Previous Issues Volume 2, Issue 1 - 2017
To Review the Treatment Response to Combined Intravenous Steroid and Orbital Irradiation in Asian Patients with Active Thyroid Eye Disease (TED)
Hsi-Wei Chung1, 2*,Lay Leng Seah1, 2,
1Singapore National Eye Centre, Singapore.
2Department of Ophthalmology, Yong Loo Lin School of Medicine, National University of Singapore, Singapore.
3Department of Clinical Sciences, Duke-NUS Graduate Medical School, Singapore.
Corresponding Author: Hsi-Wei Chung, Singapore National Eye Centre, 11 Third Hospital Avenue, Singapore, 168751, Tel: (65) 6322 8311; E-Mail: [email protected]
Received Date: 04 Jan 2017 Accepted Date: 09 Jan 2017 Published Date: 13 Jan 2017
Copyright © 2017 Chung HW
Citation: Chung HW and Seah LL. (2017). To Review the Treatment Response to Combined Intravenous Steroid and Orbital Irradiation in Asian Patients with Active Thyroid Eye Disease (TED). Mathews J Ophthalmol. 2(1): 011.
ABSTRACT
Aim: To review treatment response to combined intravenous steroid and orbital irradiation (DXT) in Asians with active thyroid eye disease (TED). Design: Retrospective case series. Methods: Single centre retrospective case series on active TED patients who received IV steroids and DXT at the Singapore National Eye Centre from 2005 to 2015. Patients who did / did not undergo surgical decompression were analysed for baseline difference in risk factors, time to receiving therapy, prior failed treatment and symptoms at presentation according. Results: Sixteen patients (11 Males: 5 Females) were studied [mean ages 55.2 (Std dev 11.4, range 40-86) years]. 81 % (Thirteen patients) had active and severe TED (developed within a year of their thyroid dysfunction diagnosis). Seven patients (43.8%) had compressive optic neuropathy (CON) and eight patients (50%) had exposure keratopathy at presentation. Five patients (31.3%) had failed treatments prior to presentation. The main indication for surgical decompression was CON and progressive active disease. Other indications included reduction of exophthalmos prior to strabismus surgery. Surgical decompression patients received IV steroids at 1.8 (+/- 2.0) months and DXT treatment 4.4 (+/- 4.1) months upon diagnosis of active disease. This is similar to the non-surgical group at 1.7 (+/- 1.5) and 5.3 (+/- 12.2) months respectively. However, the decompression patients needed >1 cycle of IV steroids (n=3) for disease stabilization, higher percentage of patients with risk factors at baseline (diabetes, hypertension, active smoking and failed prior treatments). VISA inflammatory scores were significantly improved (p< 0.0005) in the non-surgical group post combination therapy. The surgical decompression group had stable scores post decompression. No significant deterioration in extraocular motility function (diplopia and ocular restriction) and visual acuity were seen in both groups. Conclusion: Combined intravenous steroid therapy with DXT helps to reduce inflammation and maintain vision in our patients.The study also supports current literature of the early use of radiotherapy with intravenous steroids for active severe TED.
KEYWORDS
Thyroid Eye Disease; Radiotherapy; Intravenous Steroid Therapy; Compressive Optic Neuropathy.
INTRODUCTION
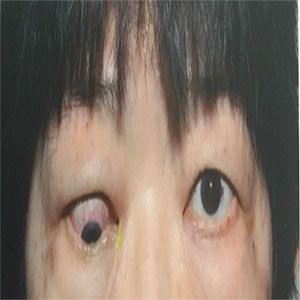
Thyroid Eye Disease (TED) is an autoimmune disorder characterised by orbital soft tissue inflammation, fat hypertrophy and extra ocular muscle enlargement [1]. Rundel has described a biphasic course in TED with an active phase lasting 6-18 months that is followed by an inactive phase [2]. While the majority of patients present with mild symptoms of proptosis and eyelid retraction, a small proportion (5-10%) of patients may develop dysthyroid optic neuropathy, which is caused by apical compression of the optic nerve by enlarged extra ocular muscles [3]. Cellular leucocyte infiltration, deposition of glycosaminoglycans and odema predominate in the active phase of TED. It is this early phase that is targeted by immunosuppressive therapies and radiotherapy with the aim of limiting potential sequelae of restrictive strabismus (Figure 1), orbital inflammation and even compressive optic neuropathy (CON).
Figure 1: Example of severe myopathy with suppression of binocular diplopia in a patient who underwent orbital decompression to reduce orbital congestion prior to strabismus surgery.
Intravenous (IV) steroid therapy is the preferred choice of first line medical therapy for active, severe thyroid eye disease due to its anti-inflammatory properties. However, it has been reported that up to 40% of patients may have limited response to steroid treatment [4]. This is further compounded by cumulative side effects and toxicity profile of intravenous steroids. Radiotherapy has been used as a 2nd line immune suppression for TED for more than 85 years [5]. However recent studies are pointing to synergistic efficacy with intravenous steroid therapy and with implications for reduced risk of compressive optic neuropathy if used early in the active disease stage [6]. The aim of this study was to review the treatment response to combined intravenous steroid and orbital irradiation in Asian patients with active TED.
MATERIALS AND METHODS
This was a single centre retrospective case control review on active TED patients who had received both IV steroids and orbital irradiation (DXT) from 2005 to 2015 at the Singapore National Eye Centre. The study protocol was approved by the Singhealth ethics committee and adheres to the tenets of the Declaration of Helsinki. Data collected also included patient demographics, duration of TED (from onset of thyroid eye symptoms to time of diagnosis of active eye disease), smoking status, pre-existing comorbidities (diabetes, hypertension), thyroid status at presentation (hypo, hyper or euthyroid), previous treatment failure (defined as poor or no response to steroid therapy), time of treatment with IV steroids and orbital irradiation (including number of intravenous steroid infusion cycles). The diagnosis of TED was diagnosed based on George Bartley’s criteria [7, 8]. The VISA (vision, inflammation, strabismus, and appearance) classification protocol developed by Dolman and Rootman was used for classification of TED activity and severity [9]. Active disease was defined by the VISA inflammatory score >4 out of 8. Severe disease was defined by the presence of persistent proptosis, significant soft tissue signs, restrictive myopathy or vision threatening complications of CON, disabling diplopia and or exophthalmos causing exposure keratopathy. Extraocular motility was assessed by the degree of diplopia and ocular restriction. It was measured with Hess and binocular single vision (BSV) charts. Severity of diplopia was graded from 0 to 3, with 0= No diplopia, 1 = diplopia with gaze, 2 = intermittent diplopia, 3 = constant diplopia. Severity of ocular restriction was graded from 0 to 3 and measured in degrees using the Hirschberg’s light reflex, with 0 = >45 degrees, 1 = 30 - 45 degrees, 2 = 15-30 degrees, 3 = < 15 degrees). Inflammatory scores, vision threatening presentations were also documented. Compressive optic neuropathy was diagnosed based on reduced best corrected visual acuity of 2 lines or more and the presence of a relative afferent pupillary defect and/or reduced colour vision and/or visual field defects. Patients with active severe disease were counselled for IV steroid therapy. Assessment of cardiovascular function (blood pressure, heart rate, electrocardiogram), liver enzymes, hepatitis and HIV viral markers, serum glucose and electrolyte levels was carried out prior to IV steroid therapy. Under close monitoring, intravenous (IV) therapy of methylprednisolone was infused at a rate of 1g given over 1 hour each day for 3 days. Pre- and post-infusion monitoring of serum potassium, blood pressure and electrocardiography were performed. Orbital radiotherapy was given to patients with active disease who had restriction in extraocular motility and in those who had poor response to / had intolerance / required multiple cycles of intravenous steroids. DXT was given at a dosage of 20 Gy in 10 fractions over 10 days directed at the posterior two-thirds of the orbit with the globe shielded. We analysed TED patients who underwent surgical decompression versus those who did not for their 6-month post treatment outcomes of visual acuity, inflammatory scores, extra-ocular motility. Complications related to treatment as well as the development of CON was also recorded. Statistical analysis was performed using Graphpad Prism Version 7.0a. The Mann U Whitney rank sum test was used for non-parametric demographic data. Visual acuity, inflammatory scores extra ocular motility measures was analysed for patients in the surgical versus non-surgical groups with Wilcoxon sign rank test post treatment.
RESULTS
Sixteen patients (11 Males: 5 Females) were included in this study (Table 1). Their mean ages 55.2 (std dev 11.4, range 40- 86) years. 81 % (Thirteen patients) had active and severe TED, which had developed within a year of their thyroid dysfunction diagnosis. Time to presentation of active severe disease from the date of thyroid dysfunction diagnosis averaged 5.7 (+/- 3.5) months for the whole study cohort, excluding 3 outliers who had multiple relapses due to poorly controlled thyroid function.
Table 1:
| Active Severe TED Asian Patients (n=16) | |||
|---|---|---|---|
| Underwent Orbital Decompression | |||
| Yes | No | P Value | |
| Total Number | 37.5% (n=6) | 62.5% (n=10) | |
| Sex (Males: Females) | 3 : 3 | 8 : 2 | |
| Male Sex | 50% | 80% | 0.30 |
| Age in Years (Std Dev) | 54.2 (12.3) | 56.8 (10.4) | 0.43 |
| Risk Factors | |||
| Diabetes | 33.3% (n=2) | 10.0% (n=1) | 0.53 |
| Hypertension | 66.6% (n=4) | 30.0% (n=3) | 0.31 |
| Active Smokers at presentation | 33.3% (n=2) | 20.0% (n=2) | 0.99 |
| (An additional 2 were ex-smokers) | |||
| Failed Treatment previously | 16.7% (n=1) | 40.0% (n=4) | |
| Symptoms at Presentation | |||
| Compressive Optic Neuropathy* | 66.6% (n=4) | 30.0% (n=3) | |
| Central Diplopia* | 66.6% (n=4) | 30.0% (n=3) | |
| Exposure* | 50.0% (n=3) | 50.0% (n=5) | |
| ( * pts may have more than 1 presentation) | |||
| Time to presentation from diagnosis | |||
| Months (+/- std dev) | 5.2 (+/- 3.4) | 6.4 (+/-3.6) | 0.64 |
Our study patients had risk factors of male gender (68.8%), older age (55.2 +/ 11.4 years), active smoking status at the time of presentation (25%) and ex-smokers (12.5%). Seven patients had hypertension (43.8%) and three (18.8%) were diabetics. All of our patients were hyperthyroid at presentation (n=16). Seven patients (43.8%) had CON and eight patients (50%) had exposure keratopathy at presentation. The latter group’s symptoms were due to severe myopathy and poor bell’s reflex leading to severe hypotropia in four patients (Figure 1). Amongst the eight (50%) patients who had diplopia, five patients had suppression secondary to severe myopathy. Foreign referral patients made up 19% of the group (n=3) as they received inadequate treatment at the time of diagnosis in their country of residence prior to consultation at our centre. 2 of these 3 foreign patients had CON at presentation. The average time to receiving IV steroid therapy upon diagnosis of active disease at our centre was 1.7 (Std Dev 1.6, range 0-5.2) months. Concurrent receipt of orbital irradiation treatment averaged 5.0 (Std Dev 9.7, range 0.2 to 39.9) months. When patients were analysed based on surgical intervention, the decompression group was found to have received IVMP 1.8 (+/- 2.0) months and DXT treatment 4.4 (+/- 4.1) months upon diagnosis of active disease. The non-surgical group had IVMP and DXT treatment times averaging 1.7 (+/- 1.5) and 1.5 (+/- 0.7) months (excluding an outlier who refused decompression surgery) respectively. This patient received DXT approximately 40 months after diagnosis of active disease. The decompression group had 50% (n=3) of patients who required more than 1 cycle of IVMP to control their disease process. The time between steroid and radiotherapy treatments for the non-surgical group was 1.2 (+/- 1.6) months (excluding the outlier) while the timings for the surgical group was 2.6 (+/- 3.1) months. No significant differences were found between the 2 groups for time to therapy and time interval between therapies. VISA inflammatory scores were significantly improved (p< 0.0005) in the non-surgical group post steroid and radiation therapy. No significant improvement was seen in the surgical decompression group though none of the patients had an increase in their inflammatory scores post decompression. There was also no significant deterioration in extraocular motility function (diplopia and ocular restriction) as well as visual acuity in both groups. One third (n=6) of the patients in this study eventually underwent orbital decompression (Table 2). This group had greater severity of symptoms such as compressive optic neuropathy (n=4), central vision diplopia and/or exposure keratopathy at presentation. There was also a higher percentage of active smokers and patients who had poorly controlled thyroid function with multiple relapses (33%). Failed prior treatment comprised 16.7% of patients (n=1) in the decompression group and 40% (n=4) in the non-surgical group.
Table 2:
| Active Severe TED Asian Patients (n=16) | |||
|---|---|---|---|
| Underwent Orbital Decompression | |||
| Yes | No | P Value | |
| Treatment deatils | |||
| DXT (months +/- Std dev)* | 4.4 (+/- 4.1) | 1.5 (+/- 0.7) | 0.31 |
| IVMP (months +/- Std dev)* | 1.8 (+/- 2.0) | 1.7 (+/- 1.5) | 0.81 |
| (Patients may have had either treatments first) | 2.6 (+/- 3.1) | 1.2 (+/- 1.6) | 0.56 |
| (months +/- Std dev) | |||
| More than 1 cycle of IVMP | 50.0% (n=3) | 30.0% (n=3) | |
All our patients tolerated IV steroid therapy and DXT well without systemic complications. There were also no serious surgical complications post decompression surgery.
DISCUSSION
Intravenous steroid therapy is a first line medical intervention with proven anti-inflammatory benefits in active severe thyroid eye disease10. However, up to 40% of patients may have limited response to steroid treatment4 and some may suffer cumulative side effects of intravenous steroids [11]. External beam radiotherapy is not a new treatment modality for thyroid eye disease [12]. Its efficacy equals that of intravenous steroid therapy according to studies by Prummel and Mourits et al [10, 13] and it works by possibly by targeting lymphocytes and fibrocytes and in so reducing soft tissue inflammation and stabilizing strabismus [5, 13-16]. However, Goman et al reported that the use of radiotherapy is of no benefit in reducing severity in quiescent thyroid eye disease. This may be because the scarring process and inflammation reduction in quiescent states have limited potential for improvement despite radiotherapy [17]. Dolman et al showed that combination therapy with steroids and radiotherapy resulted in a 0% incidence of new onset compressive optic neuropathy while recent studies by others have advocated its use for early progressive thyroid eye disease [6, 18-20]. Some of our patients are those with active established severe thyroid eye disease and who have had previous treatment, failure of treatment response or compressive optic neuropathy on presentation. Patients in the surgical and non-surgical groups received their steroid therapy at 1.8 and 1.7 months respectively upon diagnosis of active disease at our centre followed by radiotherapy (4.4 and 5.3 months). The time between treatments at 2.6 and 1.2 months respectively. One of the indications for surgical decompression was CON and progressive active disease which was non-responsive to combined steroid and DXT therapy (n=4). Other indications (in 2 of our 6 patients) included severe myopathy which required prior decompression to reduce orbital congestion before strabismus surgery. The decompression group had a higher percentage of patients who received more than 1 cycle of intravenous steroid therapy. It also had more patients with risk factors at baseline such as diabetes, hypertension, active smoking status and failed prior treatments. It is therefore not surprising that most of these patients are the ones needed orbital decompression to alleviate compressive optic neuropathy and to control disease progression. Despite multiple cycles of steroid therapy for the above patients and others in this study, therapy was received with no safety complications as we follow a strict safety protocol in our centre. Orbital radiotherapy was also well tolerated. Our study is limited by its retrospective nature and small numbers. This could partly account for inflammatory scores, strabismus and restriction as well as visual acuity in the surgical group failing to reach statistical change post treatment. Other reasons would include the heterogeneous presenting backgrounds of our patient cohort which includes foreign referrals who have received treatment in foreign countries or who have had failed treatment prior to presentation in our clinic. However, this study has shown that combination steroid and radiotherapy helps to reduce inflammation and maintain vision in our cohort of patients. It also supports current literature in the early use of radiotherapy with intravenous steroids for active severe thyroid eye disease.
REFERENCES
- Rootman J and Dolman PJ. (2003). Thyroid Orbitopathy. In: Diseases of the Orbit: A Multidisciplinary Approach. Lippincott Williams & Wilings.
- Hales IB and Rundle FF. (1960). Ocular changes in Graves' disease. A long-term follow-up study. Q J Med. 29: 113-126.
- Dolman PJ and Rath S. (2012). Orbital radiotherapy for thyroid eye disease. Curr Opin Ophthalmol. 23(5): 427-432.
- Juler and FAI. (1913). Acute purulent keratitis in exophthalmic goitre treated by repeated tarsorrhaphy, resection of cervical sympathetic and x-rays; retention of vision in one eye. Trans Ophthalmol Soc UK. 33: 58-62.
- Gorman CA, Garrity JA, Fatourechi V, Bahn RS, et al. (2001). A prospective, randomized, double-blind, placebo-controlled study of orbital radiotherapy for Graves' ophthalmopathy. Ophthalmology. 108(9): 1523-1534.
- Shams PN, Ma R, Pickles T, Rootman J, et al. (2014). Reduced risk of compressive optic neuropathy using orbital radiotherapy in patients with active thyroid eye disease. Am J Ophthalmol. 157(6): 1299-1305.
- Bartley GB. (1994). The epidemiologic characteristics and clinical course of ophthalmopathy associated with autoimmune thyroid disease in Olmsted County, Minnesota. Trans Am Ophthalmol Soc. 92: 477-588.
- Bartley GB. (1995). The differential diagnosis and classifcation of eyelid retraction. Trans Am Ophthalmol Soc. 93: 371-389.
- Dolman PJ and Rootman J. (2006). VISA Classification for Graves orbitopathy. Ophthal Plast Reconstr Surg. 22(5): 319-324.
- Yang M, Wiersinga WM, Soeters MR and Mourits MP. (2014). What is the aim of immunosuppressive treatment in patients with graves' orbitopathy? Ophthal Plast Reconstr Surg. 30(2): 157-161.
- Moli LR, Baldeschi L, Saeed P, Regensburg N, et al. (2007). Determinants of liver damage associated with intravenous methylprednisolone pulse therapy in Graves' ophthalmopathy. Thyroid Off J Am Thyroid Assoc. 17(4): 357- 362.
- Kazim M and Garrity JA. (2012). Orbital radiation therapy for thyroid eye disease. J Neuro-Ophthalmol Off J North Am Neuro-Ophthalmol Soc. 32(2): 172-176.
- Prummel MF, Mourits MP, Blank L, Berghout A, et al. (1993). Randomized double-blind trial of prednisone versus radiotherapy in Graves' ophthalmopathy. Lancet Lond Engl. 342(8877): 949-954.
- Prummel MF, Terwee CB, Gerding MN, Baldeschi L, et al. (2004). A randomized controlled trial of orbital radiotherapy versus sham irradiation in patients with mild Graves' ophthalmopathy. J Clin Endocrinol Metab. 89(1): 15-20.
- Mourits MP, Kempen-Harteveld MLV, Garcia MB, Koppeschaar HP, et al. (2000). Radiotherapy for Graves' orbitopathy: randomised placebo-controlled study. Lancet Lond Engl. 355(9214): 1505-1509.
- Kahaly GJ, Rosler HP, Pitz S, Hommel G, et al. (2000). Lowversus high-dose radiotherapy for Graves' ophthalmopathy: a randomized, single blind trial. J Clin Endocrinol Metab. 85(1): 102-108.
- . Nardi M. (2009). Squint surgery in TED -- hints and fints, or why Graves’ patients are difficult patients. Orbit Amst Neth. 28(4): 245-250.
- . Kim JW, Han SH, Son BJ, Rim TH, et al. (2016). Efficacy of combined orbital radiation and systemic steroids in the management of Graves’ orbitopathy. Graefes Arch Clin Exp Ophthalmol Albrecht Von Graefes Arch Klin Exp Ophthalmol. 254(5): 991-998.
- Marcocci C, Bartalena L, Bogazzi F, Bruno-Bossio G, et al. (1991). Orbital radiotherapy combined with high dose systemic glucocorticoids for Graves’ ophthalmopathy is more effective than radiotherapy alone: results of a prospective randomized study. J Endocrinol Invest. 14(10): 853-860.
- Hahn E, Laperriere N, Millar BA, Oestreicher J, et al. (2014). Orbital radiation therapy for Graves’ ophthalmopathy: measuring clinical efficacy and impact. Pract Radiat Oncol. 4(4): 233-239.