Previous Issues Volume 3, Issue 1 - 2018
Ultrasound-Guided Percutaneous Needle Treatment and Steroid Injection for Calcific Tendinopathy of the Shoulder: Can the Orthopedic Surgeon do it?
Stefan Moosmaye*, Ingerid B. Aasen
Martina Hansens Hospital, Department of Orthopedic Surgery, P.O.B. 823, 1306 Sandvika, Norway.
Corresponding Author: Stefan Moosmayer, Martina Hansens Hospital, Department of Orthopedic Surgery, P.O.B. 823, 1306 Sandvika, Norway,Tel: +47 67521750; E-Mail: [email protected]
Received Date: 07 May 2018 Accepted Date: 18 May 2018 Published Date: 23 May 2018
Copyright © 2018 Moosmayer S
Citation: Moosmayer S and Aasen IB. (2018). UltrasoundGuided Percutaneous Needle Treatment and Steroid Injection for Calcific Tendinopathy of the Shoulder: Can the Orthopedic Surgeon do it?.Mathews J Orthop. 3(1): 020.
ABSTRACT
Background: Ultrasound-guided percutaneous needle treatment and steroid injection (barbotage) for calcific tendinopathy is usually performed by radiologists experienced in interventional ultrasound. We wanted to assess the early learning curve of an orthopedic surgeon.
Methods:One orthopedic surgeon without prior experience with the method treated 52 consecutive patients with barbotage. Shoulder function was measured repeatedly over 24 months on the American Shoulder and Elbow Surgeons (ASES) score and ultrasound and x-ray examination were performed at baseline and after 3 and 24 months. Patients with insufficient effect from barbotage were offered surgical treatment. Primary outcome was the change on the ASES score from baseline to 24 months.
Results: After 24 months, 15 patients had crossed over to surgery, 37 patients were still treated by barbotage-only. In the barbotage-only group the ASES score had improved by39.2 ± 18.6 points from 47.6 to 86.8 points (p = .001). Deposit size at baseline, volume of extracted calcium and change of deposit appearance on x-rays were compared between patients with and without effect from barbotage but were found to be unrelated to the outcome.
Discussion:Our results may encourage orthopedic surgeons with access to ultrasound equipment to perform barbotage on patients suffering from calcific tendinopathy.
Keywords: Calcific Tendinopathy; Ultrasound-Guided Needle Treatment; Barbotage; Interventional Ultrasound; Calcific Deposit; Shoulder
INTRODUCTION
Calcific tendinopathy is a painful disorder of the shoulder of unknown etiology [1]. It is characterized by the formation of deposits of calcium crystals in one or several of the rotator cuff tendons. Tendon inflammation located around the deposit and increased intratendineous pressure are considered to contribute to pain. The course of the disease is often self-limiting with spontaneous calcium resorption and resolution of symptoms over several months [2]. Oral analgesics or a steroid injection together with physiotherapy are recommended as primary treatment measures. Incases with persisting symptoms and delayed, incomplete or absent resorption, removal of the calcification may provide symptomatic relief. Different methods including ultrasoundguided percutaneous needle treatment and steroid injection (barbotage), extracorporal shockwave therapy (ESWT), and surgical treatment are possible treatment alternatives.
Barbotage was introduced in the early 1990s as a treatment measure for calcific tendinopathy of the shoulder [3]. It is miniinvasive, well tolerated by the patients and can be performed as an outpatient procedure. The technique is performed by introducing one or two needles into the calcific deposit under sonographic guidance and by flushing out the calcification with saline water. A supplementary steroid injection is given to control the inflammation that exists around the deposit or may arise if calcific material leaks into the bursa during the procedure [4]. The efficacy of barbotage has been assessed in several cohort studies and good short- and medium-term results have been reported [5-11]. A systematic review of the efficacy of barbotage in the treatment of calcific tendinopathy found the technique to be safe and effective with an estimated average of pain improvement of 55% [12]. In comparison studies it was shown that, after one year, barbotage was superior to ESWT while when compared to a steroid injection it was superior after one year but not after five years [13-15]. Treatment by barbotage in earlier studies has been performed by experienced radiologists and it has been stated that the technique must be carried out by radiologists specifically trained in musculoskeletal and interventional ultrasound [8]. Now, as diagnostic ultrasound of the shoulder has been adopted by many orthopedic surgeons this statement may be challenged.
The primary aim of the study was therefore to assess prospectively short- and medium-term results from barbotage performed by an orthopedic surgeon with limited experience with the method. We hypothesized that the results from the oorthopedist’s early learning curve would be comparable to those reported from experienced radiologists. Secondary aims of the study were to explore associations between treatment outcomes and factors such as deposit size and radiological appearance at baseline, volume of the extracted calcium and change of the radiological deposit appearance after treatment.
PATIENTS AND METHODS
The study was performed in one Norwegian secondary care institution as a prospective cohort study with follow-up over 24 months. It was approved by our institution's q ethics committee (Committee for Medical Research Ethics - South East Norway, reference number 2012/773). Written informed consent was obtained from all participants prior to inclusion.
Barbotage was performed by one orthopedic surgeon (SM) experienced in diagnostic ultrasound of the shoulder but with no experience in ultrasound-guided lavage of calcific deposits. Before study start the orthopedic surgeon had observed two barbotage procedures performed by an experienced colleague. Further knowledge was acquired by studying method descriptions in relevant publications and by viewing instructional videos. From July 2012 to August 2013, he treated by barbotage 52 consecutive patients who were referred by general practitioners for further management of long standing shoulder pain from calcific tendinopathy. Criteria for inclusion and exclusion are given in Table 1.
Table 1: Criteria of inclusion and exclusion.
| Inclusion criteria |
|---|
| At least 6 months of shoulder pain Moderate to severe pain localized on the top and/or lateral side of the shoulder, aggravated by activities above shoulder level Pain at night when lying on the affected shoulder A painful arc together with a positive Hawkin’s test or Neer’s sign for impingement [16, 17] A finding of one or more calcifications =5 mm in size on a standard anterior posterior radiograph, localized proximally to the greater tubercle, taken not more than 4 weeks prior to the intervention and classified as Mole type A, B or C [18, 19] A sonographic finding of one or more calcifications =5 mm in size on the short or long axis view, localized in the supraspinatus or infraspinatus tendon |
| Exclusion criteria |
| An age < 18 years A subacromial injection with a corticosteroid or treatment by ESWT during the last 3 months before inclusion Sonographic signs of a rotator cuff tear (full or partial thickness) or of a dislocated long head of the biceps tendon The presence of clinical and radiological signs of a recent spontaneous release of the calcific deposit such as a sudden change in size or density of the deposit on ultrasound together with an acute onset of extreme shoulder pain Clinical signs of shoulder instability, glenohumeral arthritis, AC pathology, inflammatory arthropathy, fibromyalgia, frozen shoulder or cervical radiculopathy Previous surgery or barbotage of the relevant shoulder Knowledge of an ongoing pregnancy and women who are nursing An inability to comply with follow-up |
Data collection at baseline Demographic patient information was collected and the size of the deposit was measured sonographically on short and long axis views. The appearance of the calcification on radiographs was classified according to Molé into type A (sharply delineated, dense, homogenous), type B (sharply delineated, dense, multiple fragments) or type C (heterogeneous, fluffy) [16, 17]. Shoulder function was measured on the self-report section of the ASES score [20]. Baseline results are given in Table 2.
Table 2: Patient and deposit characteristics at baseline (n = 52 if not otherwise stated).
| Characteristics | |
|---|---|
| Age (years) | 54 (8.1) |
| Gender (female/male) | 35/17 |
| ASES score (points) | ASES score (points) |
| VAS score for pain (cm) | 5.7 (1.6) |
| Dominant side affected | 33 |
| Right side dominant | 46 |
| Smoker | 12 |
| Duration of symptoms (months) | 33 (21.7) |
| Bilateral calcification | 8 |
| Traumatic onset of symptoms | 5 |
| Earlier treatment | |
| Physiotherapy | 43 |
| Steroid injection | 26 |
| NSAID | 12 |
| Shock wave treatment | 9 |
| Acupuncture | 4 |
| Chiropractor | 3 |
| None | 4 |
| Deposit size on ultrasound (mm) | |
| Anterior-posterior plane, horizontally | 12.6 (3.4) |
| Anterior-posterior plane, vertically | 6.2 (1.1) |
| Medial-lateral plane, horizontally | 14.1 (3.5) |
| Location related to rotator cuff tendons | |
| Supraspinatus | 33 |
| Supraspinatus and Infraspinatus | 16 |
| Infraspinatus | 3 |
| Mole classification (n=51) | |
| Type A | 33 |
| Type B | 17 |
| Type C | 1 |
ASES, American Shoulder and Elbow Surgeons; VAS, Visual analog scale; NSAID, Non-steroidal Anti-inflammatory Drugs. Continuous variables are presented as mean (standard deviation). Categorical variables are presented as number of patients.
INTERVENTION
The intervention was performed with the patient placed in the supine position with the arm internally rotated (hand behind the back). After sterile skin preparation, with the transducer wrapped in a sterile drape, and by using sterile jelly, the calcific deposit was sonographically identified, usually on the lateral transversal view. A Sonoline Antares scanner (Siemens Medical Systems, Erlangen, Germany) equipped with a linear-array transducer of 8.5 to 11.5 MHz was used. A 21-gauge needle was then introduced into the shoulder under sonographic guidance, and the pathway and the subacromialsubdeltoid bursa were anesthetized with 10 ml of 1% lidocain hydrochlorid with hydrochloride with 5 μg/ml of adrenaline
A new 18-gauge needle connected to a 10 ml syringe with 8 ml of saline solution was used to puncture the calcification with freehand technique and under constant sonographic monitoring. With the tip of the needle placed in the centre of the deposit, the calcification was flushed. If backflow of calcific material was identified in the syringe, lavage of the deposit was performed by successive propulsion and aspiration with the syringe plunger. To avoid reinjection of the calcium into the deposit, the needle and the syringe were hold in the horizontal plane during the procedure. The syringe was substituted when the fluid had become cloudy and the procedure was repeated until the backflow became clear. Extracted calcium was poured into a test tube with a measurement scale with 0.1 ml increments on which the volume of the calcium could be read after it had settled at the bottom. In cases where no material could be extracted, repeated perforation of the deposit was performed to possibly initiate or accelerate spontaneous resorption.
At the end of the procedure, a new 21-gauge needle was introduced into the subacromial-subdeltoid bursa and one injection of a mixture of 9 ml of 1% Lidocain hydrochlorid and 1 ml (20 mg) of Triamcinolon was given. A video of the procedure has been published earlier [21].
Post-intervention treatment After the intervention, normal use of the shoulder was allowed without restrictions, but heavy shoulder labour had to be avoided for 2 weeks. In case of post-interventional pain exacerbation, non-prescription analgesics could be used. All patients were referred to supervised physiotherapy with particular emphasis on the correction of upper body posture and the restoration of scapulothoracic and glenohumeral muscular control and stability. Physiotherapy was given by local physiotherapists and we have no precise information on the exact number of treatments actually performed and whether our treatment recommendations were followed or not.
Data collection at follow-up Repeated scoring on the ASES score was performed after 1 and 4 weeks and after 3, 6, 12 and 24 months with the score change from baseline to 24 month control as the primary outcome measure. Clinical, radiologic and sonographic reassessment was performed after 3 and 24 months. Follow-up radiographs were compared to baseline radiographs and deposits were classified as unchanged, reduced in volume or density, or as nearly or completely disappeared. Assessment of the radiographs was performed independently by two orthopedic surgeons (SM, IBA), blinded for the patients treatment results. In case of differing results, these were discussed until a consensus was achieved.
For assessment of possible influences on the outcome of factors such as deposit size at baseline, radiographic deposit appearance at baseline, volume of extracted calcium, and change of the radiographic deposit appearance after treatment, the study group was divided in treatment successes and treatment failures. A treatment success was defined as an increase on the ASES score exceeding the score’s minimal clinical important change of 17 points after 24 months, a treatment failure as a score change of less than 17 points after 24 months or a crossover to secondary surgery [22].
Change of treatment Patients who reported inadequate improvement during follow-up were re-examined clinically and sonographically by the orthopedic surgeon (SM) no earlier than three months after study treatment was given, and a supplementary ASES score was performed. Patients who were still symptomatic to a degree that required further treatment were offered secondary treatment by acromioplasty if the deposit had disappeared or was less than 5 mm, or by a second barbotage if the deposit still exceeded 5 mm. Surgically treated patients were followed as a separate group (the secondary surgery group) while patients treated with a second barbotage still remained in the barbotage-only group. Follow-up of patients after supplementary treatment was according to the initial follow-up schedule.
Statistical analysis Sample size was calculated for a t-test analysis of our primary outcome, the change of the ASES score from baseline to 24 month follow-up. Based on the score’s minimal clinically important change, which has been reported to be between 12 and 17 points, we calculated sample size for a detectable difference of 15 points, a significance level of 0.05 and a power of 0.9 [22]. With an expected standard deviation of score changes from baseline to final follow-up of 25 points we found a necessary sample size of 31 subjects. As crossover to surgical treatment was expected in at least one third of patients we wanted to include 52 patients.
Outcome data on the ASES score were assessed for normality and are given as means with 95% CI. Changes on the ASES score from baseline to 3 and 24 month follow-ups were assessed by paired t-tests. Deposit size at baseline, radiographic deposit appearance at baseline, volume of extracted calcium, and change of the radiographic deposit appearance during follow-up were compared between treatment successes and failures. Results are expressed in percentages and significance testing was performed by the Fisher's exact test for 2x2 tables and by the Chi squared test for larger tables.
RESULTS AND DISCUSSION
RESULTS
Technical practicability In all 52 cases the barbotage procedure was performed without any immediate or delayed procedural complications. Backflow of calcific material was achieved in 44 cases. In 10 cases the aspirated volume was =0.1 ml, in 14 cases between 0.1 and 0.4 ml and in 20 cases =0.4 ml. In 8 cases no calcific material was aspirated and decompression of the deposit by needling was performed.
Outcome on the ASES score All 52 patients were available for follow-up after 3 and 24 months. At 3 month follow-up all study patients were still in the barbotage-only group, which means that no patient had crossed over to secondary surgery. Improvement on the ASES score after 3 months was by 24.6 points (95% CI 18.3 to 30.8 points, p = .001) from 46.9 points at baseline to 71.5 points.
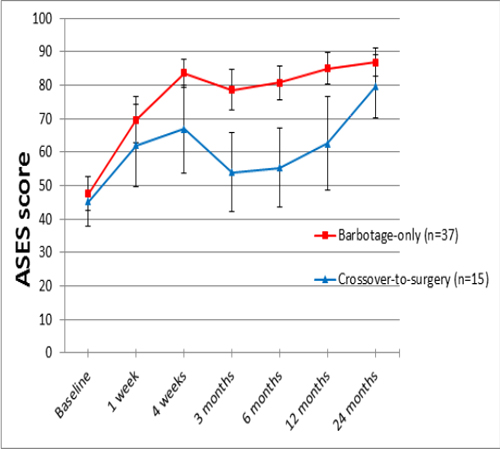
At 24 month follow-up thirty seven patients had been treated by barbotage-only and 15 patients had crossed over to secondary surgery, 12 patients during the first year and 3 patients during the second year. In the barbotage-only group, a second barbotage was performed in 3 cases more than 1 year after the primary treatment. At 24 month follow-up the barbotageonly group showed an improvement on the ASES score of 39.2 points (95% CI 33.0 to 45.4 points, p = .001) from 47.6 points at baseline to 86.8 points. In the crossover group the ASES score had decreased by a mean of 1.4 points from 45.2 points at baseline to 43.8 points at the last measurement before surgery, but increased postoperatively by 34.4 points (95% CI 23.1 to 45.7 points, p = .001) to 79.6 points after 24 months. The development of the ASES score through all follow-ups in both groups is shown in Figure 1.
Figure 1: American Shoulder and Elbow Surgeons (ASES) score (means with 95% confidence intervals) through all follow-ups for the barbotageonly and the crossover-to-surgery groups.
Successes and failures
Four of the patients in the barbotage-only group had increased by less than 17 points after 24 months and were considered as failures, even if they declined any further treatment. Together with the 15 crossovers this resulted in a number of 19 failures (37%) and of 33 treatment successes (63%) after 24 months.
Baseline findings and outcome There was no statistical difference in deposit size at baseline between treatment successes and failures. On sonography, mean deposit size in the medial-lateral plane was 13.8 mm for treatment successes and 14.8 mm for failures (difference 1 mm, 95% CI: -3.0 to 1.1 mm, p = .4). In the anterior-posterior plane the corresponding values were 12.5 mm for treatment successes and 12.7 mm for failures (difference 0.3 mm, 95% CI -2.2 to 1.7 mm, p = .8).
Classification of the calcific deposit on x-rays showed 33 deposits of Mole type A, 17 of type B and 1 of type C. At final follow-up 24 patients (73%) with a type A deposit and 8 patients (47%) with a type B deposit were classified as treatment successes, but the Mole type was found to be unrelated to the outcome as a treatment success or failure (p = .1).
Further analyses were performed to explore possible associations between the outcome and other baseline factors such as patient age, gender, affected side, smoking status, duration of symptoms, condition of the contralateral shoulder and baseline score but none of them showed significance.
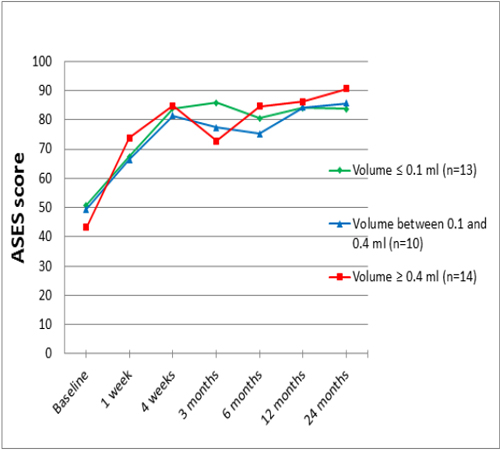
Extracted volume and outcome According to the volume of extracted calcium the study group was divided in three groups with volumes of =0.1 ml (n=18), between 0.1 and 0.4 ml (n=14) and =0.4 ml (n=20). Comparison of the number of treatment successes and failures after 24 months in the three groups showed the lowest percentage of successes in the group with the lowest extracted volume and the highest percentage of successes in the group with the highest extracted volume with 56% (10 of 18), 64% (9 of 14) and 70% (14 of 20) respectively. However, testing of differences in the distribution of treatment successes and failures among the three categories of extracted calcium did not show significance (p = .7)
Figure 2 displays the results for the three groups on the ASES score through all follow-ups for the 37 patients who were treated by barbotage-only.
Figure 2: Results for three subgroups with different volumes of extracted calcium on the American Shoulder and Elbow Surgeons (ASES) score in the barbotage-only group (n=37).
Development of deposit appearance and outcome Repeated x-ray examination was possible for 48 patients after 3 months and for 46 patients after 24 months. An overview over the changes in deposit appearance after 3 and 24 months, whether unchanged, reduced in volume or density, or nearly or completely disappeared, is given in Table 3.
Table 3: Development of deposit appearance over time.
| Development of the deposit on x-rays | Time from baseline | |
|---|---|---|
| 3 months (n=48) | 24 months (n=46) | |
| Unchanged | 10 (21) | 4 (9) |
| Lower volume or density | 25 (52) | 6 (13) |
| Nearly or completely disappeared | 13 (27) | 36 (78) |
Data are given as number of patients with the percentage in parentheses The percentage of nearly or completely disappeared deposits increased from 27% (13 of 48) after 3 months to 78% (36 of 46) after 24 months.
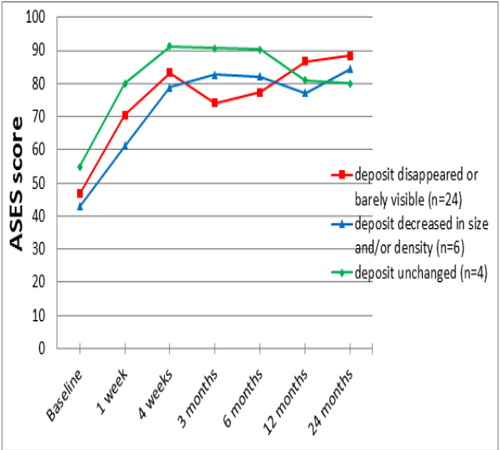
Assessment of the relation between the disappearance or persistence of the deposit and the treatment result, whether a success or a failure, was performed based on the 24-month results. We found 23 (64%) treatment successes among the 36 shoulders where the deposit had nearly or completely disappeared and 8 (80%) treatment successes among the 10 shoulders where the deposit was unchanged or reduced in volume or density. The association between the change in deposit appearance and a successful or unsuccessful outcome was not significant (p = .5), which means that a successful outcome was not more likely in shoulders where the deposit had nearly or completely disappeared compared to shoulders where the deposit was unchanged.
Figure 3 shows the results on the ASES score for the three subgroups with different radiologic development of the deposit after 24 months in the barbotage-only group.
Figure 3: Results on the American Shoulder and Elbow Surgeons (ASES) score for three subgroups with different radiologic development of deposit appearance after 24 months in the barbotage-only group (n=34).
DISCUSSION
In this study, we assessed the early learning curve of an orthopedic surgeon with barbotage of the shoulder. A successful result after 24 months was achieved in 33 of 52 (63%) of the study patients. This is within the range of success rates of 60% to 75% which have been reported in studies, where barbotage was performed by radiologists with specific training in musculoskeletal and interventional ultrasound [5-11, 23]. Also the technical practicability in this early series of an orthopedic surgeon seemed good with a backflow of calcific material in 44 (85%) of the study patients and with no treatment related complications. Vagal reactions as reported in about 5% of patients in earlier studies did not occur in our study, presumably because we performed treatment with the patient in the supine and not in the sitting position [7, 9].
Several studies have presented the results from radiologists with barbotage. Yoo et al. [11] followed 35 shoulders for 6 months on the ASES score and classified those who reported no pain relief or who crossed over to surgery as treatment failures. He found a successful outcome after 6 months in 25 shoulders (71%) with an increase of 36.6 points on the ASES score. Six patients had proceeded to surgery. This is comparable to our interim result after 6 months where 33 of 55 patients (63%) were classified as treatment successes with a rise of 33.1 points on the ASES score (Figure 1) and where 6 patients had crossed over to surgery.
Del Cura et al. [9] followed 67 shoulders for 12 months on the Shoulder Pain and Disability (SPADI) score. Sixteen patients had to be re-treated by a second barbotage. He found an improvement of 35.5 points on the SPADI score which may be compared to an improvement of 39.2 points after 24 months on the ASES score in our barbotage-only group (Figure 1).
Del Castillo-González et al. [7] re-examined 121 patients 12 months after a barbotage procedure on a visual analogue scale (VAS) for pain (0 meaning no pain, 10 meaning worst pain). Nineteen of the patients were treated by re-barbotage after 3 months. He found a decrease on VAS from 7.5 cm to 0.8 cm with no relapses during the second year.
A direct comparison of our results with those from these earlier studies is difficult because of the use of different outcome scores, different follow-up periods and a retrospective study design in some of them. Still, it seems that our results are similar to those achieved by experienced radiologists and this should encourage orthopedic surgeons with experience in diagnostic ultrasound to use barbotage in the treatment of calcific tendinopathy.
Lavage of the deposit in calcific tendinopathy is performed based on the assumption that the deposit causes symptoms.
The exact pathway linking the calcification to symptoms, however, is still a matter of debate. The deposit may act as a mechanical obstacle, as a trigger for an inflammatory reaction or may lead to increased intratendineous pressure. In all cases one would expect a better outcome in patients where a large volume of calcific material can be extracted and in patients where the calcific deposit has disappeared after the treatment. In our study a significant association between the volume of extracted calcium and the outcome could not be demonstrated. Although we found an increasing percentage of treatment successes with increasing volumes of extracted calcium, the association did not reach significance. This is in accordance with the studies from Del Cura et al. [9] and Del Castillo-González et al. [7]. Both compared the results from patients with and without backflow of calcific material and could not find differences in outcome between the two groups. Contrarily, Aina et al. [5] achieved backflow in 33% of their patients and found a clinically significant improvement on the SPADI score in the group with backflow but not in the patients where no backflow had been observed. Our finding of similar results after extraction of small or large volumes of calcific material is compatible with the idea that intratendineous pressure contributes to pain. Extraction of a small volume may be sufficient to relieve intratendineous pressure and a complete emptying of the deposit might not be necessary
When we compared deposit appearance between baseline and after 3 and 24 months, we found that many of the deposits were still visible on x-rays after 3 months but had resolved completely or nearly completely after 24 months (Table 3). This is in accordance with earlier findings [7, 9]. Interestingly, early disappearance of the calcification could also be observed in some of the patients with no backflow during the procedure which may indicate that needling alone can induce a process of resorption.
Assessment of the distribution of treatment successes and failures across the groups with deposit disappearance, partial disappearance or persistence on x-rays showed no association either after 3 months or after 24 months. After 24 months we found 36% of failures in the group where the deposit was no longer visible on x-rays. However, in our study the number of patients with an unchanged or a partially disappeared deposit after 2 years was low (n = 4 and 6) and it is possible that a larger study would have given a different result. This was the case in the studies from De Cura et al. [9] and Del Castillo-González et al.[7] who, after one and two years respectively, found better results in patients where a complete or nearly complete disappearance of the calcification could be demonstrated on x-rays. Still, our finding of 36% of treatment failures in patients with deposit disappearance indicates, that there may be pain sources other than the calcific deposit in these patients.
This study has several limitations, most of them related to the absence of a control group. First, due to the cyclic and often self-limiting course of the disease, improvement in our study patients may, at least partly, be related to a benign natural course of the condition. However, all patients were chronic cases with symptom duration of more than 6 months and showed rapid improvement already 1 and 4 weeks after the treatment (Figure 1). This should indicate, that our results truly reflect the effect of the treatment measures.
Second, the respective contribution to the result of each of our treatment measures is difficult to assess. Barbotage treatment not only includes needling and lavage of the deposit but also a steroid injection and a post-treatment physiotherapy regime. Without a control group it is difficult to assess how much of the treatment effect truly relates to the lavage of the deposit and how much to the adjuvant treatment measures. However, steroid injections and/or physiotherapy alone had been tried in the majority of study patients prior to the study and had not shown sufficient treatment effects. Still, they may have contributed to the study's treatment result when they were used together with lavage of the calcification.
Finally, the sample size for the study was calculated for our main outcome, the change of the ASES score from baseline to 24 months. The number of patients included yields presumably insufficient power for the exploration of possible associations between patient and deposit characteristics on one side and treatment successes and failures on the other and existing associations may have been overlooked.
CONCLUSION
Our results achieved by an orthopedic surgeon with no prior experience with barbotage, were comparable to those reported from more experienced radiologists and might encourage orthopedic surgeons to include barbotage in their armamentarium for the treatment of calcific tedinopathy. With no control group included our study represents a mere description of the orthopedist’s early learning curve but does not allow any conclusions about the true effect of the removal of a calcific deposit on shoulder pain. Our secondary finding of a treatment failure in 13 of 36 patients in whom the deposit had disappeared at 2 year follow-up may question the pathway between the deposit and symptoms.
REFERENCES
- Jerosch J, Strauss JM and Schmiel S. (1998). Arthroscopic treatment of calcific tendinitis of the shoulder. J Shoulder Elbow Surg. 7(1): 30-37.
- Uhthoff HK and Loehr JW. (1997). Calcific tendinopathy of the rotator cuff: pathogenesis, diagnosis and management. J Am Acad Orthop Surg. 5(4): 183-191.
- Farin PU, Jaroma H and Soimakallio S. (1995). Rotator cuff calcifications: treatment with US-guided technique. Radiology. 195(3): 841-843.
- Hackett L, Millar NL, Lam P and Murell GA. (2016). Are the Symptoms of Calcific Tendinitis Due to Neoinnervation and/or Neovascularization? J Bone Joint Surg Am. 98(3): 186-192.
- Aina R, Cardinal E, Bureau NJ, Aubin B, et al. (2001). Calcific shoulder tendinitis: treatment with modified US-guided fine-needle technique. Radiology. 221(2): 455-461
- Bazzocchi A, Pelotti P, Serraino S, Battaglia M, et al. (2015). Ultrasound imaging-guided percutaneous treatment of rotator cuff calcific tendinitis: success in shortterm outcome. Br J Radiol Epub ahead of print. 89(1057): 20150407.
- Del Castillo-González F, Ramos-Álvarez JJ, Rodríguez-Fabián G, González-Pérez J, et al. (2014). Treatment of the calcific tendinopathy of the rotator cuff by ultrasoundguided percutaneous needle lavage. Two years prospective study. Muscles Ligaments Tendons J. 4(2): 407-412.
- De Conti G, Marchioro U, Dorigo A, Boscolo N, et al. (2010). Percutaneous ultrasound-guided treatment of shoulder tendon calcifications: Clinical and radiological follow-up at 6 months. J Ultrasound. 13(4): 188-198.
- Del Cura JL, Torre I, Zabala R, and Legórburu A. (2007). Sonographically guided percutaneous needle lavage in calcific tendinitis of the shoulder: Short- and Long-term results. AJR Am J Roentgenol. 189(3): W128-134.
- Lin JT, Adler RS, Bracilovic A, Cooper G, et al. (2007) Clinical outcomes of ultrasound-guided aspiration and lavage in calcific tendinosis of the shoulder. HSS J. 3: 99-105.
- Yoo JC, Koh KH, Park WH, Park JC, et al. (2010). The outcome of ultrasound-guided needle decompression and steroid injection in calcific tendinitis. J Shoulder Elbow Surg. 19(4): 596-600.
- Lanza E, Banfi G, Serafini G, Lacelli F, et al. (2015). Ultrasound-guided percutaneous irrigation in rotator cuff calcific tendinopathy: what is the evidence? A systematic review with proposals for future reporting. Eur Radiol. 25(7): 2176-2183.
- Del Cura JL, Torre I, Zabala R, and Legórburu A. (2007). Sonographically guided percutaneous needle lavage in calcific tendinitis of the shoulder: Short- and Long-term results. AJR Am J Roentgenol. 189(3): W128-134.
- De Witte PB, Selten JW, Navas A, Nagels J, et al. (2013). Calcific tendinitis of the rotator cuff. A randomized controlled trial of ultrasound guided needling and lavage versus subacromial corticosteroids. Am J Sports Med. 41(7): 1665-1673
- De Witte PB, Kolk A, Overes F, Nelissen R, et al. (2017). Rotator cuff calcific tendinitis: Ultrasound guided needeling and lavage versus subacromial corticosteroids. Am J Sports Med. 45(14): 3305-3314.
- Hawkins RJ and Kennedy JC. (1980). Impingement syndrome in athletes. Am J Sports Med. 8(3): 151-158.
- Neer CS 2nd. (1983). Impingement lesions. Clin Orthop Relat Res. 173: 70-77.
- Molé D, Kempf JF, Gleyze P, Rio B, et al. (1993). Results of endoscopic treatment of non-broken tendinopathies of the rotator cuff. Calcifications of the rotator cuff [in French]. Rev Chir Orthop. 79(7): 532-541.
- Seil R, Litzenburger H, Kohn D, and Rupp S. (2006). Arthroscopic treatment of chronically painful calcifying tendinitis of the supraspinatus tendon. Arthroscopy. 22(5): 521-527.
- Richards RR, An KN, Bigliani LU, Gartsman GM, et al. (1994). A standardized method for the assessment of shoulder function. J Shoulder Elbow Surg. 3(6) 347-352.
- Moosmayer S, Ekeberg OM, Hallgren HB, Heier I, et al. (2017). KALK study: ultrasound guided needling and lavage (barbotage) with steroid injection versus sham barbotage with and without steroid injection - protocol for a randomized, double-blinded, controlled, multicenter study. BMC Musculoskelet Disord. 18(1):138.
- Tashjian RZ, Deloach J, Green A, Porucznik CA, et al. (2010). Minimal clinically important differences in ASES and simple shoulder test scores after nonoperative treatment of rotator cuff disease. J Bone Joint Surg Am. 92(2): 296-303.
- Farin PU, Räsänen H, Jaroma H and Harju A. (1996). Rotator cuff calcifications: treatment with ultrasound-guided percutaneous needle aspiration and lavage. Skeletal Radiol. 25(6): 551-554.