Previous Issues Volume 1, Issue 1 - 2018
Age Assessment in Forensic Cases: Anthropological, Odontological and Biochemical Methods for Age Estimation in the Dead
Joe Adserias-Garriga*1 , Sara C. Zapico2
1Fundació Universitat de Girona: Innovació i Formació, Universitat de Girona, Spain
2Department of Chemistry and Biochemistry and International Forensic Research Institute. Florida International University.
Corresponding Author: Adserias-Garriga, Fundació Universitat de Girona: Innovació i Formació, Universitat de Girona, Spain, Tel: (+34) 649 664 437; Email: [email protected]
Received Date: 11 Sep 2017 Accepted Date: 03 Jan 2018 Published Date: 08 Jan 2018
Copyright © 2018 Adserias-Garriga J
Citation: Adserias-Garriga J and Zapico SC. (2018). Age Assessment in Forensic Cases: Anthropological, Odontological and Biochemical Methods for Age Estimation in the Dead. Mathews J Foren. 1(1): 001.
ABSTRACT
One of the main goals of forensic science is determining the age of an individual to perform the biological profile. This is important in criminal cases and mass disaster scenarios, where the skeletons are often incomplete, which makes difficult their identification. The knowledge and application of anthropological methods and new biochemical approaches is essential to correct age determination, leading to the identification of an individual. Forensic anthropology and odontology methods estimate age through the macroscopic changes in bones and teeth due to growth and development in subadult individuals, and degenerative changes in adults. When growth has ceased, age estimation in adults is basically based on the degenerative changes of bone and teeth, and is generally less precise than in subadults. Due to the accuracy of the age estimate decreases as the age of the individual increases, other methods have been developed for estimating the age of the adult individual based on changes at the biochemical level due to the physiological process of aging. Biochemical methods are accurate, however, they have some limitations, so its use in combination with anthropological methods can be very useful for accurately estimating age.
KEYWORDS
Age Estimation; Biological Profile; Hman Identification.
INTRODUCTION
Anthropological and Odontological Methods for age estimation Traditionally, forensic anthropology and odontology methods estimate age through the macroscopic changes in bones and teeth due to growth and development in subadult individuals, and degenerative changes in adults. Selection of methods to be employed in age estimation depends upon the materials available for examination, their condition and the age category of the individual [1].
According to the age group to which the individual belongs, different methods of age estimation can be used. In this way, dental development could be used in fetal individuals [2-4], the presence of ossification nuclei [5], or long bones development [6]. In the case of children, dental development can be used [4, 7], the presence of ossification centers and the fusion of the epiphyses [3,8], and development of hand and wrist bones [9]. Dental development has been demonstrated to reflect chronological age more accurately than osteological development. Dental development appears to be under stronger genetic control, while osteological development is more influenced by environmental factors such as biomechanics, physiological stress and nutrition [1]. So that, in cases of age estimation of inmature individuals (fetal, neonatal, infant, child and adolescent) a special attention on dental age estimation methods should be considered.
In adolescents and young adults the most relevant age indicators are the development of the third molar [10], development of hand and wrist bones [9,11], spheno-occipital fusion [6,12] (Scheuer and Black, 2000; Madeline and Elster, 1995), and fusion of the sternal end of the clavicle [13, 14]; It is advisable to use as many indicators as possible, to obtain better results.
As in the case of children, adolescents and young adults (up to about 20-25years of age) skeletal indicators of age are based on the individual's growth and development; but when growth ends and the development of the individual is completed, age indicators are based on degenerative changes in the skeletal system.
There are different indicators of bone age in the adult as the pubic symphysis [15, 16], auricular surface of the coxae [17], the acetabulum surface [18], the sternal end of the ribs [19- 21], as well as degenerative changes in teeth [22-24]. Also, at a microscopic level an estimate of age can be achieved by the number of osteons [25-27].
All these mentioned methods based on different skeletal indicators have certain limitations that must be taken into account when applying them in forensic cases. Suchey-Brooks method [15] as well as Lamendin method [24] show greater accuracy in individuals between 20 and 40 years old. While the Isçan method [19-21] is more reliable for individuals over 60 years old. The Lovejoy method based on auricular region changes and his method based on the closure of cranial sutures [17, 28] have shown inter- and intraindividual variation. Also, the microscopic method based on the number of osteones present in the bone [25-27] is harder to implement than the observational methods mentioned. Table 1 resumes the different skeletal indicators for age assessment.
Table 1: Different indicators of skeletal age for different age groups [15-28].
When growth has ceased, age estimation in adults is basically based on the degenerative changes of bone and teeth, which can be affected by pathology.
Therefore, the estimation of age in adult individuals is generally less precise than in subadults [29]. Because of that reason, most researches suggest that when determining age in adult individuals, assessing multiple age indicators provides more accurate results than using a single indicador [1]. So that, it is always recommended to use as many indicators as possible in order to achieve the most accurate results.
Age estimation accuracy decreases as the age of the individual increases, other methods have been developed for estimating the age of the adult individual. These newer methods are based on changes at the biochemical level due to the physiological process of aging [30].
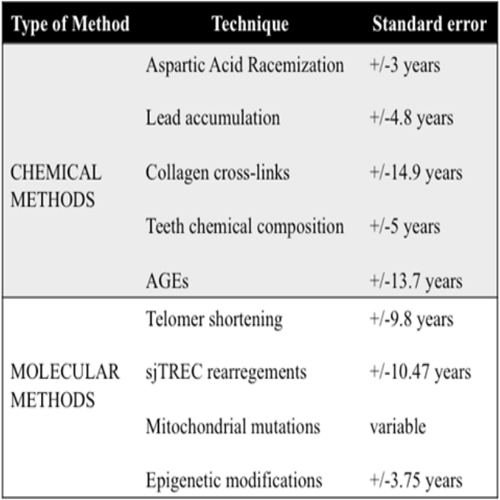
Biochemical methods for age estimation Biochemical methods for determining age are based on the natural process of aging, which induces alterations in tissues and organs at different biochemical levels [30]. According to these levels, methods for determining age are divided into chemical methods, including racemization of aspartic acid, lead accumulation, collagen cross-links, chemical composition of the teeth and analysis of advanced glycation end products (AGEs). Molecular methods include the analyses of telomere shortening, sjTRECs rearregements, the study of mitocondrial mutations and more recently epigenetic modifications [31, 32].
In living organisms, the most common amino acids have the L optical form. The racemization converts these amino acids into their D form, producing conformational alterations in proteins, which affect their biological activities and their chemical properties [33, 34]. These alterations in proteins can be correlated to the progressive changes associated with aging [35]. Aspartic acid has the fastest racemization range, becoming D-Asp, and being the most widely used amino acid in age estimation studies. Hence, aspartic acid racemization has been applied to different tissues, showing its accuracy in dentin [36, 37], cementum [36], intervertebral discs [38], elastine [39] and bone [40]. Despite the accuracy of this method (+/-3 years of error regarding chronological age), it can not be applied to bodies exposed to high temperaturas.
Since lead is one of the most common contaminants, measurement of its accumulation in dentin has been studied for the determination of age. The study of Al-Qattan and Elfawal [41] found an error of 1.3 + 4.8 years in the Kuwaiti population. Despite this result, more research is needed in this line to be able to assure that this technique can be applied to determine the age in forensic science.
Collagen matrix of cartilage, bone, dentin and other skeletal materials is stabilized through covalent bonds between collagen molecules [42]. A component of these linkages is deoxypyridoline (DPD). This component has been studied in relation to age, obtaining an error in the estimate of +14.9 years [43].
The chemical structure of the teeth changes with aging, particularly, the mineralization of the dentin and the reduction of the dentinal tubules, inducing an increase of the transparency of the root with the age. Structural and biochemical changes in dentin have been studied by several researchers in relation to age [5, 44], although the error in the estimation is not very high, additional studies are required to determine its possible application in the forensic context.
Advanced glycation endproducts occur through the Maillard reaction between reduced sugars and amino groups of the proteins, which induce different modifications in those proteins. These compounds acumúlate with age and have been correlated with complications derived from aging and associated diseases [44]. Few studies have been developed for forensic purposes, but they relate the accumulation of these compounds with age [45, 46]. Also, because of the color change caused by these compounds, this change has been related to age in some tissues [47].
Telomeres are at the end of the chromosomes, with each cell division, these terminations are shortened, limiting the proliferation of human cells and inducing senescence, differentiation or cell death. Different studies show that during the aging process telomeres are shortened [48, 49]. Because of this some researchers have studied them to determine the age [50, 51, 52], however, the mean error of this technique is about 10 years, being greater than in other techniques previously described.
During lymphocyte development in the thymus each immature T lymphocyte undergoes somatic rearrangement of its receptor (TCR, T-cell receptor), to generate a variety of TCR molecules. During this process, the DNA sequences of this TCR are eliminated and circularized in what are called "signal joint TCR excision circles, sjTRECs". These products do not replicate in cell division, for this reason they have been analyzed in relation to age [53, 54, 55], progressive decreasing of these products with the age. Like the prior technique, the associated error is 10 years.
According to mitochondrial theory of aging, mitochondrial DNA (mtDNA) is near the inner membrane of the mitochondria, which makes it more susceptible to damage generated by the free radicals released by the electron transport chain. This produces mtDNA mutations. An increase in the oxidative damage of mtDNA induces a deterioration in the mitochondrial respiratory function, producing more free radicals and consequently, an accumulation of lesions that can not be repaired in mitochondrial DNA [56]. Different studies have shown the correlation between mitochondrial mutations and age in different tissues [56-60]. However, further research is required in this line to determine the level of accuracy of this method.
Recently the correlation between methylation levels and age has been stablished in blood samples and dentin, being for blood samples the associated error of 3.75 years, with respect to chronological age and 4.86 error, with respect to chronological age for dentine [31]. These results are encouraging for its possible application in forensic science, although this research is still starting, it requires the analysis of more methylation markers to increase the efficacy and accuracy of this method.
Biochemical methods available for age estimation with its standard errors are presented in table 2.
Table 2: Biochemical methods available for age estimation with its standard errors [30-52].
CONCLUSIONS
In this article, anthropological and biochemical methods for the correct determination of age have been discussed. Biochemical methods seem to be more accurate for this purpose, particularly the racemization of aspártico acid and more recently the epigenetic modifications, however, they have some limitations, so they could be useful in combination with other anthropological techniques. The selection and application of one technique or another depends on the forensic context and the availability of the human remains.
REFERENCES
- (2013). Scientific Working Group for Forensic Anthropology (SWGANTH) Age Estimation Issue.
- Schour I and Massler M. (1941). The development of the human dentition. Journal of the American Dental Association 28: 1153-1160.
- Ubelaker DH. (1987). Estimating age at death from immature human skeletons: an overview. Journal of Forensic Sciences. 32(5): 1254-1263.
- AlQahtani SJ, Hector MP and Liersidge HM. (2010). Brief communication: The London atlas of human tooth development and eruption. American Journal of Physical Anthropology 142(3): 481-490.
- Kosa F, Antal A and Farkas I. (1990). Electron probe microanalysis of human teeth for the determination of individual age. Medicine, science, and the law. 30(2):109-114.
- Scheuer L and Black S. (2000). Developmental Juvenile Osteology. Academic Press.
- Demirjian A, Goldstein H and Tanner JM. (1973). A new system of dental age assessment. Human Biology 45(2), 211-227.
- Lynnerup N, Belard E, Buch-Olsen K, Sejrsen B, et al. (2008). Intra- and interobserver error of the Greulich- Pyle method as used on a Danish forensic sample. Forensic Science International 179(24): e241-e246.
- Greulich WW and Pyle SI. (1959). Radiographic Atlas of Skeletal Development of the Hand and Wrist. 2nd ed. 91(1): 53.
- Mincer HH, Harris EF and Berryman HE. (1993). The A.B.F.O. study of third molar development and its use as an estimator of chronological age. Journal of Forensic Sciences. 38(2): 379-390.
- Tanner JM, Whitehouse RH, Marshall WA and Carter BS. (1975). Predictionofadulthei ghtfromheight, bone age, and occurrence of menarche at ages 4-16 with allowance for midparent height. Arch Dis Child 50: 14-26.
- Madeline LA and Elster AD. (1995). Suture closure in the human chondrocranium: CT assessment. Radiology 196(3): 747-756.
- Schulz R, Muhler M, Mutze S, Schmidt S, et al. (2005). Studies on the time frame for ossification of the medial epiphysis of the clavicle as revealed by CT scans. International Journal of Legal Medicine 119(3): 142-145.
- Schmidt S, Muhler M, Schmeling A, Reisinger W, et al. (2007). Magnetic resonance imaging of the clavicular ossification. International Journal of Legal Medicine 121(4): 321-324.
- Brooks S and Suchey J. (1955). Age at death: reliability of cranial and pubic age indicators, Am. J. Phys. Anthropol. 13(4): 567-597.
- Todd TW. (1921). Age changes in the pubic bone IV: the interpretation of variations in symphyseal area. Am. J. Phys. Anthropol. 4(4): 407-424.
- Lovejoy CO, Meindl RS, Pryzbeck TR and Mensforth RP. (1985). Chronological metamorphosis of the auricular surface of the ilium: a new method for the determination of adult skeletal age at death. American Journal of Physical Anthropology. 68(1): 15-28.
- Rouge-Maillart C, Jousset N, Vielle B, Gaudin A, et al. (2007). Contribution of the study of acetabulum for the estimation of adult subjects. Forensic Science International. 171(2-3): 103-110.
- Iscan MY, Loth SR and Wright RK. (1984). Age estimation from the rib by phase analysis: white males. Journal of Forensic Sciences 29(4): 1094-1104.
- Iscan MY, Loth SR and Wright RK. (1985). Age estimation from the rib by phase analysis: white females. Journal of Forensic Sciences. 30(3): 853-863.
- Iscan MY, Loth SR and Wright RK. (1987). Racial variation in the sternal extremity of the rib and its effect on age determination. Journal of Forensic Sciences 32(2): 452-466.
- Cameriere R, Ferrante L and Cingolani M. (2004). Precision and reliability of pulp/tooth area ration (RA) of second molar as indicador of adult age. Journal of Forensic Sciences 49(6): 1319-1323.
- Kvaal SI and Solheim T. (1994). A non-destructive dental method for age estimation. Journal of Forensic Odontostomatol. 12(1): 6-11.
- Lamendin H, Baccino E, Humbert JF, Tavernier JC, et al. (1992). A simple technique for age estimation in adult corpses: The two criteria dental method. Journal of Forensic Sciences. 37(5): 1373-1379.
- Ahlqvist J and Damsten O. (1969). A modification of Kerley's method for the microscopic determination of age in human bone. Journal of Forensic Sciences 14(2): 205-212.
- Cool SM, Hendrikz JK, Wood WB. (1995). Microscopic age changes in the human occipital bone. Journal of Forensic Sciences. 40(5): 789-796.
- Kerley ER and Ubelaker DH. (1978). Revisions in the microscopic method of estimating age at death in human cortical bone. American Journal of Physical Anthropology. 49(4): 545-546.
- Meindl RS and Lovejoy CO. (1985). Ectocranial suture closure: a revised method for the determination of skeletal age at death base don the latero-anterior sutures, American Journal of Physical Anthropology 68(1): 57-66.
- Adams B. (2007). "Forensic Anthropology". Chelsea House Publications. New York.
- Balin AK and Allen RG. (1989). Molecular bases of biologic aging. Clinics in geriatric medicine. 5(1): 1-21.
- Bekaert B, Kamalandua A, Zapico SC, Van de Voorde W, et al. (2015). Improved age determination of blood and teeth samples using a selected set of DNA methylation markers. 10(10): 1-9.
- Ubelaker DH, Buchholz BA and Bergman LE. (2006). Analysis of artificial radiocarbon in different skeletal and dental tissue types to evaluate dateof death. Journal of Forensic Science. 51(3): 484-488.
- Helfman PM and Bada JL. (1975). Aspartic acid racemization in Toth enamel from living humans. Proceedings of the National Academy of Sciences of the United States of America. 72(8): 2891-2894.
- Masters PM, Bada JL and Zigler JS Jr. (1977). Aspartic acid racemisation in the human lens during ageing and in cataract formation. Nature. 268(5615): 71-73.
- Helfman PM, Bada JL and Shou MY. (1977). Considerations on the role of aspartic acid racemization in the aging process. Gerontology 23(6): 419-425.
- Ohtani S. (1995). Estimation of age from the teeth of unidentifiedcorpses using the amino acid racemization method with reference to actual cases. The American journal of forensic medicine and pathology 16(3): 238- 242.
- Ritz S, Schutz HW and Schwarzer B. (1990). The extent of aspartico acid racemization in dentin: a possible method for a more achurate determination of age at death? Zeitschrift fur Rechtsmedizin Journal of legal medicine 103(6): 457-462.
- Ritz S and Schutz HW. (1993). Aspartic acid racemization in intervertebral discs as an aid to postmortem estimation of age at death. Journal of forensic sciences 38(3): 633-640.
- Dobberstein RC, Tung SM and Ritz-Timme S. (2010). Aspartic acid racemisation in purified elastin from arteries as basis for age estimation. International journal of legal medicine. 124(4): 269-275.
- Ohtani S, Matsushima Y, Kobayashi Y and Yamamoto T. (2002). Age estimation by measuring the racemization of aspartic acid from total amino acid content of several types of bone and rib cartilage: a preliminary account. Journal of forensic sciences. 47(1): 32-36.
- Al-Qattan SI and Elfawal MA. (2010). Significance of teeth lead accumulation in age estimation. Journal of forensic and legal medicine 17(6): 325-328.
- Eyre D. (1987). Collagen cross-linking amino acids. Methods in enzymology. 144: 115-139.
- Martin-de las Heras S, Valenzuela A and Villanueva E. (1999). Deoxypyridinoline crosslinks in human dentin and estimation of age. International journal of legal medicine 112(4): 222-226.
- Ager JW 3rd, Nalla RK, Balooch G, Kim G, et al. (2006). On the increasing fragility of human teeth with age: a deepUV resonance Raman study. Journal of bone and mineral research: the official journal of the American Society for Bone and Mineral Research. 21(12): 1879-1887.
- Bank RA, Bayliss MT, Lafeber FP, Maroudas A, et al. (1998). Ageing and zonal variation in post-translational modification of collagen in normal human articular cartilage. The age-related increase in non-enzymatic glycation affects biomechanical properties of cartilage. The Biochemical journal. 330(Pt 1): 345-351.
- Sato Y, Kondo T and Ohshima T. (2001). Estimation of age of human cadavers by immunohistochemical assessment of advanced glycation end products in the hippocampus. Histopathology. 38(3): 217-220.
- Pilin A, Pudil F and Bencko V. (2007). The image analysis of colour changes of different human tissues in the relation to the age. Part 2: practical applicability. Soudni lekarstvi / casopis Sekce soudniho lekarstvi Cs lekarske spolecnosti J Ev Purkyne. 52(2): 26-30.
- Harley CB, Futcher AB and Greider CW. (1990). Telomeres sorteen during ageing of human fibroblasts. Nature. 345(6274): 458-460.
- Melk A, Ramassar V, Helms LM, Moore R, et al. (2000). Telomere shortening in kidneys with age. Journal of the American Society of Nephrology: JASN. 11(3): 444-453.
- Ren F, Li C, Xi H, Wen Y, et al. (2009). Estimation of human age according to telomere shortening in peripheral blood leukocytes of Tibetan. The American journal of forensic medicine and pathology. 30(3): 252-255.
- Takasaki T, Tsuji A, Ikeda N, et al. (2003). Age estimation in dental pulp DNA based on human telomere shortening. International journal of legal medicine. 117(4): 232-234.
- Tsuji A, Ishiko A, Takasaki T and Ikeda N. (2002). Estimating age of humans based on telomere shortening. Forensic science International. 126(3): 197-199.
- Ou X, Zhao H, Sun H, Yang Z, et al. (2011). Detection and quantification of the age-related sjTREC decline in human peripheral blood. International journal of legal medicine. 125(4): 603-608.
- Pido-Lopez J, Imami N and Aspinall R. (2001). Both age and gender affect thymic output: more recent thymic migrants in females than males as they age. Clinical and experimental immunology 125(3): 409-413.
- Zubakov D, Liu F, van Zelm MC, Vermeulen J, et al. (2010). Estimating human age from T-cell DNA rearrangements. Current biology : CB. 20(22): R970-971.
- Zapico SC and Ubelaker DH. (2015). Relationship Between Mitochondrial DNA Mutations and Aging. Estimation of Age-atdeath. The journals of gerontology Series A, Biological sciences and medical sciences.
- Lacan M, Theves C, Amory S, Keyser C, et al. (2009). Detection of the A189G mtDNA heteroplasmic mutation in relation to age in modern and ancient bones. International journal of legal medicine. 123(2): 161-167.
- Lacan M, Theves C, Keyser C, Farrugia A, et al. (2011). Detection of age-related duplications in mtDNA from human muscles and bones. International journal of legal medicine. 125(2): 293-300.
- Meissner C, von Wurmb N and Oehmichen M. (1997). Detection of the age-dependent 4977 bp deletion of mitochondrial DNA. A pilot study. International journal of legal medicine. 110(5): 288-291.
- Theves C, Keyser-Tracqui C, Crubezy E, Salles JP, et al. (2006). Detection and quantification of the age-related point mutation A189G in the human mitochondrial DNA. Journal of forensic sciences. 51(4): 865-873.
.png)