; 3.85 kcal/g) and HFD (60% of energy derived from fat; D12492; 5.24 kcal/g) were purchased from Research Diets, Inc. (New Brunswick, NJ, USA).
Diets
L-carnitine tartrate was provided by Mr. Satoshi Oudo of Lonza Japan Co., Ltd (Tokyo, Japan). M2 medium (M7167) was purchased from Sigma-Aldrich Japan Corporation (Tokyo, Japan). Triglyceride (432-40201) and Cholesterol E-tests (439-17501) were purchased from Wako Pure Chemical Industries, Ltd. (Osaka, Japan). All other chemicals used were of the highest commercially available purity.
Bleeding
Four-week-old male mice were preliminarily monitored for one week and then switched to experimental feed and monitored for 25 weeks (until 30 weeks of age). Breeding was performed in an environment containing 5 to 6 animals per cage at room temperature (20-25°C) with a humidity of 50-70% and light/dark time control (7:00 to 19:00). Mice were divided into three groups: CTD (N=10), HFD (N=10), and HFD + L-carnitine groups (HFD+C, N=11). For the HFD+C group, drinking water was switched from tap water to 1% L-carnitine water after 14 weeks of breeding (18 weeks of age) in addition to HFD feeding. Animals were fed ad libitum, and food and drinking water were exchanged three times per week. Body weights were measured once per week using an animal weighing balance system (LIBROR EB-3200S-A, 3,200 g capacity and 0.1 g, readability, Shimadzu Corporation, Kyoto, Japan). This experiment was conducted according to the "Ethics Committee Regulations on Experimental Research for Animals at Wayo Womens University" under the review of and approval by the Ethics Committee (Reception Number 1104, 1221).
Mating Test
From the CTD, HFD, and HFD+C groups, five random male mice were used for the mating test. Eight-week-old female mice were preliminarily monitored for one week and then used for the mating test. Individually housed mice (30 weeks of age) were cohabited with two virgin female mice (9 weeks of age) for 5 days, after which males and females were separated. Approximately 13 days after the last day of cohabitation, each female was euthanized by cervical spine fracture under isoflurane aspiration anesthesia. After confirming the death, the abdomen was opened, the uterus was excised and opened, and the number and viability of fetuses were carefully examined. The fetuses were euthanized under isoflurane aspiration anesthesia after confirmation of viability.
Blood Sampling and Removal of Organs
Mice were fasted for 18 hours immediately before the end of the breeding period. Whole blood was then collected by cardiac puncture after laparotomy under isoflurane aspiration anesthesia. After euthanasia, the liver, kidney, testis, and visceral fat (perirenal, peritesticular, and mesenteric fat) were removed, and organ and visceral fat weights were measured.
Fasted Serum Glucose (GLC), Cholesterol, and Triglyceride (TG) Measurements
Whole blood was allowed to clot at room temperature and then centrifuged at 1,700 × g for 10 min to harvest the serum. Serum GLC, TG, total cholesterol (TC), and high-density lipoprotein cholesterol (HDL-C) concentrations were measured using a biochemical automatic analyzer (Dri-Chem 4000, Fuji Film Medical, Tokyo, Japan) and specimen-specific slides. Quantification of serum components was performed using the calibration curve included in the instrument.
Sperm Harvest, Sperm Motility, and Progressive Measurements
Peritesticular fat was removed from the epididymis, which was then immersed in 1 mL of M2 medium. After adhering fat was carefully removed, the cauda epididymis was excised, and the new M2 medium was transferred to a 1 mL container. Spermatozoa were released into the M2 medium by making a tiny cut in the cauda epididymis with a micro shear blade and squeezing.
After gently stirring with a microspatula, the supernatant was retrieved with a micropipette and transferred to a 1.5 mL tube. Computer-assisted sperm analysis (CEROS, Hamilton Thorne, Inc., Beverly, MA, USA) was used to obtain quantitative parameter values for sperm motility. Sperm dispersed in M2 medium was loaded into a computer-assisted sperm analysis assay chamber (Hamilton Thorne, Inc.). Sperm tracks (1.5 s, 30 frames) were captured at 60 Hz and analyzed using HTM-CEROS Version 12.3 software (Hamilton Thorne, Inc.). The parameters during measurements were: minimum contrast, 30; minimum cell size, 4 pixels; straightness threshold, 50.0%; path velocity cut off, 10.0 μm/s; progressive minimum path velocity, 50 μm/s; static head size, 0.13 to 2.43; static head intensity, 0.10 to 1.52; and static elongation, 5 to 100.
Liver Lipid Analysis
The liver sample was shredded with scissors until it became pasty. The sample (0.5 g) was then transferred to a glass Teflon homogenizer, and 4.5 mL of physiological saline was added. After homogenizing for a minimum of 10 strokes, 2 mL of homogenate was transferred to a 40-mL lidded glass centrifuge tube. Methanol (3.3 mL) was added and stirred for 5 s × 3 times with a vortex mixer. Chloroform (6.7 mL) was then added and stirred with a vortex mixer for 5 s × 3 times. After 5 s × 3 stirring operations were repeated 12 times (1 hour) every 5 min, centrifugation was performed at 2,000 rpm/min at 4°C for 10 minutes. The organic phase (3 mL of the lower layer) was transferred to a 10-mL lidded test tube, and the 1 organic solvent was evaporated in the draft chamber. After evaporation, 1 mL of 2-propanol solution containing 10% Triton-X 100 was added to the test tube, and the lipid was dissolved. For quantification of TG and TC levels, Triglyceride and Cholesterol E-tests (Wako) were used, respectively.
Statistical Analysis
Results are expressed as mean ± SD. After Students T test was used to assess significance, Holms multiple comparison test was used [26]. Chi square test was used for fertility and fetal life and death analyses. A value of p <0.05 was considered statistically significant. Microsoft Excel 2010 and R were used for statistical processing.
RESULTS
Comparison of CTD and HFD mice and the anti-obesity effect of L-carnitine supplementation Estimated total energy intake, estimated L-carnitine intake, and body weight
Because the mice were reared together (5 to 6 mice per cage), the mean total energy intake per mouse during the rearing period was estimated by dividing the total energy intake in the total amount of feed provided to each cage during the rearing period by the number of mice in the cage. The mean total energy intakes of the HFD and HFD+C mice were approximately 1.2 times that of the CTD mice (Table 1). The estimated L-carnitine intake of HFD+C mice was calculated by dividing the total amount of water by the number of animals being reared in each cage. The estimated L-carnitine intake per mouse was 30.1 mg/day or 0.6 mg/g/day by weight. The total estimated L-carnitine intake per mouse over a 12-week period was 2538.6 mg. HFD mouse weights at the end of the rearing period were approximately 1.6 times those of the CTD mice.
Table 1: Mean food intakes, weights, and weight increments.
| CTD | HFD | HFD+C | |
|---|---|---|---|
| Food intake (kcal) | 1896 | 2261 | 2291 |
| Baseline body weight (g) | 20.9±0.7 | 20.1±0.4 | 20.2±0.3 |
| Body weight at 18 weeks (g) | 30.2±1.5a | 47.2±3.8b | 48.0±2.0b |
| Final body weight (g) | 31.4±1.8a | 50.1±2.9b | 47.4±2.9c |
| Weight increment during feeding experiment (g) | 10.5±2.3a | 29.9±3.3b | 27.4±3.0b |
Values except food intake are means ± SD. Means in a row with different letters differ significantly (p<0.05).
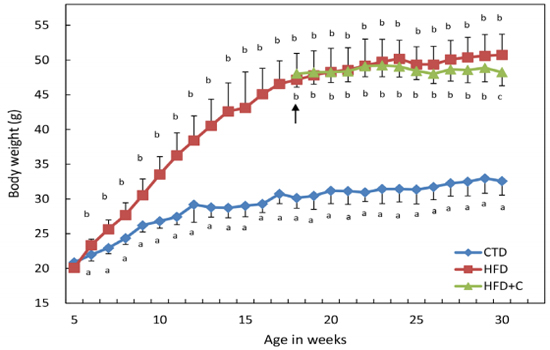
HFD mouse weights were significantly increased compared with those of CTD mice at 6 weeks of age. HFD+C mouse weights increased more gently compared with those of HFD mice from the start of L-Carnitine administration, and by 30 weeks of age, this difference became statistically significant (Figure-1).
Figure 1: Average body weight change during the feeding experiments. Mice were divided into three groups and fed diets as described in Materials and Methods. HFD-fed mice were divided into two groups at 18 weeks of age. The HFD group continued to receive tap water as drinking water. The HFD+C group received 10% (w/v) Carnitine-containing water as drinking water from 18 to 30 weeks of age. The arrow indicates when HFD-fed mice were divided into the two groups. Body weights of each animal were measured weekly. Data are means ± SD for 12 mice/group. For the HFD group from ages 5 to 17 weeks, the data are from 24 mice. Means with different letters in the same week differ significantly (p<0.05).
Organ Weights and Amount of Accumulated Fat
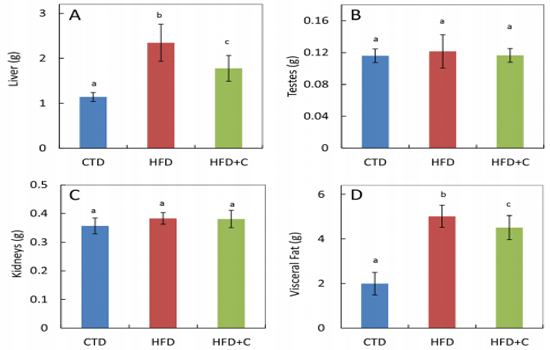
Liver weights in HFD mice at the end of the rearing period were approximately 2.1 times those of the CTD mice. In HFD+C mice, liver weights were approximately 0.8 times those of HFD mice (Figure 2). There were no differences between the three groups in testis weights (Figure 2). Visceral fat weights (combined total of perirenal, peritesticular, and mesenteric fat) in HFD mice were approximately 2.5 times those of CTD mice. In HFD+C mice, visceral fat weights were approximately 0.9 times those of HFD mice (Figure 2).
Figure 2: Liver (A), testes (B), kidney (C), and visceral fat (D) weights at 30 weeks of age. Data are means ± SD for 12 mice/group. Means with different letters differ significantly (p<0.05).
Blood GLC, blood lipid, and hepatic lipid levels
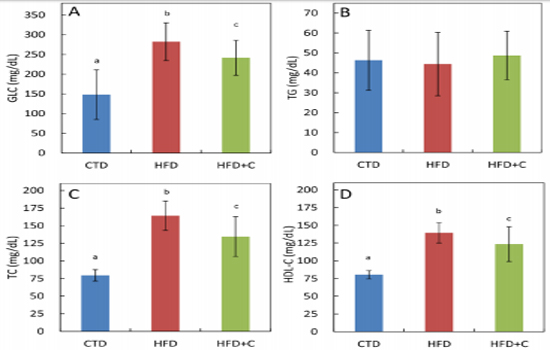
Blood GLC levels in HFD mice were approximately 1.9 times those of CTD mice. Although there was no significant difference between HFD+C mice and HFD mice, there was a trend toward mitigation of HFD-induced blood glucose levels in the HFD+C group (Figure 3). There were no significant differences between the three groups in blood TG levels (Figure 3). TC levels in HFD mice were approximately 2.1 times those of CTD mice. In the HFD+C mice, TC levels were 0.8 times those of HFD mice (Figure 3). Blood HDL-C levels in HFD mice were approximately 1.7 times those of CTD mice. There was no significant difference between the HFD+ C and HFD mice (Figure 3). TG levels per 1 g of liver in HFD mice were approximately 2.6 times those of CTD mice
Figure 3: Serum glucose (GLC) (A), triglyceride (TG) (B), total cholesterol (TC) (C), and HDL16 cholesterol (HDL-C) (D) levels at 30 weeks of age. Data are means ± SD for 12 mice/group. Means with different letters differ significantly (p<0.05).
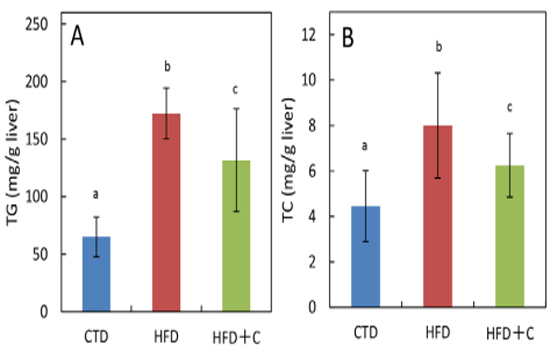
In HFD+C mice, TG levels were approximately 0.8 times those of HFD mice (Figure 4). TC levels per 1 g of liver in HFD mice were approximately 1.8 times those of CTD mice. In HFD+C mice, TC levels were approximately 0.8 times those of HFD mice (Figure 4).
Figure 4: Hepatic triglyceride (TG) (A) and total cholesterol (TC) (B) contents at 30 weeks of age. Data are means ± SD for 12 mice/group. Means with different letters in the same week differ significantly (p<0.05).
Fertility Male Mouse Fertility
Male mouse fertility was determined by confirming via autopsy the presence of embryos in female mice with whom males had mated. The total numbers of fertile male mice were as follows: 5/5 CTD mice, 0/5 HFD mice, and 1/5 HFD+C mice. No fertility was observed in male HFD mice (Table 2).
Table 2: Summary of mating experiment.
| CTD | HFD | HFD+C | |
|---|---|---|---|
| Fertile/total number of male mice | 5/5 | 0/5 | 1/5 |
| Pregnant/total number of mated female mice | 8/10 | 0/10 | 2/10 |
Number of embryos and percentage of live embryos
The numbers of impregnated females by experimental males were 8 of 10 impregnated by CTD mice and 2 of 10 impregnated by HFD+C mice. No female mice were impregnated by male HFD mice (Table 2). The mean number of live embryos per embryos in a single females uterus was 8.1±1.4 in CTD mice and 8.5±0.7 in HFD+C mice (Table 3). The mortality rate for embryos was 3% (2 of 67) in CTD mice and 6% (1 of 18) in HFD+C mice. There was no difference between the CTD mice and the HFD+C mice in the percentage of live embryos.
Table 3: Summary of mating experiment.
| CTD | HFD | HFD+C | |
|---|---|---|---|
| Number/litter | 8.1 ± 1.3 | 0 | 8.5 ± 0.5 |
| Pregnant/total number of mated female mice | 8/10 | 0/10 | 2/10 |
Values are means ± SD.
Sperm motility and forward progression
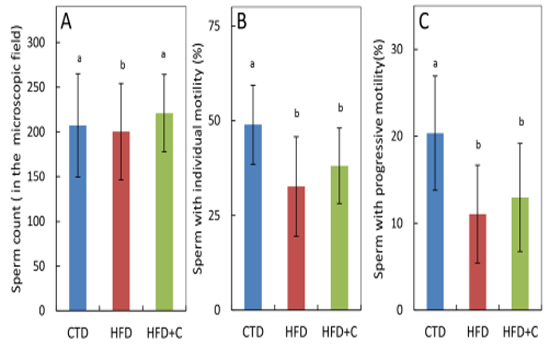
Sperm motility and forward progression were measured using sperm extracted from mouse epididymides during autopsies. There were no significant differences among the three groups in number of sperm in the visual measurement field (Figure 5). Mean sperm motility in HFD mice was approximately 0.7 times that of CTD mice. Mean sperm forward progression in HFD mice was approximately 0.5 times that of CTD mice (Figure 5).
Figure 5:Sperm count (A), sperm motility (B), and progressive sperm motility (C) of cauda epididymal sperm samples at 30 weeks of age. Preparation of sperm samples and determination of quantitative parameters of sperm motility were performed as described in Materials and Methods. Sperm countwas determined by the number of sperm in the microscopic field recognized by the sperm analyzer software. Data are means ± SD for 12 mice/group. Means with different letters in the same week differ significantly (p<0.05).
Both sperm motility and forward progression of HFD+C mice were 1.2 times those of HFD mice. Although L-carnitine caused a slight recovery of motility, these observations were not statistically significant. The percentage of motile sperm and sperm forward progression in the single HFD+C mouse that were fertile was 21.0% and 7.8%, respectively. Sperm motility in this mouse was not high (Table 4).
DISCUSSION
In this study, we investigated fertility in HFD-induced obese male mice to clarify the mechanisms underlying male infertility associated with obesity. We also investigated the efficacy of Lcarnitine, which has been reported to be effective in improving male infertility in humans and obese male mice with reduced fertility
HFD-fed mice displayed significant increases in body weight, liver and visceral fat weights, and blood GLC, blood TG, blood HDL-C, hepatic TG, and hepatic TC levels compared with those of CTD mice that were administered a standard diet (Table 1, Figures 2, 3, and 4). These findings confirmed obesity and the blood biochemical changes that occur in association with obesity. No changes were observed in blood TG levels or testes and liver weights. Sperm motility, sperm forward progression, and fertility of HFD mice were significantly reduced compared with those of CTD mice (Figure 5). These results are consistent with the results of a previous study conducted by Ghanayem et al., which indicated that obesity caused decreased sperm quality and male infertility [20]. Although no fertility was observed in HFD mice under the conditions of the present study, Ghanayem
Table 4: Sperm motility and number of fetuses in female mice mated with fertile male mice.
| Group | ID of male mice | Sperm with mortility (%) | Sperm with progressive motility (%) | ID of mated female mice | Existence of fetuses | Number of fetuses | |
|---|---|---|---|---|---|---|---|
| Live | Dead | ||||||
| CTD | |||||||
| CTD-01 | 41.8 | 13.8 | F01 | Yes | 8 | 1 | |
| F02 | Yes | 9 | 0 | ||||
| CTD-02 | 57.8 | 24.5 | F03 | Yes | 10 | 0 | |
| F04 | Yes | 7 | 0 | ||||
| CTD-03 | 47.7 | 22.8 | F05 | Yes | 7 | 1 | |
| F06 | Yes | 10 | 0 | ||||
| CTD-04 | 58.6 | 26.2 | F07 | Yes | 7 | 0 | |
| F08 | No | - | - | ||||
| CTD-05 | 57.8 | 24.5 | F09 | No | - | - | |
| F10 | Yes | 7 | 0 | ||||
| HFD | |||||||
| HFD-01 | 44.9 | 16.2 | F11 | No | - | - | |
| F12 | No | - | - | ||||
| HFD-02 | 22.1 | 7.8 | F13 | No | - | - | |
| F14 | No | - | - | ||||
| HFD-03 | 46.2 | 19.3 | F15 | No | - | - | |
| F16 | No | - | - | ||||
| HFD-04 | 37.7 | 18.2 | F17 | No | - | - | |
| F18 | No | - | - | ||||
| HFD-05 | 4.3 | 1.8 | F19 | No | - | - | |
| F20 | No | - | - | ||||
| HFD+C | |||||||
| HFD+C-01 | 29.3 | 10.6 | F21 | No | - | - | |
| F22 | No | - | - | ||||
| HFD+C-02 | 41 | 19.5 | F23 | No | - | - | |
| F24 | No | - | - | ||||
| HFD+C-03 | 21 | 7.8 | F25 | Yes | 9 | 0 | |
| F26 | Yes | 8 | 1 | ||||
| HFD+C-04 | 49.2 | 15.5 | F27 | No | - | - | |
| F28 | No | - | - | ||||
| HFD+C-05 | 52.6 | 20.2 | F29 | No | - | - | |
| F30 | No | - | - | ||||
et al. reported 12% fertility in their HFD mice. In their experiment 71 male HFD mice were used for mating, while in the present study only five male HFD mice were used for mating. Thus, we believe our smaller sample size may have affected our results. Future studies with larger numbers of animals in each group may result in identifying fertile males in the HFD group, although in small percentages.
In this study, we also investigated whether L-carnitine was effective in improving fertility in male HFD mice. L-carnitine transports long-chain fatty acids into mitochondria and promotes fatty acid metabolism [24]. Compared with HFD mice, HFD+C mice displayed reduced body weight, visceral fat, hepatic weight, and blood TC, hepatic TG, and hepatic TC levels (Table 1, Figures 2, 3, and 4). Although we observed a tendency toward improved blood GLC and HDL-C levels, these trends were not statistically significant. Similarly, we observed a tendency toward reduced sperm motility and forward progression that were also not statistically significant (Figure5). One in five male HFD mice which received L-carnitine was determined to be fertile, however, this did not represent a statistically significant difference. Compared with HFD and other HFD+C mice, sperm motility and sperm forward progression percentages in the single fertile HFD+C mouse were not high. This indicates that from the point of view of motility, sperm quality was not improved in this case (C03 in Table 4). Based on this result, it is difficult to conclude that the fertility observed in one HFD+C mouse was the result of L-carnitine. Besides, diet-induced obesity raises a risk of sperm DNA damage and a ratio of teratozoospermia [15, 21], those could not be analyzed in the present study because of our limited research environment. Future studies involving these issues with larger numbers of animals would be required. men with asthenospermia. They reported that although sperm motility in healthy men with phospholipid-hydroperoxide glutathione peroxidase (PHGPx) activity in their testes improved during the L-carnitine administration period, it did not improve in men with low PHGPx activity in the testes [27]. Therefore, we believe that for L-carnitine to result in improved sperm motility, it is necessary for the subject to have normal PHGPx activity in the testes. There are no reports investigating the correlation between obesity and PHGPx activity in testes. In the present study, decreased sperm motility because of obesity was not clearly improved by L-carnitine administration. L-carnitine has been used as a supplement to control obesity, and in recent years, it has come to be used as a supplement that improves sperm quality. A previous study by Tsujihara et al. reported that L-carnitine intake in addition to exercise led to marked improvement in obesity [28]. This finding suggests that improving ones overall physical condition, including a re-examination of L- carnitine intake and exercise, might facilitate L-carnitines ability to improve infertility.
ACKNOWLEDGEMENT
This research was supported by Grant-in-aid for Scientific Research (C) from Japan Society for the Promotion of Sciences (KAKENHI No. 23617023) (TS).
COMPETING INTERESTS
The authors declare that they have no competing interests
AUTHORS CONTRIBUTIONS
MN and TS were involved in the design of the original study, data acquisition, data analysis, and writing the manuscript. All authors read and approved the final manuscript.
REFERENCES
- (2016). National Institute of Population and Social Security Research. The 15th Japanese National Fertility Survey in 2015.
- Zegers-Hochschild F, Adamson GD, de Mouzon J, Ishihara O, et al. (2009). International Committee for Monitoring Assisted Reproductive Technology (ICMART) and the World Health Organization (WHO) revised glossary of ART terminology. Fertil Steril. 92: 1520-1524.
- KUBO H. (2009). Epidemiology of Infertility and Recurrent Pregnancy Loss in Society with Fewer Children. The Japan Medical Association Journal. 52: 23-28.
- (2014). Japan Society of Obsterics and Gynecology. HUMAN+ administrative office ed: Dictionary in women and men.
- Hamatani T and Yoshimura Y. (2009). Infertility in Diagnosis, Treatment and Management of Gynecologic Disease. Japan Society of Obstercs and Gynecology.
- Benedict MD, Missmer SA, Vahratian A, Meeker JD, et al. (2011). Secondhand tobacco smoke exposure is associated with increased risk of failed implantation and reduced IVF success. Hum Reprod. 26: 2525-2531.
- Maheshwari A, Stofberg L and Bhattacharya S. (2007). Effect of overweight and obesity on assisted reproductive technology--a systematic review. Hum Reprod Update. 13: 433-444.
- Rossi BV, Berry KF, Hornstein MD, Cramer DW, et al. (2011). Effect of alcohol consumption on in vitro fertilization. Obstet Gynecol. 117: 136-142.
- World Health Organization. BMI classification.
- Ng M, Fleming T, Robinson M, Thomson B, et al. (2014). Global, regional, and national prevalence of overweight and obesity in children and adults during 1980-2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet. 384: 766-781.
- (2007). Ministry of Health, Labor and Welfare, Japan. Overview of National Health and Nutrition Examination Survey 2007.
- (2015). Ministry of Health, Labor and Welfare, Japan. Overview of National Health and Nutrition Examination Survey 2015.
- Bellver J, Melo MA, Bosch E, Serra V, et al. (2007). Obesity and poor reproductive outcome: the potential role of the endometrium. Fertil Steril. 88: 446-451.
- Kurauchi H, Narumi M and Suto T. (2012). Obesity in women. Diagnosis and Treatment. 100:1901-1906.
- Dupont C, Faure C, Sermondade N, Boubaya M, et al. (2013). Obesity leads to higher risk of sperm DNA damage in infertile patients. Asian J Androl. 15: 622-625.
- Martini AC, Tissera A, Estofan D, Molina RI, et al. (2010). Overweight and seminal quality: a study of 794 patients. Fertil Steril. 94: 1739-1743.
- Palmer NO, Bakos HW, Fullston T, Lane M. (2012). Impact of obesity on male fertility, sperm function and molecular composition. Spermatogenesis. 2: 253-263.
- Mounzih K, Lu R and Chehab FF. (1997). Leptin treatment rescues the sterility of genetically obese ob/ob males. Endocrinology. 138: 1190-1193.
- Brannian JD, Furman GM and Diggins M. (2005). Declining fertility in the lethal yellow mouse is related to progressive hyperleptinemia and leptin resistance. Reprod Nutr Dev. 45: 143-150.
- Ghanayem BI, Bai R, Kissling GE, Travlos G et al. (2010). Diet-induced obesity in male mice is associated with reduced fertility and potentiation of acrylamide-induced reproductive toxicity. Biol Reprod. 82: 96-104.
- Fan Y, Liu Y, Xue K, Gu G et al. (2015). Diet-induced obesity in male C57BL/6 mice decreases fertility as a consequence of disrupted blood-testis barrier. PLoS One. 10: e0120775
- Moradi M, Moradi A, Alemi M, Ahmadnia H et al. (2010). Safety and efficacy of clomiphene citrate and L-carnitine in idiopathic male infertility: a comparative study. Urol J. 7: 188-193.
- Arcaniolo D, Favilla V, Tiscione D, Pisano F et al. (2014). Is there a place for nutritional supplements in the treatment of idiopathic male infertility? Arch Ital Urol Androl. 86: 164-170.
- Zhou X, Liu F and Zhai S. (2007). Effect of L-carnitine and/ or L-acetyl-carnitine in nutrition treatment for male infertility: a systematic review. Asia Pac J Clin Nutr. 16(Suppl 1): 383-390.
- Palmer NO, Bakos HW, Owens JA, Setchell BP et al.(2012). Diet and exercise in an obese mouse fed a high-fat diet improve metabolic health and reverse perturbed sperm function. Am J Physiol Endocrinol Metab. 302: E768-E780
- Sidak ZK. (1967). Rectangular confidence resions for the means of multivariate normal distributions. J Am Stat Assoc. 62: 626-633.
- Garolla A, Maiorino M, Roverato A, Roveri A et al. (2005). Oral carnitine supplementation increases sperm motility in asthenozoospermic men with normal sperm phospholipid hydroperoxide glutathione peroxidase levels. Fertil Steril. 83: 355-361.
- Tsujihara N and Tani Y. (1997). Effects of Carnitine Administration and Voluntary Exercise on the Lipid Metabolism of Rats Fed on a High-Fat Diet. J Home Econ Jpn. 48: 5-9.