Previous Issues Volume 2, Issue 1 - 2017
Chemical and Physical Analysis of the Effect of In-Office Tooth Bleaching Agent on Three Esthetic Composite Resin Restorations
Ameer Hamdi AL-Ameedee1*
1Faculty of Dentistry, University of Babylon, Babylon, Hilla, Iraq.
Corresponding Author: Ameer Hamdi AL-Ameedee, Faculty of Dentistry, University of Babylon, Babylon, Hilla, Iraq, Tel: 030 - 249551; E-Mail: [email protected]
Received Date: 22 Jul 2016 Accepted Date: 23 Dec 2016 Published Date: 28 Dec 2016 Copyright © 2017 AL-Ameedee AH
Citation: AL-Ameedee AH. (2017). Chemical and Physical Analysis of the Effect of In-Office Tooth Bleaching Agent on Three Esthetic Composite Resin Restorations. Mathews J Dentistry. 2(1): 013.
ABSTRACT
In the present study we evaluate the physical and chemical analysis effects of the in-office bleaching gel (H2O2 25%) on three contemporary restorative composite resins by using scanning electron microscope (SEM), atomic force microscope (AFM), energy dispersive X-ray spectroscopy (EDAX), Fourier transform infrared spectrometer (FTIR) and Vickers micro hardness taster (HVS), out of all the tested composite resins in comparison with two follow-up examinations (before bleaching and after one week of bleaching).
Materials and Methods: A total of 300 discus composite resins; Beautifil II, a Nano hybrid composite resin (Giomer); IPS Empress Direct, a Nano hybrid composite resin (Ceromer); and the Ceram-x-mono, a Nano ceramic micro hybrid composite resin (Ormocer); (n=100 for each group), were selected for an measurements evaluation of before (n=50 for each group) and after one week of bleaching treatment (n=50 for each group) by micro hardness tester measurements and chemical analysis using SEM, AFM, EDAX images and FTIR (n=10 for each test).
Results: The Vickers's hardness revealed a significant increase in micro hardness of the Beautifil II and IPS Empress Direct; also the SEM, AFM, EDAX images and FTIR revealed a great diversity in ultrastructure. The results obtained served to validate that the methods employed in this study, SEM, AFM, EDAX, and FTIR were useful in examining and analysing the physical and chemical changes of bleaching on the composite resins.
Conclusions: It was also found that the changes of the arrangement of filler and chemical composition on the composite resins caused by the effect of bleaching had an effect on the chemical and physical properties. Further studies are needed in search of clinical applications that optimally match the differing properties of these materials.
KEYWORDS
SEM; EDAX; AFM; FTIR; Vickers Hardness Tester; Beautifil II; IPS Empress Direct; Ceram.x.mono; Bleaching Materials.
INTRODUCTION
Bleaching teeth treatments are deemed acceptable among the population with increases in the production of new types bleaching systems. In addition, there are increased numbers of patients with composite resin restorations placed in their teeth. As all composite resins undergo chemical and physical property changes with their increasing use as bleaching agents, the goal of dental restoration is to conserve sound tooth structure during the preparation and restorative phases [1, 2]. On the development of restorative materials for adhesive procedures, the introduction of composite resins to the dental profession by Bowen has since generated much discussion from clinicians, researchers, and manufacturers [3].
Composite resins are acceptable restorative materials for the management of fracture or dental caries in the anterior teeth [4, 5]. The recent resin-based materials have shown increasing quality and longevity [6]. The dental composite is made up of four major components: an organic polymer matrix, inorganic filler particles, a coupling agent, and an initiator-accelerator system. The resin forms the matrix of the composite material, and the coupling agent binds the individual filler particles together. The most used monomer is bisphenol-A-glycidyl methacrylate (Bis-GMA) diluted either with triethyleneglycol dimethacrylate (TEGDMA) or with Di-urethane methacrylate (DUMA) [5, 7].
To enhance strength, stiffness, reduce dimensional changes and improve handling, fillers are added to the polymeric part of the composite. Recently, most of the dental resin has been filled with silicate particles based on oxides of barium, aluminium, strontium, or zirconium and zinc [8].
The highest mechanical properties could be obtained by using high concentrations of filler particles of varied sizes into the resin [8-10]. Different classifications according to the average size of the filler particles have been placed and they found there was no superiority of any specific filler as every type of filler revealed advantages and disadvantages during clinical application [3, 5, 8, 11]. They revealed that one of the most important causes of reinforcing composites was a suitable bond between the fillers and the resin matrix. The dental resin microstructure and properties were studied extensively [4, 9, 12]. They used a scanning electron microscope (SEM) successfully for evaluating the filler particles' numbers, shapes, and sizes. The null hypothesis of present study for the effects of in-office bleaching will have no effect on the three resin composites.
MATERIALS AND METHODS
Three hundred disc-shaped composite resin specimens were prepared from three types of resin composites; group one was prepared using Beautifil II (1), group two was prepared using IPS Empress Direct(2) and group three was prepared using Ceram.x.mono(3) composite restoration; according to the manufacturer's instructions (n=100 for each group). For each fabricated disc, a notch was marked on the untreated surface in order to distinguish the bleached tested surface. Five mm-diameter and two mm-thick discs were fabricated using a custom-made Teflon mold. The shade (A1) was standardized for all restorative materials to eliminate this variable and was inserted as one increment into the Teflon mold; covered by a Mylar strip(4) followed by a microscopic glass plate, then 200 g of pressure was applied for 45 sec to expel excess material from the mold and to reduce voids [13]. This technique ensures equal pressure and creates a flat surface on all the specimens being fabricated; light polymerization (5) was performed for 40 seconds at the top surface of specimens for each disc to simulate the clinical condition, using a halogen light of 350 mW/cm2. The distance between the light tip and the specimen surfaces was standardized by touching the glass slide (1.2 mm thickness). Then the specimens were polished using Sof-Lex system polishing discs (6) with a slow-speed hand piece, rotating in one direction, followed by a 30-second ultrasonic cleaner. All the specimens were stored in distilled water at 37 Co using environmental water bath (7) for one week before bleaching application to ensure complete polymerization [14, 15].
Bleaching Procedures
The bleaching was performed using Zoom Bleaching kite (8) (25% HP). After applying a layer of gel (approximately 2 mm thick) to the disc surface (one hundred fifty disc-shaped composite resin specimens were prepared from three types of resin composites all groups), the Zoom light source was applied for four bleaching sessions of 15 minutes each, after that the gel was removed with a plastic spatula and sterile gauze, then all the disc specimens were cleaned under running water for 1 minute and later stored in distilled water for one week at 37o C [15-17].
Measurement
All groups specimens remain in distilled water at 37o C using an environmental water bath till the time of measurement, the measuring technique was made for each group separately. Measurement baseline time follow ups for before bleaching treatment for one hundred fifty disc-shaped composite resin specimens were prepared from three types of resin composites all groups and after one week of for each specimen; the mean and standard deviation of each group were calculated [18, 19]. The lighter color indicates raised areas of the specimen surface. The topography picture and diagram curve for the centre area of the disc specimen were converted to arithmetic data for the surface roughness value (Sa) to compare with other specimen groups [7, 20-23].
Although the arithmetic average of the roughness profile (Ra) is commonly used for surface roughness, the arithmetic average of the three-dimensional roughness profile (Sa) was recorded for bleaching time follow ups of other one hundred fifty disc-shaped composite resin specimens were prepared from three types of resin composites for all parameters were recorded (Table 1). Data were calculated.
Table 1: The Groups Distribution According to Type of Restorations Materials.
| (B1): Beautifil II restoration before expose to H2 O2 . (n=50) | (E1): ISP Empress restoration before expose to H2 O2 . (n=50) | (C1): Ceram.x.mono restoration before expose to H2 O2 . (n=50) |
| (B2): Beautifil II restoration one week after expose to H2 O2 . (n=50) | (E2): ISP Empress restoration one week after expose to H2 O2 .(n=50) | (C2): Ceram.x.mono restoration one week after expose to H2 O2 .(n=50) |
Scanning Electron Microscope (SEM) Measurement
The measurements were recorded before (n=10 for each group) and after one week (n=10 for each group) of bleaching time intervals for the groups of the study by using scanning electron microscope(9) (SEM), the resins blocks of 2 x 5 mm were prepared as described before, and the specimens were placed on carbon stubs. The samples were coated with carbon for 5 seconds [20]. The electronic analysis was performed with Seron technologies.
The Vickers Micro Hardness Measurement
The (HVS) measurements were recorded before (n=10 for each group) and after one week (n=10 for each group) of bleaching time intervals for the groups of the study by using a Vickers micro hardness tester (HVS) (10) a 100-gram load and 20-second dwell time were applied at room temperature to the bleach surface specimens disc. The diagonal length impressions were measured and the hardness number (H) was calculated according to a standard formula (H= 1.854 P/d2). Where (P) represents the indentation load, and (d) indicates the diagonal length impression for each disc specimen, five indentations were made on the bleach treated top surface, not closer than (1) mm to the adjacent indentations or the margins of the specimen [20-22].
Fourier Transform Infrared Spectrometer (FTIR) Measurement
The measurements were recorded before (n=10 for each group) and after one week (n=10 for each group) of bleaching time intervals for the groups of the study by using Fourier transform infrared spectroscopy.(11) (FTIR) is a technique that uses infrared light to observe properties of a solid, liquid, or gas (n=10 for each group). It is used in many different applications to measure the absorption, emission, and photoconductivity of matter by shining a narrow beam of infrared light at the matter in various wavelengths and detecting how the matter responds to each wavelength. Once the data have been obtained, it is converted into digital information using a mathematical algorithm known as the "Fourier transform." The study of some properties can be made by the degree of conversion and hardness tests. Degree of conversion (DC) is an important parameter in determining the final physical, mechanical, and biological properties of photo-activated composite resins. The DC were determined by the proportion of the remaining concentration of the aliphatic C=C double bonds in a cured sample relative to the total number of C=C bonds in the uncured material [23-25].
Energy Dispersive X-ray Spectroscopy (EDAX) measurement
From the specimen discs (the measurements were recorded before (n=10 for each group) and after one week (n=10 for each group) of bleaching time intervals for the groups of the study), resin powders were prepared, then placed on carbon stubs to analyse using energy dispersive X-ray spectroscopy (12) (EDAX). The information was obtained after 300 seconds of measurement. An area of approximately 20×15 µm was selected for 2D analysis, which included both the resin matrix and filler particles. Relative values were obtained after 300 seconds of measurement
Atomic Force Microscope (AFM) Measurement
The surface roughness of the disc specimen (the measurements were recorded before (n=10 for each group) and after one week (n=10 for each group) of bleaching time intervals for the groups of the study) was evaluated using an atomic force microscope (13) (AFM). The surface dimensions of the different disc specimens were scanned in different directions: 1×1 µm to 10×10 µm (x, y) and 4 µm (z). The images collected were processed with software including plane fitting and x-y flattening and two-dimensional (2D) and three-dimensional (3D) images were captured [21]. The (Sa) measurements were recorded before and after one week of bleaching time intervals for the groups of the study [7, 24, 25].
Statistical Analysis
Data were collected, tabulated, and statistically analysed. A master chart was prepared in MS Excel Sheet used for tables, diagrams and analysed using Statistical Package for Social Sciences (SPSS 20, IBM) (Weiss and Weiss, (2012).
A-Descriptive Statistics 1. Arithmetic mean. 2. Standard deviation (SD). 3. Tables.
B-Inferential Statistics 1. One-way ANOVA (F-test) was used to compare between mean values of more than two groups. 2. Least Significant Differences test (LSD) was used for multiple comparisons between the groups. Probability levels of more than (0.05) were regarded as statistically non-significant, while levels less than or equal to (0.05) were considered as a significant. P < 0. 05 significant P < 0. 01 high significant P > 0. 05 non-significant
RESULTS
The analysis of the present study has been evaluated and tabulated as follows: In the present study, the measurements were recorded before (n=10 for each group) and after one week (n=10 for each group) of bleaching time intervals for the groups of the study.
Scanning Electron Microscope (SEM) analysis
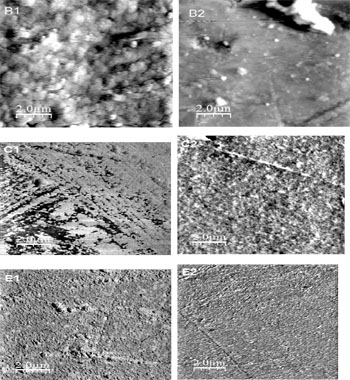
SEM micrographs revealed that all the composites showed surface alterations in their superficial surface after bleaching. Micrographs of the restorative composite resins under SEM are shown in micrographs 1) observation with the SEM micrographs shown with a backscattered electron signal provided an adequate contrast between the resin matrix and fillers. The shapes and sizes of the filler particles were different between the composites. The appearance of the Beautifil II and ISP Empress Direct groups presented a greater amount of filler particles alike where their superficially bigger filler particles were observed with the inclusion of a few nanoparticles. For the Ceram.x.mono group the SEM micrographs showed that they were alike where their homogeneously smaller filler particles were. After one week of bleaching for the Ceram.x.mono group, the backscattered micrograph showed a view which was the most similar appearance of the Beautifil II and ISP Empress Direct groups as seen in micrographs (1), in surface texture among the these composite resin groups, it is interesting to note that Beautifil II presented the most complex ultrastructure, whereby large filler particles were observed with a SEM photomicrograph (Figures 1).
Surface Micro hardness (VHN) Analysis
The mean values of Vickers hardness are shown in (Table 2, 3). It must be mentioned that in all cases, the size of the indentations were larger than the filler particles. As such, the score recorded was the average value of both the resin matrix and filler. Large differences in micro hardness between before bleaching and after one week of bleaching were observed among the composites. The results were a statistically significant increase in mean value in the Beautifil II and ISP Empress Direct groups (66.12 �2.94 VHN and 63.86 ), respectively, whereas Ce2.75), respectively, whereas Ceram.x.mono showed no statistically significant mean value effect (56.44 ±3.9 VHN).
A statistical analysis of the data using an ANOVA test between the groups before bleaching showed that there was significant difference between the surface micro hardness of these groups (P<0.05). It also showed a highly significant difference between the surface micro hardness (P<0.01) for one week after bleaching, as seen in (Table 2).
Table 2: Anova Results of the Effect of Time on Surface Microhardness Mean Values of All Groups (B&E&C).
| Statistical test | T0 | T1 | |
| between groups B&E&C | P-value | 9.586 | 17.21 |
| P-value | P<0.05* | P<0.01** |
* The mean difference is significant at the 0.05 level. ** The mean difference is high significant at the 0.01 level.
B:Beautifil II group, E: ISP Empress Direct group, C: Ceram.x.mono group. T0: Before bleaching, T1: one week after bleaching.
Table (3) shows that time and bleaching had a statistically significant effect on the surface micro hardness of the ISP Empress Direct group (P<0.01) and a significant effect on the Beautifil II group (P<0.05), while there was no statistically significant effect on the surface micro hardness of the Ceram.x.mono group at one week after the bleaching follow up times (p=0.584).
Table 3: Descriptive Statistical Study of Surface Microhardness Values (Vhn) in Kg/Mm2 of All Groups (n=10).
| T0 | T1 | ||
| Beautifil II group | Mean | 53.0394b2 | 66.12a1 |
| SD | 3.822 | 2.94 | |
| ISP Empress Direct group | Mean | 50.355b3 | 63.86a1 |
| SD | 4.576 | 2.75 | |
| Ceram.x.mono group | Mean | 58.595a1 | 56.44a2 |
| SD | 4.443 | 3.9 |
* The mean difference is significant at the 0.05 level. Different latter in same row = significant differences. Different number in same Colum = significant differences. T0: Before bleaching, T1: one week after bleaching.
Fourier Transform Infrared Spectrometer (FTIR) analysis
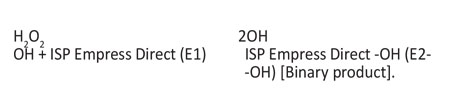
In the presenting study, the results for the Beautifil l group noticed a slight shift in the intensity and peak position of the carbonyl group (C=O), which has an absorbance in the range of 1730 in Beautifil II before bleaching group (B1), and converted or shifted to 1724, as seen in Beautifil II after one week of bleaching group (B2). Also we have another peak in the FTIR spectroscopy results The most important peak hydroxyl group (OH) has an absorbance of between (3400-3448), and this peak will remain the same because the main reaction is:
Figure 2 (B1): FTIR diagram for Beautifil Il composite group's samples before bleaching.
Figure 3 (B2): FTIR diagram for Beautifil II composite group's samples after one week of bleaching.
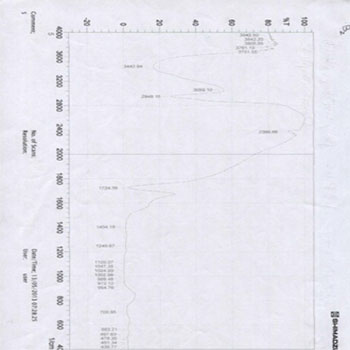
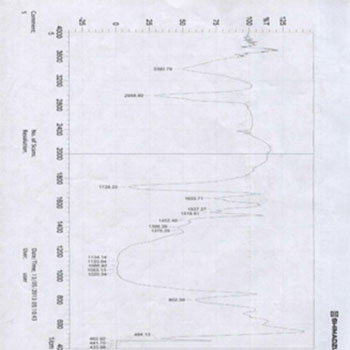
In addition, the ISP Empress Direct group results showed a slight shift in the intensity of the carbonyl group (C=O), which has an absorbance in the range of 1732 for the ISP Empress Direct group before bleaching treatment (E1) and converted or shifted to 1722 one week after bleaching treatment. Also, we have another peak in the FTIR spectroscopy, but this peak will not change, as shown in (Figures 4, 5). The most important peak hydroxyl group (OH) has an absorbance of between (3400-3500) and this peak will remain the same because the main reaction is:
Figure 4 (E1): FTIR diagram for ISP Empress Direct composite group�s samples before bleaching.
Figure 5 (E2): FTIR diagram for ISP Empress Direct composite group's samples after bleaching.
While the results of the Ceram.x.mono group (C) showed a slight shift in the intensity of the carbonyl group (C=O), which has an absorbance in the range of 1728 before bleaching treatment (C1) and converted or shifted to 1730 one week after bleaching treatment (C2), we also have another peak in FTIR spectroscopy, but this peak will not change, as shown in (Figures 6, 7). The main important peak hydroxyl group (OH) has an absorbance of between (3400-3448) and this peak will remain the same because the main reaction is:
Figure 6 (C1): FTIR diagram for Ceram.x.mono composite group�s samples before bleaching.
Figure 7 (C2): FTIR diagram for Ceram.x.mono composite group's samples after one week of bleaching.
Rastelli et al [24] said other factors can influence the DC such as the light source used, power density, wavelength, irradiation time, light-tip size, photo-activation method, distribution, quantity of inorganic fillers, the type and quantity of the photo initiator, and the color also strongly affect the DC of the composite resins.
The percentage of unreacted carbon-carbon double bonds (C=C) was determined from the ratio of absorbance intensities of aliphatic C=C (peak at 1637 cm-1) against the internal reference aromatic C=C (peak at 1610 cm-1) before and after the curing of the specimens.
EDAX Analysis
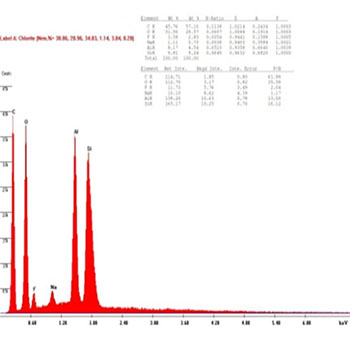
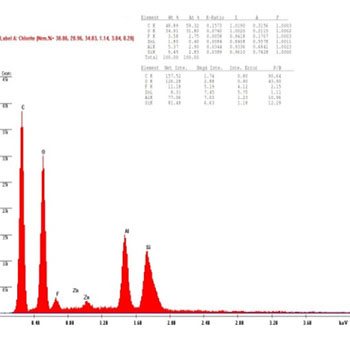
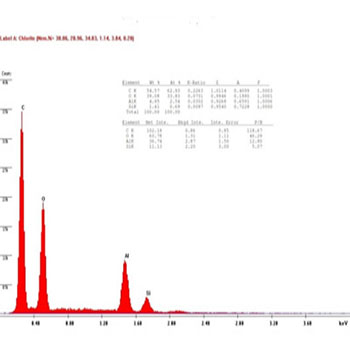
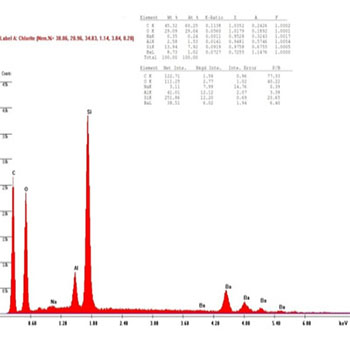
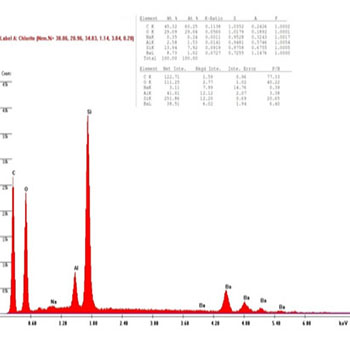
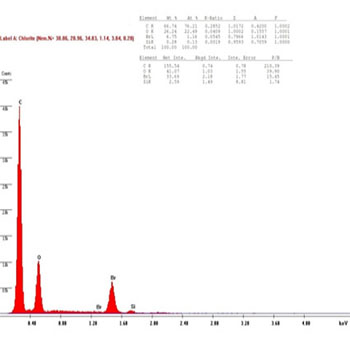
In the presenting study, The chemical compositions of the restorative composite resins, including the elements with relative values, expressed in weight percentage are presented in (Figures 8 to 13): similar elements, such as (Al), (O) and Si, were detected; however, the concentration was different in every composite resin. The filler contents showed interesting differences in elemental composition and concentration.
Figure 8 (B1): EDAX diagram for Beautifil II composite group's samples before bleaching.
Figure 9 (B2): EDAX diagram for Beautifil II composite group's samples after bleaching.
Figure 10 (E1): EDAX diagram for ISP Empress Direct composite group's samples before bleaching.
Figure 11 (E2): EDAX diagram for ISP Empress Direct composite group's samples after bleaching.
Figure 12 (C1): EDAX diagram for Ceram.x.mono composite group's samples before bleaching.
Figure 13 (C2): EDAX diagram for Ceram.x.mono composite group's samples after one week of bleaching.
The results were shown to confirm the reaction between the hydroxyl group (OH) and samples of the Beautifil II group done by using the EDAX technique; it is clear from (Figures 8, 9) that in the before bleaching and one week after bleaching groups the intensity of Al and Si reduced to 50% and 55%, respectively, whereas the amount of the (O) element reduced to 20% in the one week after bleaching group, which is attributed to the reaction between the (OH) radical and the Beautifil II samples.
Also, the result of the ISP Empress Direct group confirmed the reaction with the hydroxyl group (OH) which occurred because the intensity of the (Al) element reduced to 40%, while the amount of the (O) element remained the same. This gives a profit for the reaction of the (OH) radical with the ISP Empress Direct in the (E1) before bleaching group decreasing in intensity the (Al) element, thus causing an increase in the hardness because of the rearrangement phenomena or the adaptation on the surface of the ISP Empress Direct in the after one week after bleaching group, as shown in (Figures 10, 11).
While the result of the Ceram.x.mono before bleaching group showed the amount of the (Al) element to decrease at a very high percent 90%, and same for the (Si) element, which caused a decrease in the hardness of Ceram.x.mono in the one week after bleaching group, which is explained by a hard reaction between the (OH) radical and the Ceram.x.mono samples' surface, as shown in (Figures 12, 13).
AFM Analysis
In the presenting study, The results for Beautifil II in the one week after bleaching group showed the surface roughness (Sa) average will decrease by about 22% compared to Beautifil II in the one week after bleaching group because the amount of (Al) and (Si) decrease, as shown in (micrographs 14, 15). And, according to the results above and by use of the Vicker test tool, the hardness of the Beautifil II samples increases for one week after (H2 O2 ) treatment, which is explained by the effect of rearrangement on the surface of sample after reacting with the (OH) radical.
Also, the results the for ISP Empress Direct group the surface roughness (Sa) average will decrease for one week after bleaching treatment, which is explained by the increase in the intensity of the (Si) element after treatment as notes from the EDAX results, as shown in (micrographs 16, 17). So, according to the above results and by using the Vicker test tool, the hardness of the ISP Empress Direct samples increases after (H2 O2 ) treatment, which is explained by the effect of rearrangement on the surface of the sample after reacting with the (OH) radical.
Micrographs 14 (B1): AFM micrographs for Beautifil II composite group's samples before bleaching.
Micrographs 15 (B2): AFM micrographs for Beautifil II composite group's samples after bleaching.
Micrographs 16 (E1): AFM micrographs for ISP Empress Direct composite group's samples before bleaching.
Micrographs 17 (E2): AFM micrographs for ISP Empress Direct composite group's samples after bleaching.
Whereas the results for the Ceram.x.mono group show that the surface roughness (Sa) average will decrease in the one week after bleaching group, and this may be due to rearrangement on the Ceram.x.mono samples' surface by reducing the high percentage of (Al) and (Si), as shown in (micrographs 18, 19). So, according to the above results and by using the Vicker test tool, the hardness of the Ceram.x.mono samples decrease after (H2O2) treatment, which is explained by the effect of rearrangement on the surface of samples after reacting with the (OH) radical.
Micrographs 18 (C1): AFM micrographs for Ceram.x.mono composite group's samples before bleaching.
Micrographs 19 (C2): AFM micrographs for Ceram.x.mono composite group's samples after one week of bleaching.
DISCUSSION
In the present study, SEM micrographs revealed that all the composites showed surface alterations in their superficial surface after bleaching. Interestingly, some studies on the surface roughness for composite resin after applying bleaching gels have reported an increase, a decrease, or unchanged, or that it led to a slight roughness of resin-based composites, although it may have no clinical significance [20, 26-28]. The Vickers microhardness test results revealed significant difference between the composites (Table 2). This result could be explained in terms of the chemical composition, suggesting that a higher combined concentration of elements could make a composite resin harder.
SEM and ADEX were used in this stud to examine the composite resin particles and elements. With the SEM method, the contrast obtained with the electron signal was very useful for showing the differences between the resin matrix and filler particles. Consequently, the SEM micrographs showed a dramatic dissimilarity between the particles of the tested groups. However, the DEAX micrographs revealed that Beautifil II majesty had a more complex structure (micrographs. 1). Also, Beautifil II presented a significantly higher Vickers hardness value (Tables 2, 3).
Taken together, these results suggested that whereas composite resins could present similar compositions, the different technologies employed to manufacture them could cause them to possess dramatically different properties and particles and elements; showing that they contained splintered pre-polymerized nanofiller complexes, different new composites have been developed based on nanoparticle filler technology.
Scanning Electron Microscope (SEM) Analysis
For the Ceram.x.mono group the SEM micrographs showed that they were alike where their homogeneously smaller filler particles were shown because these composite resins were filled with nanoparticles; there were, nonetheless, some micro particles [29]. These particles seemed to be organic filler particles including smaller nanoparticles of inorganic fillers, for ISP Empress Direct, it contained some apparent Nano filler complexes (Micrograph 1) [3].
Surface microhardness (VHN) analysis
Composite resins are routinely classified on the basis of filler particle size for the purposes of research, clinical applications, and communications [3, 4, 30]. However, gaping variations exist when defining the average particle size or range of sizes of the particles for their material grouping [3, 4]. The investigation of filler technology is particularly important because filler content has been shown by numerous authors to strongly correlate with the mechanical and physical properties of composite resins [8-11, 30]. Furthermore, the storage media effect on the surface topography of the specimens, without the parameter of bleaching, was not controlled in this study. However, the specimens were stored in distilled water within 7 days in order to reduce the effects of monomers launching onto the surface of the composite resin showed that water uptake can result in stress corrosion and complete or partial debonding of fillers, which in turn can lead to increased surface roughness, reported that acid-based reactions and fluoride release are related to water uptake [31-33].
Indeed, the results from per-bleaching showed that this highly significant difference in surface roughness might have been due to differences in filler loadings; these results coincide with the results of Hubbezoglu et al [16] and Hannig et al [34], who demonstrated that the filler particles and resin matrix have different levels of response, which can lead to variations in surface roughness. Therefore, the difference in the composite roughness values obtained after applying the same bleaching regime could be related to the organic matrix and its different polymers, filler content, and size.
The results obtained with the Beautifil II composite restorations were highly significant; the decrease in surface roughness values observed with this composite might have been due to the fact that its surface was composed of S-PRG glass ionomer particles. Moreover, the oxidizing effect of bleaching through the (O-) and (OH-) free radicals may erode the Beautifil II matrix of glass ionomer particles and S-PRG fillers, which would result in surface wash off with the exposure of the cores to silica. Consequently, the significant decreases in surface roughness with the exposure time to bleaching may result in an additional oxidizing effect on the matrix, leading to water uptake by the glass ionomer with complete or partial debonding of the silica, thus causing a reduction in surface roughness. Following this, the water uptake would result in the formation of spheroidal bodies which have an eggshell-like appearance on the surface. Similarly, Kuhn, Wilson, explained the mechanism of such erosion but using a different composite material matrix, where they reported that the dissolution of glass ionomer includes a three-step surface wash off, diffusion in the solid state, and surface corrosion [35]. These results agree with Jefferson et al [36], who examined the effect of peroxidecontaining products on glass ionomer, and reported that the matrix of the specimens showed surface wash off and erosion with longer exposure of the cores to Hassanaly et al [37].
In addition, the highly significant decreases in surface roughness observed with both the Beautifil II and IPS Empress Direct composite resins might be explained by the fact that their resin matrix is composed of urethane dimethacoilate (UDMA) and (BIS-GMA), which results in harder matrices. However, this disagrees with Takahashi et al [38], who used the Z250 resin matrix with a different bleaching material applied for a different duration. In their study, the soft phase of surface was easily abraded by bleaching urethane dimethacoilate (UDMA) and (BIS-GMA), which are contained in the Z250 resin and form fewer double bonds, which results in a slightly softer matrix, which in turn leads to the debonding of the filler from the resin resulting in a rough surface. Moreover, this roughening can result in the erosion of the matrix, and the consequent debonding of the resin-filler interfaces would lead to the dislodgement and elution of the fillers. Thus, the smaller the size of the bleached particles, the smoother the surface will be obtained, which was the case with both the Beautifil II and IPS Empress Direct composite resins. Indeed, it is known that small fillers enhance the packing of particles, producing a composite with smaller inter-particle spacing, which coincides with the results of Moraes et al [25] and Hassanaly et al [37].
In addition, the modulus of elasticity of the materials used in restorations with lower modulus of elasticity tend to bend more like a tooth structure when subjected to a masticatory load and may flex and be retained, our study concurs with the previous findings [39, 40]. Our study revealed increases in smoothness and hardness only in the Beautifil II and IPS Empress Direct composite resin restorations. The increases in surface hardness are associated with an unaffected hardness of core (the deep subsurface of a resin restoration), these results explain the absence of cracking during surface roughness measurements (AFM micrographs 14 to 19, SEM micrograph 1.) and surface micro-hardness tests (Table 3) and will eventually have an increasing impact force (preventing fracturing of the restoration during mastication). These aspects are obvious evidence for the absence of cracking during surface hardness tests and surface roughness micrographs.
Fourier Transform Infrared Spectrometer (FTIR) Analysis
The degree of conversion% is an important tool to determine the final physical, mechanical, and biological properties of composite resins, since it has been shown that composite properties tend to improve as the degree of conversion attained during photo-polymerization is increased [41]. In addition, increased cure may result in a lower amount of uncured, potentially leachable monomer, leading to a more biocompatible restoration [42]. Moreover, uncured functional groups can act as plasticizers, reducing the mechanical properties [43]. The minimum degree of conversion percentage (%) for a clinically satisfactory restoration has not been precisely established. Nevertheless, a negative correlation of in vivo abrasive wear depth with a percentage degree of conversion (%) has been found for values in the range of 55-65%. This suggests that, at least for occlusal restorative layers, degree of conversion (%) values below 55% may be contraindicated [44]. According to some authors the dimethacrylate monomers used in restorative materials exhibit considerable residual unsaturation in the final material, with a degree of conversion (%) ranging from 55 to 75% under conventional irradiation conditions [45-47]. A lower degree of conversion could affect the longevity of the composite restoration, because an incomplete conversion may result in unreacted monomers, which might dissolve in a wet environment. In addition, reactive sites (double bonds) are susceptible to hydrolization or oxidation and, thereby, lead to a degradation of the material [48, 49].
Energy Dispersive X-ray Spectroscopy (EDAX) Analysis
The increasing in the micro hardness as result of the photopolymerization of the composite resins exhibits an incomplete conversion of the double bonds. This leaves a significant proportion of the methacrylate group's unreacted, depending on the monomer and filler composition, initiator system, and light-curing procedure [50, 51]. Increasing the conversion results in higher surface hardness, flexural strength, flexural modulus, fracture toughness, and diametral tensile strength. This improvement in its properties may be because of increased cross-linkage [41, 52].
Atomic Force Microscope (AFM) Analysis
Studies found a decline of silica and silisium content and a washout after bleaching, indicating the erosion of the composite surfaces [36]. The filler load directly related to the surface area that has been taken up by filler particles versus resin matrix, since roughening suggested resulting from erosion of the matrix, the consequent debonding of the resinfiller interfaces would lead to dislodgement as to elution of the fillers. Thus, the smaller the size of the leached particles the smoother the resulting surface that happens the in Beautifil II and ISP Empress Direct groups. Indeed, it is known that small fillers enhance the packing of the particles, producing a composite with smaller inter-particle spacing [25, 37, 53, 54]. They revealed reductions in the feldspatic porcelain surface (SiO2) content of 4.82 and 4.44% for the Nite-White and Rembrandt bleaching agents, respectively. The (SiO2) forms the matrix and would thus affect the surface hardness. This small amount of released (SiO2) was not considered to be of clinical significance. The difference of the ultrastructure, size of filler particles, volume/weight fraction of the filler, and the chemical composition of the composite resins had an effect on the Vickers hardness and the reason of the difference of our present results [55]. Few studies have been performed that deal with the effects of bleaching agents on restorative materials. However, it is difficult to compare the results of those studies due to the assortment of restorative materials used and no published studies that are available on the effects of bleaching on some of restorative materials are used in the present study. The differences in the results of different studies might be attributed to different methodologies and materials involved [15]. Kim et al [30], stated that composite resins with a higher hardness produced a positive effect on wear resistance. In a study by Rode et al [56], it was reported that light curing units promote a similar degree of conversion and microhardness for the same composite resin. However, the shape of the filler particles, as well as the particle size and filler content, can significantly affect the light transmittance characteristics. Arikawa et al [57] and Emami et al [58] suggesting that the characteristic of incident light also affects light absorbance by dental composites. Arikawa et al [59] concluded that markedly inhomogeneous light emitted from a light curing unit could result in inhomogeneous polymerization in some areas of the restoration below the light guide tip.
The composition of composite resins is complex and the use of EDAX obtained general information about the elemental compositions of the composite resins [29]. The common resin matrix elements such as (C) and (O) were detected in all the materials with comparable concentrations. Nevertheless, Ceram.x.mono presented the highest concentration of (C) after bleaching, suggesting that this composite was slightly lower-filled than the others and this could be related to its lowest Vickers hardness mean value. Secondary caries at the tooth-restoration margin is the most frequently cited reason for restoration replacements [60]. Based on previous studies that measured fluoride release from dental materials, fluo-ride has been shown to be effective in preventing caries development [61, 62]. Among the composite resins evaluated in this study, fluoride was detected only in Beautifil II, which contained a surface pre-reacted glass ionomer (S-PRG) filler [29]. Nanocomposites claim to provide the esthetic properties required for anterior restorations, as well as mechanical properties. In addition, some scientific data from in vitro investigative studies indicated that the majority of nanofilled composites led to higher surface quality and superior polish retention. However, in a study by Jung et al., Filtek Supreme (which is filled with nanoparticles) showed a surface quality that was no better than that of a traditional hybrid composite after polishing [63].
In the presented study, the SEM and AFM micrographs of the restorative composite resins showed a great diversity of particles and elements with regard to filler particle size, shape, and content. These findings were consistent with the significant differences in filler particle numbers and sizes of composite resins reported by Jaarda et al [4]. The size and characteristics of filler particles have been considered as significant factors in the rate of wear of composites [4, 12, 64, 60].
CONCLUSION
Within the limitations of the current study, the following conclusions were drawn:
1. The surface topography, which includes the surface roughness and surface micro-hardness, revealed increases in the smoothness and hardness of the Beautifil II and IPS Empress Direct composite resin restorations. This increasing surface micro-hardness was associated with unaffected cores, and resulted in increases in impact force (preventing fracturing of the restoration during mastication). Increasing surface microhardness could result in decreasing wear rates and extending the restorations' lives. 2. These results explain the absence of cracking during surface roughness measurements (SEM and AFM micrographs) and surface micro-hardness tests. 3. Increased surface microhardness values of the bleached Beautifil II and ISP Empress Direct groups and decreased values in the Ceram.x.mono group due to chemical changes and element rearrangements. 4. Decreased surface roughness values of the bleached Beautifil II and ISP Empress Direct groups and increased values in the Ceram.x.mono group due to chemical changes and element rearrangements. 5. The revolutionary technologies employed to manufacture composite resins have different changed their properties, especially in terms of the size, shape, and distribution of the filler particles. 6. The SEM, DEAX, and AFM methods used in this study were useful for examining the particles and elements of the composite resins. The size of the filler particles and the chemical composition of a composite resin had an impact on its Vickers hardness. 7. Increasing smoothness represented a healthier field.
REFERENCES
- Kugel G, Ferreira S, Sharma S, Barker M, et al. (2009). Clinical trial assessing light enhancement of in-office tooth whitening. Journal of Esthetic and Restorative Dentistry. 21(5), 336-347.
- Labella R, Lambrechts P, Meerbeek BV and Vanherle G. (1999). Polymerization shrinkage and elasticity of flowable composites and filled adhesives. Dental materials Journal. 15(2), 128-137.
- Lang R, Jaarda M and Wang F. (1992). Filler particle size and composite resin classification systems. Journal of oral rehabilitation. 19(6), 569-584.
- Jaarda J, Lang W and Chris A. (1993). Measurement of composite resin filler particles by using scanning electron microscopy and digital imaging. The Journal of prosthetic dentistry. 69(4), 416-424.
- Ottenga E and Mjor A. (2007). Amalgam and composite posterior restorations: curriculum versus practice in operative dentistry at a US dental school. Operative dentistry Journal. 32(5), 524-528.
- Zandinejad A, Atai M and Pahlevan A. (2006). The effect of ceramic and porous fillers on the mechanical properties of experimental dental composites. Dental Materials Journal. 22(4), 382-387.
- Domingo C, Arcis W, Osorio E, Osorio R, et al. (2003). Hydrolytic stability of experimental hydroxyapatite-filled dental composite materials. Dental Materials Journal. 19(6), 478-486.
- Faltermeier A, Rosentritt M, Faltermeier R, Reicheneder C, et al. (2007). Influence of filler level on the bond strength of orthodontic adhesives. The Angle orthodontist Journal. 77(3), 494-498.
- Xu K. (2000). Whisker-reinforced heat-cured dental resin composites: effects of filler level and heat-cure temperature and time. Journal of dental research. 79(6), 1392-1397.
- Willems G, Lambrechts P, Braem M, Vanherle G, et al. (1993). Composite resins in the 21st century. Quintessence international Journal. 24(9), 641-658.
- Silva RD, Simamoto-Junior C, Mota SD and Soares J. (2007). Mechanical properties of light-curing composites polymerized with different laboratory photo-curing units. Dental materials journal. 26(2), 217-223.
- Lim S, Ferracane L, Condon R and Adey D. (2002). Effect of filler fraction and filler surface treatment on wear of micro filled composites. Dental Materials Journal. 18(1), 1-11.
- Cristina J, Christine W and Palma R. (2007). Microhardness of posterior composite resin at varying post irradiation time. Journal of Oral Science. 15(4), 77-80.
- Villalta H, Okte Z, Garcia-Godoy F and Powers M. (2006). Effects of staining and bleaching on color change of dental composite resins. The Journal of prosthetic dentistry. 95(2), 137-142.
- Mohammadi N, Kimyai S, Abed-Kahnamoii M, EbrahimiChaharom E, et al. (2012). Effect of 15% carbamide peroxide bleaching gel on color stability of giomer and microfilled composite resin: An in vitro comparison. Medicina Oral, Patologia Oraly Cirugia Bucal Journal. 17(6), e1082-e1088.
- Hubbezoglu I, Akaoglu B, Dogan A, Keskin S, et al. (2008). Effect of bleaching on color change and refractive index of dental composite resins. Dental materials journal. 27(1), 105-116.
- Palaniappan S, Bharadwaj D, Mattar L, Peumans M, et al. (2011). Nanofilled and microhybrid composite restorations: Five-year clinical wear performances. Dental materials Journal. 27(7), 692-700.
- Wattanapayungkul P and Yap U. (2003). Effects of in-office bleaching products on surface finish of tooth-colored restorations. Operative dentistry Journal. 28(1), 15-19.
- Ratto V, Langry C, Rudd E, Balhorn L, et al. (2004). Force spectroscopy of the double-tethered concanavalin-A mannose bond. Biophysical journal. 86(4), 2430-2437.
- Alameedee A, Ragab A, Osman E, Hussein F. (2014). Effect of Aqueous Suspensions of Titanium Dioxide in Photoreactor with Radiation Source on Color Changes of Three Contemporary Composite Resins. Asian Journal of Chemistry. 26(14), 4507-4512.
- Atali Y and Topbasi B. (2011). The effect of different bleaching methods on the surface roughness and hardness of resin composites. Journal of Dentistry and Oral Hygiene. 3(2), 10-17.
- Mujdeci A and Gokay O. (2006). Effect of bleaching agents on the microhardness of tooth colored restorative materials. Journal of Prosthetic Dentistry. 95(4), 286-289.
- Costa X, Galvao R, Jacomassi P, Bernardi I, et al. (2011). Continuous and gradual photo-activation methods: influence on degree of conversion and crosslink density of composite resins. Journal of thermal analysis and calorimetry. 103(1), 219-227.
- Rastelli S, Jacomassi P and Bagnato S. (2008). Degree of conversion and temperature increase of a composite resin light cured with an argon laser and blue LED. Laser physics. 18(12), 1570-1575.
- Moraes P, Rocha F, Menegazzo M, Arujo D, et al. (2008). Infrared spectroscopy: a tool for determination of the degree of conversion in dental composites. Journal of Applied Oral Science. 16(2), 145-149.
- Cooley R and Burger K. (1991). Effect of carbamide peroxide on composite resins. Quintessence International Journal. 22(10), 817-821.
- Bailey T and Swift EJ. (1992). Effects of home bleaching products on composite resin. Quintessence International Journal. 23(7), 489-494.
- Monaghan P, Trowbridge T and Lautenschlager E. (1992). Composite resin color change after vital tooth bleaching. Journal of Prosthetic Dentistry. 67(6), 778-781.
- Vilchis J, Hotta Y and Yamamoto K. (2008). Examination of six orthodontic adhesives with electron microscopy, hardness tester and energy dispersive X-ray microanalyzer. The Angle orthodontist Journal. 78(4), 655-661.
- Kim H, Ong L and Okuno O. (2002). The effect of filler loading and morphology on the mechanical properties of contemporary composites. The Journal of prosthetic dentistry. 87(6), 642-649.
- Polydorou O, Hellwig E, Auschill M. (2006). The effect of different bleaching agents on the surface texture of restorative materials. Operative dentistry Journal. 31(4), 473-480.
- Soderholm M, Zigan M, Ragan M, Fischlschweiger W, et al. (1984). Hydrolytic degradation of dental composites. Journal of Dental Research. 63(10), 1248-1254.
- Yap U, Tham Y, Zhu Y and Lee K. (2002). Short-term fluoride release from various aesthetic restorative materials. Operative dentistry Journal. 27(3), 259-265.
- Hannig C, Duong S, Becker K, Brunner E, et al. (2007). Effect of bleaching on subsurface micro-hardness of composite and a polyacid modified composite. Dental Materials Journal. 23(2), 198-203.
- Kuhn T, Wilson D. (1995). The dissolution mechanism of silicate and glass ionomer dental cements. Biomaterials Journal. 18, 128-132.
- Jefferson L, Zena B and Giammara B. (1992). Effects of carbamide peroxide on dental luting agents. Journal of endodontics. 18(3), 128-132.
- Hassan Aly R, Mosallam R and Abialhassan M. (2009). Effect of ozonated water on Marginal integrity and surface topography of class V restorative system. Cairo Dental Journal. 2, 281-293.
- Surface characterization of current composites after toothbrush abrasion. Takahashi R, Jin J, Nikaido T, Tagami J, et al. (2013). Surface characterization of current composites after toothbrush abrasion. Dental materials journal. 32(1), 75-82.
- Heymann O, Sturdevant R, Bayne S, Wilder D, et al. (1991). Examining tooth flexure effects on cervical restorations: A two-year clinical study. Journal of the American Dental Association. 122(5), 41-47.
- Demirc M, Sancakli SH and Uysal o. (2008). Clinical evaluation of a polyacid modified resin composite (Dyract) in class V carious lesions: 5years results. Journal of Clinical Oral Investgations. 12(2), 157-163.
- Lovell G, Lu H, Elliott E, Stansbury W, et al. (2001). The effect of cure rate on the mechanical properties of dental resins. Dental Materials Journal. 17(6), 504-511.
- Yap U, Soh S, Han S, Siow S, et al. (2004). Influence of curing lights and modes on cross-link density of dental composites. Operative Dentistry-University of Washington. 29(4), 410-415.
- Asmussen E and Peutzfeld A. (2001). Influence of pulsedelay curing on softening of polymer structures. Journal of Dental Research. 80(6), 1570-1573.
- Soares S, Liporoni and Martin A. (2007). The effect of soft-start polymerization by second generation LEDs on the degree of conversion of resin composite. Operative dentistry. 32(2), 160-165.
- Kurachi C, Tuboy M, Magalhaes V and Bagnato S. (2001). Hardness evaluation of a dental composite polymerized with experimental LED-based devices. Dental Materials Journal. 17(4), 309-315.
- Rode M, Kawano Y and Turbino L. (2007). Evaluation of curing light distance on resin composite microhardness and polymerization. Operative Dentistry Journal. 32(6), 571-578.
- Hammesfahr D, OConnor T and Wang G. (2002). Lightcuring technology: past, present, and future. Compendium of Continuing Education in Dentistry Journal. 23(9), 18-24.
- Yap U, Wong Y and Siow S. (2002). Composite cure and shrinkage associated with high intensity curing light. Operative dentistry Journal. 28(4), 357-364.
- Oysaed H, Ruyter E and Kleven S. (1988). Release of formaldehyde from dental composites. Journal of dental Research. 67(10), 1289-1294.
- Cook D, Metcalf D, Nicola A, Burgess W, et al. (1985). Malignant transformation of a growth factor-dependent myeloid cell line by Abelson virus without evidence of an autocrine mechanism. 41(3), 677-683.
- Prasanna V, Prabha N and Tharanatha N. (2007). Fruit ripening phenomena - An overview. Critical reviews in food science and nutrition Journal. 47(1), 1-19.
- Ruyter E and Svendsen A. (1978). Remaining methacrylate groups in composite restorative materials. Acta odontol scand journal. 36(2), 75-82.
- Anusavice K and Phillips R. (2012). Phillips science of dental materials, 12th edition. USA: Elsevier Health Sciences.
- Anusavice K. (1996). Reducing the failure potential of ceramic based restorations. Part 1: Metal ceramic crowns and bridges. Journal of General Dentistry. 44(6), 492-494.
- Vilchis S, Yamamoto S, Kitai N and Yamamoto K. (2009). Shear bond strength of orthodontic brackets bonded with different self-etching adhesives. American Journal of Orthodontics and Dentofacial Orthopedics. 136(3), 425-430.
- Rode M, Kawano Y and Turbino L. (2007). Evaluation of curing light distance on resin composite microhardness and polymerization. Operative Dentistry Journal. 32(6), 571-578.
- Arikawa H, Kanie T, Fujii K, Takahashi H, et al. (2008). Effect of inhomogeneity of light from light curing units on the surface hardness of composite resin. Dental materials journal. 27(1), 21-28.
- Emami N, Sjodahl M and Soderholm M. (2005). How filler properties, filler fraction, sample thickness and light source affect light attenuation in particulate filled resin composites. Dental Materials Journal. 21(8), 721-730.
- Arikawa H, Kanie T, Fujii K, Takahashi H, et al. (2007). Effect of filler properties in composite resins on light transmittance characteristics and color. Dental materials journal. 26(1), 38-44.
- Faltermeier A, Rosentritt M, Reicheneder C and Mussig D. (2006). Experimental composite brackets: influence of filler level on the mechanical properties. American Journal of Orthodontics and Dentofacial Orthopedics. 130(6), 699, e9-e14.
- Xu K, Weir D, Sun L, Takagi S, et al. (2007). Effects of calcium phosphate nanoparticles on Ca-PO4 composite. Journal of dental research. 86(4), 378-383.
- Chan D, Yang L, Wan W and Rizkalla S. (2006). Fluoride release from dental cements and composites: a mechanistic study. Dental Materials Journal. 22(4), 366-373.
- Jung M, Eichelberger K and Klimek J. (2007). Surface geometry of four nanofiller and one hybrid composite after onestep and multiple-step polishing. Operative Dentistry Journal. 32(4), 347-355.
- Atai M, Yassini E, Amini M and Watts C. (2007). The effect of a leucite-containing ceramic filler on the abrasive wear of dental composites. Dental materials Journal. 23(9), 1181- 1187.