Previous Issues Volume 1, Issue 1 - 2016
MEG Evaluation of the Function of Alpha and Beta Rhythms After Visual Stimulation
Photios Anninos1, Athanasia Kotini1, Adam Adamopoulos1, Triandafillos Gemousakakis1
1Lab of Medical Physics, School of Medicine, Democritus University of Thrace, University Campus, Dragana, Alexandroupoli, 68100,Greece.
Corresponding Author: Photios Anninos, Em. Professor, Lab of Medical Physics, School of Medicine, Democritus University of Thrace, University Campus, Dragana, Alexandroupoli, 68100, Greece, Tel: +30 2531 039000;
E-Mail: [email protected] Received Date: 22 Aug 2016 Accepted Date: 24 Sep 2016 Published Date: 28 Sep 2016 Copyright © 2016 Anninos P Citation: Anninos P, Kotini A, Adamopoulos A and Gemousakakis T. (2016). MEG Evaluation of the Function of Alpha and Beta Rhythms After Visual Stimulation. Mathews J Neurol. 1(1): 004.
ABSTRACT
Objective: In this study, we investigated the function of alpha and beta rhythms after visual stimulation using Magnetoencephalography (MEG) recordings.
Materials And Methods: Ten (10) Caucasian healthy volunteers (6 female, 4 male) aged (mean: 37.6±5.3) participated in the study. The subjects were measured with a 122-channel MEG system in a magnetically shielded room of low magnetic noise. We applied Fast Fourier Transform to all channels and evaluated the alpha and beta rhythm of the volunteers before and after visual stimulation.
Results: Before stimulation, the alpha rhythm was located at the occipital lobe due to mental relaxation and blocked attention. We observed also an activation of the beta rhythm at the frequency range 19-25 Hz due to thought processes. After stimulation, we observed an intense MEG field with a spread of the alpha and beta rhythms throughout the brain without specific location. The decrease in the alpha and beta rhythms is due to the stimulus because each stimulus entering the brain is maintained for a certain period of time representing the short-term memory of the particular stimulus.
Conclusion: The results suggest that the MEG is a efficacious modality in the investigation of alpha and beta rhythms after visual stimulation. This cortical activation might have applicability in modulation of brain status. This might be important to patients' groups because the alpha rhythm could be used as a neurophysiological marker for the activity of the pineal gland.
KEYWORDS
Meg; Visual Stimulation; Alpha Rhythm; Beta Rhythm.
INTRODUCTION
Magnetoencephalography (MEG) is a functional neuroimaging technique for mapping brain activity by recording magnetic fields produced by electrical brain currents, using very sensitive magnetometers and it gives improved spatial resolution with particularly high temporal resolution. Since the MEG signal is a direct measure of neuronal activity, its temporal resolution is comparable with that of intracranial electrodes. MEG complements other brain activity measurement techniques such as electroencephalography (EEG), positron emission tomography (PET), and functional magnetic resonance imaging (FMRI). It is a non-invasive method and uses no ionizing radiation, as opposed to PET. MEG can resolve events with a precision of greater than ten milliseconds (msec), while fMRI, can at best resolve events with a precision of several hundred milliseconds (msec). MEG is also being used to better localize responses in the brain. The responses in the brain before, during, and after the introduction of stimuli can be mapped with greater spatial resolution than was formerly potential used with EEG [1-13].
The pineal gland controls the hormone system and at night it is releasing one important hormone the melatonin in the blood stream and from the blood stream to the brain. Sandyk reported a case of a patient with multiple sclerosis in whom visual perception worsened throughout the course of the day and improved at night [14]. These changes in vision appeared to correspond to the circadian secretion of melatonin which is coupled to the circadian temperature rhythms. Ter Huurne et al [15] investigated whether aberrant modulation of alpha oscillations contributes to attention problems in Attentiondeficit/ hyperactivity disorder (ADHD) patients with the use of MEG. They suggested that aberrant modulations of alpha oscillations reflect attention problems because of ADHD and might be related to the neurophysiological substrate of the disorder. Babiloni et al [16] investigated if simple delayed response tasks affect latency and amplitude of MEG midline alpha rhythms (6-12 Hz) in early dementia. They found that the alpha peak was later in latency in the demented and normal elderly subjects than in the normal young subjects and it was stronger in amplitude in the demented patients than in the normal subjects. Anninos et al [8] in a MEG study discussed the potential essential role of the pineal gland in the long term anticonvulsant effects of external artificial magnetic stimulation because the pineal gland has been shown to be a magnetosensitive organ which forms part of a combined compasssolar clock system and exerts an inhibitory action on seizure activity. Sandyk et al [17] based on MEG measurements suggested that patients with nocturnal epilepsy or those experiencing exacerbation of seizures premenstrually may benefit from the administration of agents which block the secretion or action of melatonin.
The aim of this study was to investigate the function of alpha and beta rhythms after visual stimulation by means of MEG recordings because this cortical activation might have applicability in the modulation of brain status and in clinical use.
MATERIALS AND METHODS
Subjects The MEG recordings were carried out in ten (10) Caucasian healthy volunteers (6 female, 4 male) (mean age: 37.6±5.3 years) in a magnetically shielded room with a whole head 122-channel biomagnetometer (model: Neuromag-122, Helsinki, Finland) (Table 1) [1-13]. All subjects had normal visual acuity and were not on any medication. The first MEG measures represent the baseline without visual stimulation. Afterwards we applied visual stimulation with different information in order to find out how the brain elaborates them (Figure 1). Informed consent was obtained from the participants prior to the procedure. The research was approved by the Research Committee of the Democritus University of Thrace.
Table 1: Volunteers' profile (F:female, M:male).
| A/A | SEX | AGE | WEIGHT | ETHNIC |
|---|---|---|---|---|
| 1 | F | 35 | 71 | Caucasian |
| 2 | F | 43 | 75 | Caucasian |
| 3 | F | 39 | 70 | Caucasian |
| 4 | F | 45 | 65 | Caucasian |
| 5 | F | 39 | 69 | Caucasian |
| 6 | F | 30 | 76 | Caucasian |
| 7 | M | 40 | 85 | Caucasian |
| 8 | M | 38 | 78 | Caucasian |
| 9 | M | 39 | 90 | Caucasian |
| 10 | M | 28 | 79 | Caucasian |
Figure 1: The 122-channel MEG system in the magnetically shielded room. One of the images for the visual stimulation (small icon).
Data Acquisition
The MEG recordings were filtered with cut off frequencies at 0.3 Hz and 40 Hz. The MEG sampling frequency was 256 Hz and the associated Nyquist frequency was 128 Hz. We took 4 MEG recordings of 32 seconds each with 4 stimuli (icons). The images illustrated delicious sweets. As an experimental paradigm one picture is illustrated in Figure 1 (small image). We preferred coloured stimuli instead of neutral ones in order to have more profound effects. The time interval between each stimulus was 1 min. The total experimental time was about 5 min.
Data analysis
The MEG workstation software analyzes the MEG data and gives the isocontour field maps (ISO-fields) in the whole scalp. We used the Fast Fourier Transform (FFT) algorithm to obtain the power spectra of the MEG data. A software program has been developed in our lab in order to detect the 1st dominant frequency of the power spectra of each channel after the application of FFT on the MEG raw data and construct a map for its spatial distribution over the scalp. Different colours in the maps represent different dominant frequencies. The numbers in the map squares represent the 122 MEG channels in every parts of the brain area according to Table 2.
Table 2: This table shows the brain regions and the corresponding channels.
| Brain Regions | Channels |
|---|---|
| Right Temporal | 1-14 , 111-120 |
| Right Parietal | 5-6,11-16,97-100, 109, 110 , 115-122 |
| Left Temporal | 43-50 , 55-62,67-74 |
| Left Parietal | 47-52,59-64,71-74,79,80,87-90 |
| Frontal | 17-42 |
| Occipital | 75-86,91-96 , 101-110 |
| Vertex | 13-16,49-54,61-66,73,74,89,90,99,100 ,117-122 |
RESULTS
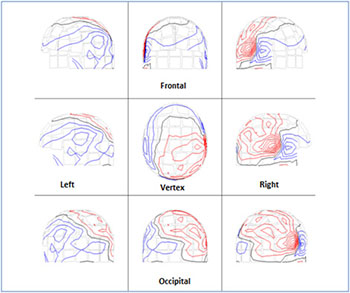
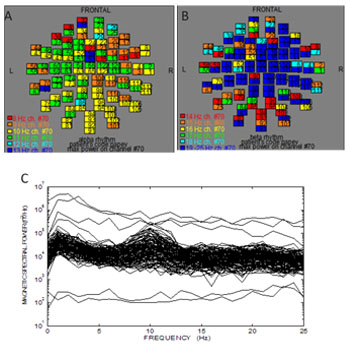
The following results referred to the volunteer (no 8) as representative. (Figure 2) shows the ISO-fields without external visual stimulation. (Figure 3A) exhibits the spatial distribution of the alpha rhythm (8-13 Hz) over the scalp for all the MEG channels without stimulation after the application of the FFT on the MEG raw data. (Figure 3B) shows the beta rhythm (14-25 Hz) before stimulus. We observed an activation of the channels between the frequencies 19-25 Hz due to thought processes. (Figure 3C) shows the overlapping power spectra from these 122 channels after FFT. We viewed a dominant frequency at 10 Hz that indicates the physiological state of the subject. The alpha rhythm is intense due to mental relaxation and blocked of attention. The alpha rhythm was located primarily at the occipital lobe (MEG channels: 75-86, 91-96, 101-110, Table 2).
Figure 2: The scalp isocontour (ISO) field distribution without visual stimulation at the left - right temporal, left - right parietal, frontal , vertex and occipital regions . The red and blue lines indicate the incoming and outgoing magnetic field.
Figure 3: Frequency distribution from all channels without visual stimulation at all brain regions (Table 1). A) The spatial distribution of the alpha rhythm (8-13 Hz). Dominant alpha rhythm at the occipital lobe (MEG channels: 75 -86, 91-96, 101- 110). The numbers in each square represent the 122 MEG channels B) The spatial distribution of the beta rhythm (14-25 Hz). The squares represent the MEG channels C) Power spectra analysis: Overlapping the power spectra from all channels. Dominant frequency at 10 Hz.
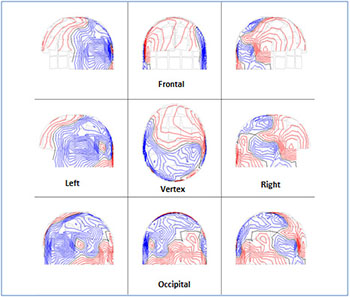
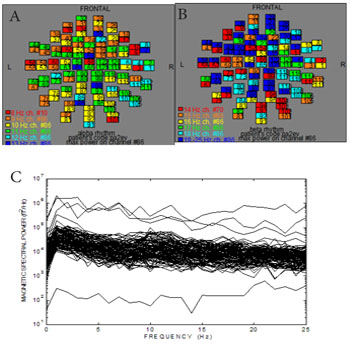
Afterwards we applied external stimuli such as images with different information (Figure 1). Figure 4 shows the ISO-fields with external visual stimulation. We showed an intense activation of the MEG signals all over the scalp. Figure 5A exhibits the spread of the alpha rhythm throughout the brain due to the stimulus without specific location. Figure 5B shows the behavior of the beta rhythm after stimulus. We observed a spread of the beta rhythm without specific location. The activity of the frequencies 19-25 Hz has been decreased. Figure 5C shows the overlapping power spectra from these 122 channels after FFT. We viewed an attenuation of the alpha rhythm due to the attention to the stimulus. Comparing Figures 2,4 and Figures 3,5 we evaluated the alpha and beta activity in the maps before and after external visual stimulation at every region (Table 2).
Figure 4: The scalp isocontour (ISO) field distribution after 352,7 ms visual stimulus at the left -right temporal, left -right parietal, frontal , vertex and occipital regions. We observe an intense activation of the MEG signals. The red and blue lines indicate the incoming and outgoing magnetic field.
Figure 5: Visual stimulation after 352.7 ms. A) The alpha and the B) beta rhythm after optical stimulation at all brain regions (Table 1). The numbers in each square represent the 122 MEG channels C) Power spectra analysis : Overlapping the power spectra from all MEG channels. We observe an attenuation of the magnetic spectra power (fT2 /Hz) of the alpha rhythm as a result of stimulus.
DISCUSSION
From our measurements we extracted useful conclusions regarding the position and the intensity of the MEG recordings after the application of an external visual stimulus. Our raw data showed an intense activation of the MEG signals. This was clearly identified in the ISO-fields between (Figure 2 without stimulus) and (Figure 4 with stimulus). Comparing Figures 3,5 we observed an attenuation of the alpha rhythm at the occipital lobe and the widespread of the beta rhythm due to the visual stimulation.
Recently, the alpha-band activity (10Hz) has drawn a lot of consideration. Lange et al [18] suggested that reduced alphaband power does not always predict improved visual processing, but rather increased excitability. Some researchers found that the power of pre-stimulus alpha-band activity in parietooccipital areas was associated negatively with the individual perception in visual detection and discrimination tasks [19- 21]. Other researchers found that intermediate levels of prestimulus alpha-power in the visual and somatosensory area increases perception and evoked responses while low and high levels have a negative effect [22-24]. Alpha-band power is reconciled by attention and has been associated to the inhibition of task unrelated areas [24-27].
Okazaki et al [28] examined brain oscillatory responses related to visual perceptual change of short-term duration in the absence of morphologic change by MEG. Two types of stimulus conditions were created, the 'face-target' (F-T) condition and the 'saxophone-target' (S-T) condition. They found significantly greater synchronization in the beta (14-30-Hz) frequency band, ranging from 250 to 450ms predominantly over the occipital and parietal regions, after stimulus alternation for the S-T condition than for the F-T condition. Kinsey et al [29] in a research study by MEG concluded that the role of alpha and beta activity in object processing might related principally to changes in visual attention.
In (Figure 5) we illustrated that the alpha and beta rhythms and the power spectra after 352.7 ms at the beginning of visual stimulation. An external stimulus prompts all the cortical neurons to fire simultaneously causing a spike to appear in some 300ms later. The alpha rhythm is classically described as a bilateral posterior rhythm of substantially constant frequency in the range of 8-13Hz and is enhanced by mental relaxation and blocked attention. Since the full expression of alpha rhythm has been shown to occur during puberty, it is possible that the establishment of alpha rhythm is subject to neuroendocrine influences. Nocturnal plasma melatonin levels have been shown to decline progressively throughout childhood reaching a nadir in puberty. This progressive decline in melatonin secretion during childhood facilitates the maturation of the alpha rhythm. Our study population was in adulthood (mean 37.66±5.3 years) because we had volunteers only at these ages.
In a number of studies Anninos et al [1-3, 5, 7, 8, 30-33] using an electronic device demonstrated the significant effects of pi-co-Tesla (pT) (1pT=10-12 Tesla) external transcranial magnetic stimulation (pT-TMS) to patients with CNS disorders. Specifically, using an electronic device invented by them they were able to increase the abnormal (2-7Hz) frequencies of the brain activity towards frequencies of less than or equal to those frequencies of the alpha frequency range (8-13Hz) of each patient [30]. It is known that magnetic fields modify the activity of the pineal gland, which has been shown to control dopaminergic, and endogenous opioid functions [34, 35]. In addition, exposure of an organism or biological material to magnetic fields has been reported to induce mutagenic, immunological, metabolic, endocrine, morphological, developmental, behavioral and anticonvulsant effects [36, 37].
The monitoring of brain activity by MEG requires particularly sensitive sensors made superconductive by liquid helium and data acquisition in shielded rooms cutting out the ambient magnetic fields in order to obtain the best quality signals. The major limitation of the study is the high cost of the MEG system and the liquid helium for its operation.
Our results shown that MEG is a valuable tool for the estimation of the role of alpha and beta rhythms in healthy volunteers. The significant observation is that there is an intense activation of the MEG data and a decrease in the alpha and beta rhythms due to stimulus. This fact encourage the use of MEG for the estimation of the above rhythms in patients with CNS disorders (autism, dyslexia, personality disorders, epilepsy, schizophrenia, Parkinson etc.). This might be important and helpful to patients' groups because the presence of alpha rhythm could be used as a neurophysiological marker for the activity of the pineal gland and for the disorders associated with the absent or delayed maturation of it.
ACKNOWLEDGEMENT
Funding for this work was provided by a collaboration of GGET (General Secretariat of Research and Technology, GREECE) and ERGO AEBE, INC, GREECE under the research program titled "Foundation of a Laboratories Network and purchase of a Multichannel Biomagnetometer SQUID (Superconducting Quantum Interference Device), in order to develop an expert system for automatic acquisition, analysis, evaluation and exploitation of MEG signals that are emitted from different organs of the human body" (Grant Number: 80623).
REFERENCES
- Anninos P, Adamopoulos A, Kotini A and Tsagas N . (2000). Nonlinear Analysis of Brain Activity in Magnetic Influenced Parkinson Patients. Brain Topogr. 13(2), 135-144.
- Anninos P, Adamopoulos A, Kotini A, Tsagas N, et al. (2007). MEG evaluation of Parkinson's diseased patients after external magnetic stimulation. Acta Neurol Belg. 107(1), 5-10.
- Anninos P, Kotini A, Adamopoulos A and Tsagas N. (2003). ?agnetic stimulation can modulate seizures in epileptic patients. Brain Topogr. 16(1), 57-64.
- Anninos P, Kotini A, Adamopoulos A, Hatzimihael A, et al. (2006). Identification of taste quality with the use of MEG. J Integr Neurosci. 5(4), 535-540.
- Anninos P, Kotini A, Anninou N, Adamopoulos A, et al. (2008). MEG recordings of patients with CNS disorders before and after external magnetic stimulation. J Integr Neurosci 7(1), 17-27.
- Anninos P, Kotini A, Dipla V, Tsalkidis A, et al. (2010). MEG evaluation of Febrile Seizures in Young Children. J Child Neurol. 25(1), 61-66.
- Anninos P, Tsagas N, Jacobson J and Kotini A. (1999). The biological effects of magnetic stimulation in epileptic patients. Panminerva Med. 41(3), 207-215.
- Anninos PA, Tsagas N, Sandyk R and Derpapas K. (1991). Magnetic stimulation in the treatment of partial seizures. Int J Neurosci. 60(3-4), 141-171.
- Gemousakakis T, Anninos P, Zissimopoulos A, Seimenis I, et al. (2013). A study on the age dependency of gustatory states: Low-frequency spectral component in the resting-state MEG. J Integr Neurosci. 12(4), 1-13.
- Gemousakakis T, Kotini A, Anninos P, Zissimopoulos A, et al. (2011). MEG evaluation of taste by gender difference. J Integr Neurosci. 10(4), 537-545.
- Kotini A, Anninos P and Tamiolakis D. (2007). MEG mapping in multiple sclerosis patients. Eura Medicophys. 43(3), 345-348.
- Kotini A and Anninos P. (2002). Detection of non-linearity in schizophrenic patients using MEG. Brain Topogr. 15(2), 107- 113.
- Kotini A, Anninos P, Adamopoulos A and Prassopoulos P. (2005). Low Frequency MEG Activity and MRI Evaluation in Parkinson's Disease. Brain Topogr. 18(1), 59-63.
- Sandyk R. (1995). Diurnal variations in vision and relations to circadian melatonin secretion in multiple sclerosis. Int J Neurosci. 83(1-2), 1-6.
- Ter Huurne N, Onnink M, Kan C, Franke B, et al. (2013). Behavioral consequences of aberrant alpha lateralization in attention-deficit/hyperactivity disorder. Biol Psychiatry. 74(3), 227-233.
- Babiloni C, Cassetta E, Chiovenda P and Del Percio C. (2005). Alpha rhythms in mild dements during visual delayed choice reaction time tasks: a MEG study. Brain Res Bull. 65(6), 457-470.
- Sandyk R, Tsagas N and Anninos PA. (1992). Melatonin as a proconvulsive hormone in humans. Int J Neurosci. 63(1-2), 125-135.
- Lange J, Halacz J, van Dijk H, Kahlbrock N, et al. (2012). Fluctuations of prestimulus oscillatory power predict subjective perception of tactile simultaneity. Cereb Cortex. 22(11), 2564-2574.
- Romei V, Gross J and Thut G. (2010). On the role of prestimulus alpha rhythms over occipito-parietal areas in visual input regulation: correlation or causation? J Neurosci. 30(25), 8692-8697.
- Van Dijk H, Schoffelen JM, Oostenveld R and Jensen O. (2008). Prestimulus oscillatory activity in the alpha band predicts visual discrimination ability. J Neurosci. 28(8), 1816- 1823.
- Wyart V and Tallon-Baudry C. (2009). How ongoing fluctuations in human visual cortex predict perceptual awareness: baseline shift versus decision bias. J Neurosci. 29(27), 8715- 8725.
- Zhang Y and Ding M. (2010). Detection of a weak somatosensory stimulus: role of the prestimulus mu rhythm and its top-down modulation. J Cogn Neurosci. 22(2), 307-322.
- Lange J, Oostenveld R and Fries P. (2013). Reduced occipital alpha power indexes enhanced excitability rather than improved visual perception. J Neurosci. 33(7), 3212-3220.
- Rajagovindan R and Ding M. (2011). From prestimulus alpha oscillation to visual-evoked response: an inverted-U function and its attentional modulation. J Cogn Neurosci. 23(6), 1379 -1394.
- Haegens S, Handel BF and Jensen O. (2011). Top-down controlled alpha band activity in somatosensory areas determines behavioral performance in a discrimination task. J Neurosci. 31(14), 5197-5204.
- Van Ede F, de Lange F, Jensen O and Maris E. (2011). Orienting attention to an upcoming tactile event involves a spatially and temporally specific modulation of sensorimotor alpha and beta-band oscillations. Neurosci. 31(6), 2016-2024.
- Jensen O and Mazaheri A. (2010). Shaping functional architecture by oscillatory alpha activity: gating by inhibition. Front Hum Neurosci. 4,186.
- Okazaki M, Kaneko Y, Yumoto M and Ariman K. (2008). Perceptual change in response to a bistable picture increases neuromagnetic beta-band activities. Neurosci Res. 61(3), 319- 328.
- Kinsey K, Anderson SJ, Hadjipapas A and Holliday IE. (2011). The role of oscillatory brain activity in object processing and figure-ground segmentation in human vision. Int J Psychophysiol. 79(3), 392-400.
- Anninos PA and Tsagas N. (1995). Electronic apparatus for treating epileptic individuals US patent 5453072.
- Anninos P, Adamopoulos A and Kotini A. (2015). MEG as a Medical Diagnostic Tool in the Greek Population. Acta Medica (Hradec Kralove). 58(3), 71-78.
- Anninos P, Adamopoulos A, Kotini A and Tsagas N. (2016). MEG evaluation of pico-Tesla external TMS on multiple sclerosis patients. Mult Scler Relat Disord. 8, 45-53.
- Anninos P, Adamopoulos A, Kotini A and Tsagas N. (2016). Combined MEG and pT-TMS study in Parkinson's disease. J Integr Neurosci. 15(2), 145-162.
- Aldegunde M, Miquez I and Veira J. (1985). Effects of pinealectomy on regional brain serotonin metabolism. Int J Neurosci. 26(1-2), 9-13.
- Lissoni P, Esposti D, Esposti G, Mauri R, et al. (1986). A clinical study on the relationship between the pineal gland and the opioid system. J Neural Trans. 65(1), 63-73.
- Jankovic BD, Maric D, Ranin J and Veljic J. (1991). Magnetic fields, brain and immunity: effect on humoral and cell-mediated immune responses. Int J Neurosci. 59(1-3), 25-43.
- Malinin GI, Gregory WD, Morelli L, Sharma VK, et al. (1976). Evidence of morphological and physiological transformation of mammalian cells by strong magnetic fields. Science. 194(4267), 844-846.