Previous Issues Volume 1, Issue 2 - 2016
Association Between Cumulative Time of Use of Antiretroviral Drugs and Vitamin D Deficiency in Brazilian Hiv-Infected Patients
Guilherme Almeida Rosa da Silva1, Filipe Anibal Carvalho Costa2, Walter de Araujo Eyer-Silva1, Jorge Francisco da Cunha Pinto1, Fernando Raphael de Almeida Ferry1, Marcelo Costa Velho Mendes de Azevedo1, Rogerio Neves Motta1
1Professor of Internal Medicine from the Federal University of the State of Rio de Janeiro, UNIRIO, Gaffrée Guinle University Hospital.
2Public Health Researcher from Oswaldo Cruz Foundation - FIOCRUZ
Corresponding Author: Guilherme Almeida Rosa da Silva, Professor of Internal Medicine from the Federal University of the State of Rio de Janeiro - UNIRIO - Gaffrée Guinle University Hospital, Brazil, Tel: +55 21 2264-5844; E-Mail: [email protected]
Received Date: 04 Oct 2016 Accepted Date: 25 Oct 2016 Published Date: 27 Oct 2016
Copyright © 2016 Silva GARD
Citation: Silva GARD, Costa FAC, Silva WDAE, Pinto JFDC, et al. (2016). Association Between Cumulative Time of Use of Antiretroviral Drugs and Vitamin D Deficiency in Brazilian Hiv-Infected Patients. Mathews J HIV AIDS. 1(2): 010.
ABSTRACT
Objective: The objective of this study is to estimate the prevalence of vitamin D deficiency among HIV-infected outpatients treated at a university hospital in Brazil. Look for associations between vitamin D deficiency and factors such as age, sex, time since diagnosis, cumulative time of use of different antiretroviral count, CD4 + lymphocytes, and plasma viral load of HIV. Design and Methods: We analyzed 125 patients aged above 18 years, of both sexes, who had recorded measurements of 25(OH)D, CD4+ lymphocyte count, and viral load count, all obtained in the same sample. Were excluded from the study any who had taken any vitamin supplement in the last year, carriers of chronic renal failure with creatinine clearance less than 30 ml/min, or liver failure. Linear regression was used to analyze continuous variables or Student`s t-test for categorical variables. The deficient patients were whose 25(OH)D concentrations were below 32 ng/dL. Results: A total of 72.8% of patients had vitamin D deficiency. It was found no association between vitamin D and the variables: age, gender, time since diagnosis, duration of use of antiretroviral therapy, HIV viral load, and with none current antiretroviral regimen. A trend for the association was found between the plasma concentration of vitamin D and CD4+ lymphocyte count (negative β, R2 = 0.029, p = 0.058). There was a statistically significant association between the cumulative usage time of efavirenz and plasma concentrations of vitamin D (negative β, R² = 0.071 and p = 0.03).
INTRODUCTION
The vitamin D (VD) deficiency, measured by serum 25(OH)D, is an underdiagnosed condition and generally not evaluated by clinicians, especially those located in countries abundant in sunlight. Part of this omission is due to the ignorance of the consequences of subclinical deficiency of this nutrient and the inability of professionals to recognize clinically manifest forms of VD scarcity [1]. It is estimated that around 36% of healthy adults and 57% of hospitalized patients in the United States (U.S.) have some degree of VD deficiency [2]. This condition is searched at the highest rate in residents of countries with less sun exposure, elderly, malnourished, and in patients with acquired immunodeficiency syndrome (AIDS) [3,4]. The VD is considered a steroid hormone since the ’60s and has its principal function attributed to the metabolism of calcium and phosphate. This control is accomplished through a homeostatic balance of the parathyroid gland, bone, kidney, and intestine [5]. Besides classic rickets in children and osteopenia in different degrees in adults, chronic and subclinical VD deficiency has been implicated as a risk factor in pathogenic processes such as metabolic syndrome, cancer, and autoimmune diseases [6- The research interests of the various metabolic interactions caused by VD deficiency in person with AIDS are: cellular immunity dysfunction, predisposition to opportunistic infections, increased risk of Immune Reconstitution Inflammatory Syndrome (IRIS), interactions with antiretroviral treatment, increase in the prevalence of anemia, increased prevalence of osteoporosis, and increased overall mortality due to cardiovascular complications of metabolic syndrome [10-15].
Relevant Aspects of Vitamin D in AIDS A study conducted in the Netherlands, among women with average age of 36.5 years, and infected with the human immunodeficiency virus (HIV), showed a prevalence of VD deficiency of 58.1% (reference values of 10 and 14 ng/mL for winter and summer respectively) [16]. In a similar study among adults and adolescents in the U.S., the prevalence reached 87% (reference of 15ng/mL) [17]. In Italy, Spain, and again in the U.S., the prevalence of VD deficiency found among HIV positive adults were respectively 81.25%, 86%, and 74.4% with cutoffs of 10 ng/dL, 18 ng/mL, and 32 ng/dL [4,18,19]. In a survey, conducted in 200 HIV patients in the South Central U.S. (Houston, Texas), was found a prevalence of 64% of VD deficiency (< 20ng/dL) and 20.5% of the individuals had a severe deficiency (< 10 ng/dL). The African-American ethnicity, higher BMI, and smoking have been implicated as risk factors for VD deficiency [20]. Likewise, a very similar study conducted in New York, with 274 patients, found deficiency levels (< 10 ng/ mL) in 21.2% of the cases, insufficient levels (10-30 ng / mL) in 68.6%, and sufficiency (> 30 ng/mL) in 10.2%. The groups showed a logarithmic viral load inversely proportional to the levels of VD. The black people were the most affected by the deficiency [21]. In a study, conducted in HIV-infected women in Tanzania, decreased 25(OH)D (< 32ng/mL) was associated with unfavorable progression to stage III or IV of the World Health Organization (WHO)/HIV and severe anemia. Women with normal results of 25(OH)D, located in the highest quintile, had lower overall mortality compared to those followed in the lowest quintile. Nevertheless, this data was independent of CD4+ lymphocytes counts of these women [14]. When performing a nested case-control study to evaluate the prognostic value of 25(OH)D in patients with AIDS, the risk of death fell 46% if has been occurred a twofold increase in plasma 25(OH)D after one year of monitoring [22]. A Norwegian longitudinal study of 53 patients found that those with less than 25 ng/mL of 25(OH)D levels had lower survival than patients with normal levels when adjusted by CD4+ lymphocytes. The strength of association was greater in patients with CD4+ lymphocytes count below to 50 cells/mL [23]. The EuroSIDA study demonstrated that among 1985 HIV patients studied, 23.7% had VD levels below 10ng/mL, 65.3% had levels between 10 and 30 ng/ml, and only 11% had levels that can be considered normal (above 30 ng/ml). Individuals who performed levels of VD at the lowest tertile had a higher mortality during follow-up and higher clinical AIDS-related complications [24]. A French study of 2994 patients reported the prevalence of VD insufficiency/deficiency in 86.7% of patients. Of these, 55.6% had mild impairment (10-30ng/dL) and 31.1% had severe deficiency (<10ng/dL). Variables as smoking, use of antiretrovirals, and levels of CD4+ lymphocytes <350 cells/mm³ were reported as risk factors. The efavirenz (EFV) was the only drug involved with substantially increased risk OR = 1.89 (95%CI: 1.45 to 2.47) [25]. The relationship between VD deficiency and viral load was evaluated in a few studies. One of the most important studies was conducted by the University of Wisconsin in 112 volunteers. They found an association between VD deficiency and the viral load in a U-shape curve, with higher VD among the highest viral loads or in subjects with undetectable viral load [26]. In an experimental model, the presence of lipopolysaccharide (LPS) and the HIV protein gp120 increased macrophage CYP24A1 (LPS only) and CYP27B1 (both) activity. The CYP27B1 increases the conversion of 25(OH)D to 1,25(OH)2D3 and CYP24A1 accelerate the catabolism of 1,25(OH)2D3, providing a possible immune regulation by macrophages in VD concentration in response to high viremia or systemic inflammation [27]. Regarding the use of antiretrovirals, a sectional analysis of 1077 patients with AIDS showed a higher rate of severe VD deficiency and higher bone turnover measured by FA among users of EFV. The use of tenofovir (TDF) was associated with increased bone turnover, without, however, be related to VD deficiency [12]. The use of EFV in the current schema and CD4+ lymphocytes count <200 cells/mm³ were associated with vitamin D deficiency in a Belgian cross-sectional study [28]. When performed a prospective analysis comparing the use of TDF/emtricitabine plus rilpivirine or EFV was found a significant reduction in the 25(OH)D plasma concentration after 48 weeks in EFV, but not in rilpivirine group [29]. In Thailand, a prospective analysis of patients coinfected with HIV/ HBV found a statistically significant reduction in the concentration of 25(OH)D after TDF use [30]. The EFV suppresses CYP2R1, which may reduce the 25-hydroxylase activity, that should limit the formation of 25(OH) D. The induction of CYP3A4 may increase the production of 25(OH)D, however, the role of the CYP3A4 as a 25-hydroxylase has been seen as secondary [31]. The EuroSIDA study demonstrated greater association regarding the use of protease inhibitors (PI) and VD deficiency, as well as in another study they found the same association in ritonavir users [24,32]. Regarding the transmission of HIV in pregnant women with VD deficiency (< 32 ng/mL), a study of 884 HIV positives pregnant women in Tanzania, showed vertical transmission rate at 50% higher in the deficient group, after six weeks of childbirth, and doubled the transmission in initially uninfected children, after six weeks of life when breastfed. The overall risk was 46% higher. Mortality among children generated by VD deficient mothers was 61% higher during follow-up [33]. A study among 460 intravenous drug users exposed to HIV assessed the rate of transmission in relation to vitamin D receptors (VDR) haplotypes. It was observed that the polymorphisms rs11568820, rs4516035, rs10735810, rs1544410, and rs17878969 reduced in 60% the risk of HIV transmission [34]. The same author in another study concluded that genotype rs1544410_AA is associated with slower progression to AIDS and reduce the rate of contamination with HIV, by promoting greater response to 1,25(OH)2D3 stimuli [35]. Another study in Tanzania observed the relation between body mass index (BMI) and the risk of infection among pregnant women with VD deficiency (< 32 ng/mL). The result found was that deficient women had a 43% higher risk of developing cachexia, with a BMI < 18, in two years. Regarding the infections, the deficient women had a 27% greater risk of developing respiratory tract infections and 174% higher in presenting candidiasis [36]. An American study showed an association between VD deficiency and bacterial vaginosis among pregnant women not infected with HIV [37]. In Uganda, 38% of patients with TB and HIV coinfection had VD deficiency (< 20ng/mL), versus 22% of individuals with tuberculosis without HIV, and 20% of healthy individuals [38]. About osteomineral metabolism, HIV positive patient has a higher risk of developing osteoporosis and osteopenia. The prevalence of osteoporosis among HIV positive individuals reached 27.7% versus 17.7% for the control group studied in the American health system [39]. In another study comprising 492 HIV-positive patients, osteopenia prevalence was 54.6% in men and 51.1% in women. The prevalence of osteoporosis was 33.7 and 8.3% respectively. A compilation of studies points to several risk factors, which include: low BMI; advanced age; homosexual transmission; high viral load, low CD4+ lymphocytes count, TDF or IP use, smoking, hypogonadism, and alcohol consumption [40,41]. The use of bisphosphonates, such as oral alendronate 70mg/ once a week, associated with supplementation of 1.5 g of calcium per day and 400-800 IU of 1,25(OH)2D3 can be considered safe and effective for patients with HIV, as well as in the general population [42]. Studies analyzing directly VD deficiency generating bone disease among HIV individuals are scarce, despite numerous studies evaluating the high prevalence of bone disorders and risk factors involved. The dose of VD to be supplemented and the optimum levels of 25(OH)D remain uncertain [43].
METHODS
We revised 400 records from the Immunology outpatient clinic from the Gaffrée Guinle University Hospital - the Federal University of the State of Rio de Janeiro, attended from October 2013 through November 2013. Patients who were older than 18 years, of both sexes, who had recorded measurements of 25(OH)D, CD4+ lymphocyte count, and HIV viral load performed in the same sample were included in the study. In this sample, none of the patients had recorded use of any type of vitamin supplement within the last year, chronic renal failure (creatinine clearance inferior to 30 ml/min) or liver failure, which screening is performed by history, physical examination, and biochemical routine. In total 125 patients who met the inclusion criteria were selected for the study. As the request of plasma 25(OH)D has become a routine test for monitoring HIV-infected patients in this service only in the second semester from 2013, 275 patients were excluded because of the lack of 25(OH)D in the chart. The following variables were analyzed: sex, age (years), time since diagnosis of HIV infection (months), the historical use of antiretroviral drugs, cumulative time of use of different antiretroviral drugs (months) until the date of the collection of the samples, plasma HIV viral load, CD4+ lymphocytes, and 25(OH)D plasma concentration. The count of CD4+ lymphocytes was categorized into four groups: 1-100 cells /mm³, 101- 350 cells/mm³, 351-500 cells/mm³, and > 500 cells/mm³. The value of plasma HIV viral load was expressed in logarithms. The plasma concentration of 25(OH)D was also categorized into four groups: 32 ng/dL. Patients whose VD concentrations were inferior to 32 ng/dL were considered deficient. The database was constructed in SPSS version 17.0 software (SPSS Inc., Chicago, IL) We performed the study in the series of cases of the association between the concentration of VD, considered response variable, and the explanatory variables: sex, age, time since diagnosis, duration of antiretroviral treatment, accumulated time of use of different antiretroviral drugs. The correlation between the concentration of VD and continuous variables was performed by simple linear regression, yielding the angular coefficient (β), determination coefficient (R²) and p-values. The mean VD in different categories was compared using the Student`s t-test. The level of statistical significance in all analyzes was set at 5%. The study was approved by the local ethical committee
RESULTS
We obtained the descriptive characteristics of the sample+ (125 patients). Table 1 shows the data on gender, age, use of antiretrovirals, CD4+ lymphocytes, status of viral load, HIV viral load, and VD concentration.
Table 1: Description of Sample Variables
| Variable | N = 125 | % |
|---|---|---|
| Sex | ||
| Male | 72 | 57,6 |
| Female | 53 | 42,4 |
| Age | ||
| 18-30 years | 13 | 10,4 |
| 31-45 years | 55 | 44,0 |
| 45-60 years | 49 | 39,2 |
| >60 years | 8 | 6,4 |
| HAART | ||
| Yes | 104 | 83,2 |
| No | 21 | 16,8 |
| CD4+ | ||
| < 100 cells/mm³ | 5 | 4,0 |
| 100-350 cells/mm³ | 33 | 26,4 |
| 351- 500 cells/mm³ | 33 | 26,4 |
| > 500 cells/mm³ | 54 | 43,2 |
| Viral load | ||
| Detectable | 38 | 30,4 |
| Non-detectable | 87 | 69,6 |
| Viral load log | ||
| < 2 | 88 | 70,4 |
| 2-3 | 11 | 8,8 |
| 3,1-4 | 14 | 11,2 |
| 4,1-5 | 10 | 8,0 |
| 5,1-6 | 2 | 1,6 |
| VD measure | ||
| < 10 ng/dL | 5 | 4,0 |
| 10-19 ng/dL | 36 | 28,8 |
| 20-31 ng/dL | 50 | 40,0 |
| >32 ng/dL | 34 | 27,2 |
Variables: age, sex, duration of antiretroviral, and time since diagnosis were not statistically significant associated with plasma concentrations of VD in the group studied. The correlation between the age variable and serum VD produced an R2 = 0.001, p = 0.795. The mean plasma concentration of VD were not statistically significant different between males (26.69 ± 12.72 ng / dL) and females (26.36 ± 11.68 ng / dL) with p = 0.879. A relationship between duration of use of antiretrovirals and VD concentration was not significant either, (R2 = 0.008, p = 0.316). The time since HIV infection diagnosis were not correlated to plasma concentrations of VD analyzed by simple linear regression (R² = 0.009 and p = 0.281).
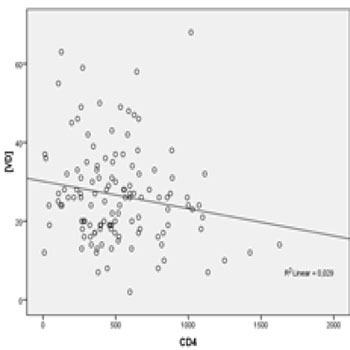
The degree of immunosuppression of the patients studied was evaluated by CD4+ lymphocytes count. Table 1 shows the distribution of patients by the level of CD4+ lymphocytes count. It was observed with the linear regression, correlation close to statistical significance between the CD4+ lymphocytes count and concentrations of VD (negative β, R2 = 0.029, p = 0.058). The linear regression between the concentration of VD and CD4+ lymphocytes count is shown in Figure 1.
Figure 1: Linear regression between plasmatic VD concentration (ng/dL) and plasmatic TCD4+ cells count (cells/mm³).
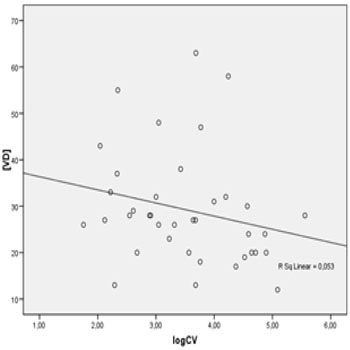
The mean concentration of VD in CD4+ lymphocytes categories were : CD4 < 100 cells/mm³ (25.6 ± 10.83 ng/dl) , 100-350 cells/mm³ (29.63 ± 13.31 ng/dl) , 351-500 cells/mm ³ (24.75 ± 10.43 ng/dl), and > 500 cells/mm³ (26.13 ± 12.85 ng / dl) .. We also evaluated the correlation between HIV viral load and concentrations of VD. To do so, we compared the mean concentration of VD among patients with detectable viral load (n = 38 patients), considering the detection limit of 40 viral copies/mm3, and undetectable viral load (n = 87 patients). The mean VD concentrations of these groups were 29.21 ± 12.18 ng/mL (detectable) and 25.39 ± 12.12 ng/mL (undetectable), p = 0.109. Among patients with detectable viral load, linear regression analysis was used to identify the relationship between the logarithmic viral load and VD concentrations. We observed a not statistically significant correlation (negative β, R2 = 0.053, p = 0.165), as shown in Figure 2.
Figure 2: Linear regression between plasmatic VD concentration (ng/dL) and HIV viral load Log.
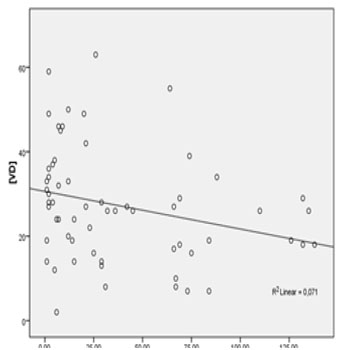
Regarding the history of antiretrovirals there was no significant difference between the mean VD concentrations among the group that used antiretroviral therapy (n = 104, mean VD = 25.91 ± 12.22 ng/dL) in comparison to the naive group (n = 21, mean VD = 29.71 ± 12.14 ng/dL), p = 0.202. The correlation between VD concentration and the time of cumulative use of different antiretrovirals (months) was evaluated. Table 2 shows the results of these analyzes. The time of cumulative use of the drugs: zidovudine (AZT), lamivudine (3TC), TDF, ritonavir (r), lopinavir (LPV), atazanavir (ATV), and nevirapine (NVP) were not significantly correlated with the VD concentration through linear regression. The duration of cumulative use of EFV was statistically significantly correlated with the VD concentration (negative β, R² = 0.071 and p = 0.03), as shown in Figure 3.
Figure 3: Linear regression between plasmatic VD concentration (ng/dL) and cumulative time of use of efavirenz (months).
Table 2: Correlation between VD plasmatic measure (ng/dL) and cumulative time of diferente antiretrovirals (months).
| Antiretroviral | β | R² | p |
|---|---|---|---|
| AZT | -0,034 | 0,023 | 0,148 |
| 3TC | -0,015 | 0,006 | 0,441 |
| TDF | -0,033 | 0,007 | 0,557 |
| R | 0,013 | 0,002 | 0,723 |
| LPV | 0,002 | 0,000 | 0,978 |
| ATV | 0,049 | 0,033 | 0,260 |
| NVP | -0,019 | 0,004 | 0,718 |
| EFV* | -0,089 | 0,071 | 0,037* |
It was not possible to assess the influence of the current use of antiretroviral drugs on VD concentrations, in view of the great variability of schemes compared to the sample number. The different types of current antiretroviral drugs and the mean concentration of VD can be seen in Table 3.
Table 3: Different HAART Distribution and VD Plasmatic Measures (ng/ dL).
| HAART | VD mean | N | Standard deviation |
| 3TC + DDI + ATV | 31,00 | 1 | - |
| 3TC + TDF + ATV | 36,50 | 2 | 13,435 |
| 3TC + TDF + ATV/r | 28,77 | 22 | 8,513 |
| 3TC + TDF + DRV/r + RAL | 35,67 | 3 | 28,006 |
| 3TC + TDF + EFV | 26,75 | 8 | 17,702 |
| 3TC + TDF + FPV/r | 24,50 | 2 | 6,364 |
| 3TC + TDF + LPV/r | 23,50 | 8 | 10,850 |
| 3TC + TDF + NVP | 17,33 | 3 | 7,024 |
| AZT + 3TC + ATV | 16,00 | 2 | 1,414 |
| AZT + 3TC + ATV/r | 19,33 | 3 | 6,028 |
| AZT + 3TC + EFV | 26,31 | 26 | 13,120 |
| AZT + 3TC + LPV/r | 21,60 | 5 | 7,893 |
| AZT + 3TC + NVP | 22,70 | 10 | 15,210 |
| AZT + 3TC + TDF + ATV/r | 26,00 | 2 | 8,485 |
| AZT + 3TC + TDF + LPV/r | 28,83 | 6 | 9,745 |
| AZT + DDI + ATV/r | 14,00 | 1 | - |
| No HAART | 29,71 | 21 | 12,141 |
| Total | 26,55 | 125 | 12,246 |
DISCUSSION
Spanish, Dutch, Americans, Norwegians, and Italians studies, like the great EuroSIDA study, indicate a prevalence of VD deficiency among people with HIV around 60-80% [4,16,23]. The present study is a cross-sectional analysis in which limitations occur to the determination of a causal relationship, because the absence of temporality. The cross-sectional studies are predisposed to prevalence bias. This study allows only the description of associations between variables. The prevalence of VD deficiency in the studied sample (< 32ng/dL) was 72.8%. This value is consistent with works done in other countries, including temperate climates, in contrast to the tropical climate of the State of Rio de Janeiro. This element suggests a strengthening of the argument that the presence of a tropical climate does not act robustly protecting the population of VD deficiency. The variables age, sex, duration of antiretrovirals, and time since diagnosis showed no statistically significant association with VD concentrations. There is no robust scientific evidence and biological plausibility that makes even the age and sex factors strongly associated with VD deficiency. More specifically on the time since diagnosis, we must take into consideration that the diagnosis can be performed at different stages of the disease, not meaning the precise time of infection. In addition, there is variability in terms of speed of progression to severe immunosuppression among individuals that make the time since diagnosis a variable full of confounding factors. The result of association analysis between VD concentrations and total time of antiretrovirals may be affected by the fact that most schemes are formed by 3 or 4 different medications, and are not necessarily held to the same drugs and schemes throughout the all history of antiretrovirals. Treatment dropouts, poor compliance, and exhibitions for different frequencies with different schemes make this analysis predisposed to confounding variable. Likewise, the analysis of the association between VD concentration and CD4+ lymphocyte count, that express the degree of immunosuppression, may be affected by the fact that antiretroviral is started when the CD4+ lymphocyte count is low, currently in Brazil < 500 cells/mm³ and previously < 350 cells/ mm³. In patients with high CD4+ lymphocyte count were included treatment-naive and chronic users of antiretroviral drugs with good adherence, that could have different antiretroviral cumulative time and could be experimented with different schemes. Those with more severe immunosuppression mainly concentrate individuals with newly diagnosis, recent begin of antiretroviral, individuals previously diagnosed with clinical and virologic failure, or poor adherence to treatment. These factors make the complex assessment of the association between VD deficiency and CD4+ lymphocyte count. A not statistically significant correlation between the CD4+ lymphocyte count and VD concentration was found, with negative β. This finding may reflect a prolonged and effective use of antiretroviral drugs, especially with schemes involving the EFV, among patients with higher CD4+ lymphocyte count, which would lead to a reduction in plasma concentration of VD.
Another endpoint would be the analysis between detectable or undetectable viral load groups and VD deficiency. This analysis may be affected by the fact that the undetectable group patients mostly were using antiretroviral and have different time of use of different drugs. The detectable group is composed of naive and patients on antiretroviral therapy with low compliance or failure. When analyzed by linear regression, the logarithmic viral load of the detectable group and VD concentration, we noted a not statistically significant correlation, with negative β. Possibly the viral fitness aggravates an inflammatory condition of the body that can negatively influence the plasma concentration of VD. In an experimental model, the presence of the gp120 protein of HIV increased macrophage expression of CYP27B1, which theoretically could reduce the plasma concentration of 25(OH)D [ 26 ]. We cannot rule out the influence of previous schemes, mainly involving the EFV among patients with high viral load due to dropouts or virologic failure. There was no statistically significant difference between the mean concentration of VD when comparing patients experienced with antiretrovirals and naïve It may be a consequence of the fact that users of antiretrovirals use a broad range of schemes and medicines, current and previous, which could act in different ways on concentrations of VD. A statistically significant correlation of cumulative time of use of EFV and VD concentration is a result that reinforces the theory that the EFV is the antiretroviral drug most associated with VD deficiency. This study suggests that the influence of EFV reducing the VD concentrations possibly can be time dependent. Studies that take into consideration the cumulative use of antiretroviral therapy and VD deficiency are scarce. According to current literature, the EFV suppresses CYP2R1, which has been suggested as a reason for reducing the concentration of 25(OH)D. Moreover EFV induce CYP3A4 which could compensate for the suppression of CYP2R1 increasing the concentration of 25 (OH)D. Currently, the role of CYP3A4 as a 25-hydroxylase activity has been seen as secondary which could explain the predominance in lowering effect of 25(OH)D concentrations caused by suppression of CYP2R1 by EFV. Clinical Impacts of the influence of EFV in concentration of 25(OH) D are undetermined.
REFERENCES
- Unger MD, Cuppari L, Titan SM, Magalhaes MCT, et al. (2010). Vitamin D status in a sunny country: Where has the sun gone? Clinical Nutrition. 29(6), 784-788.
- Holick MF. (2006). High prevalence of vitamin D inadequacy and implications for health. Mayo Clin Proc. 81(3), 353-373.
- Holick M, Matsuoka LY and Wortsman J. (1989). Age, vitamin D, and solar ultraviolet radiation. Lancet. 2(8671), 1104- 1105.
- Rodriguez M, Daniel SB, Gunawardene S and Robbins GK. (2009). High frequency of vitamin D deficiency in ambulatory HIV-positive patients. AIDS Res Hum Retroviruses. 25(1), 9-14.
- Premaor MO and Furlanetto TW. (2006). Hipovitaminose D em adultos: entendendo melhor a apresentacao de uma velha doenca. Arq Bras Endocrinol Metab. 50(1), 25-37.
- Cantorna MT. (2006). Vitamin D and its role in immunology: multiple sclerosis and inflammatory bowel disease. Prog Biophys Mol Biol. 92(1), 60-4.
- Mathieu C and Badenhoop K. (2005). Vitamin D and type 1 diabetes mellitus: state of the art. Trends Endocrinol Metab. 16(6), 261-266.
- Pittas AG, Lau J, Hu FB and Dawson-Hughes B. (2007). The role of vitamin D and calcium in type 2 diabetes. A systematic review and meta-analysis. J Clin Endocrinol Metab. 92(6), 2017-2029.
- Holick MF. (2006). Resurrection of vitamin D deficiency and rickets. J Clin Invest. 116(8), 2062-2072.
- Liu PT, Stenger S, Tang DH and Modlin RL. (2007). Cutting edge: vitamin D mediated human antimicrobial activity against Mycobacterium tuberculosis is dependent on the induction of cathelicidin. J Immunol. 179(4), 2060-2063.
- Conesa-Botella A, Mathieu C, Colebunders R, MorenoReyes R, et al. (2009). Is vitamin D deficiency involved in the immune reconstitution inflammatory syndrome? AIDS Rer Ther. 6, 4.
- Welz T, Childs K, Ibrahim F, Poulton M, et al. (2010). Efavirenz is associated with severe vitamin D deficiency and increased alkaline phosphatase. AIDS. 24(12), 1923-1928.
- Szep Z, Guaraldi G, Shah SS, Ratcliffe SJ, et al. (2011). Vitamin D deficiency is associated with type 2 diabetes mellitus in HIV infection. AIDS. 25(4), 525-529.
- Mehta S, Giovannucci E, Mugusi FM, Spiegelman D, et al. (2010). Vitamin D Status of HIV-Infected Women and Its Association with HIV Disease Progression, Anemia, and Mortality. PLoS ONE. 5(1), e8770.
- Conrado T, Miranda-Filho DB and Bandeira F. (2010). Vitamin D deficiency in HIV-infected individuals: one more risk factor for bone loss and cardiovascular disease? Arq Bras Endocrinol Metab. 54(2), 118-122.
- Fievez L, Michels M, Sweep FC, Hermus AR, et al. (2008). Vitamin D deficiency among HIV type 1-infected individuals in the Netherlands: Effects of antiretroviral therapy. AIDS Res Hum Retroviruses. 24(11), 1375-1382.
- Stephensen CB, Marquis GS, Kruzich LA, Douglas SD, et al. (2006). Vitamin D status in adolescentes and young adults with HIV infection. Am J Clin Nutr. 83(5), 1135-1141.
- Seminari E, Samaras K, Wand H, Law M, et al. (2005). Osteoprotegerin and bone turnover markers in heavily pretreated HIV-infected patients. HIV Med. 6(3), 145-150.
- Aparicio AMG, Fernandez SM, Gonzalez J, Arribas JR, et al. (2006). Abnormalities in the bone mineral metabolism in HIVinfected patients. Clin Rheumatol. 25(4), 537-539.
- Crutchley RD, Gathe J, Mayberry C, Trieu A, et al. (2012). Risk Factors for Vitamin D Deficiency in HIV-Infected Patients in the South Central United States. AIDS Res Hum Retroviruses. 28(5), 454-459.
- Kim JH, Gandhi V, Psevdos G, Espinoza F, et al. (2012). Evaluation of Vitamin D Levels Among HIV-Infected Patients in New York City. AIDS Res Hum Retroviruses. 28(3), 235-241.
- Shepherd L, Souberbielle JC, Bastard JP, Fellahi S, et al. (2014). Prognostic value of vitamin D level for all-cause mortality, and association with inflammatory markers, in HIV-infected persons. J Infect Dis. 210(2), 234-243.
- Haug C, Muller F, Aukrust P and Froland SS. (1994). Subnormal serum concentration of 1,25-vitamin D in human immunodeficiency virus infection: correlation with degree of immune deficiency and survival. J Infect Dis. 169(4), 889-893.
- Viard JP, Souberbielle JC, Kirk O, Knysz B, et al. (2011). Vitamin D and clinical disease progression in HIV infection: results from the EuroSIDA study. AIDS. 25(10), 1305-1315.
- Allavena C, Delpierre C, Cuzin L, Rey D, et al. (2012). High frequency of vitamin D deficiency in HIV-infected patients: effects of HIV-related factors and antiretroviral drugs. J. Antimicrob. Chemother. 67(9), 2222-2230.
- Bearden A, Abad C, Gangnon R, Sosman JM, et al. (2013). Cross-Sectional Study of Vitamin D Levels, Immunologic and Virologic Outcomes in HIV-Infected Adults. J Clin Endocrinol Metab. 98(4), 1726-1733.
- Pinzone MR, Di Rosa M, Celesia BM, Condorelli F, et al. (2013). LPS and HIV gp120 modulate monocyte/macrophage CYP27B1 and CYP24A1 expression leading to vitamin D consumption and hypovitaminosis D in HIV-infected individuals. Eur Rev Med Pharmacol Sci. 17(14), 1938-1950.
- Theodorou M, SersteT, Van Gossum M and Dewit S. (2013). Factors associated with vitamin D deficiency in a population of 2044 HIV-infected patients. Clin Nutr. 33(2), 274-279.
- Wohl DA, Orkin C, Doroana M, Pilotto JH, et al. (2014). Change in vitamin D levels and risck of severe vitamin D deficiency over 48 weeks among HIV-1 infected, treatment-na- eve adults receiving rilpivirina or efavirenz in a Phase III trial (ECHO). Antivir Ther. 19(2), 191-200.
- Avihingsanon A, Apompong T, Ramautarsing RA, Ubolyam S, et al. (2014). Decline in serum 25 hydroxyvitamin D levels in HIV-HBV-coinfected patients after long-term antiretroviral therapy. Antivir Ther. 19(1), 41-49.
- Ellfolk M, Norlin M, Gyllensten K and Wikvall K. (2009). Regulation of human vitamin D(3) 25-hydroxylases in dermal fibroblasts and prostate cancer LNCaP cells. Mol Pharmacol. 75(6), 1392-1399.
- Dao CN, Patel P, Overton ET, Rhame F, et al. (2011). Low Vitamin D among HIV-Infected Adults: Prevalence of and Risk Factors for Low Vitamin D Levels in a Cohort of HIV-Infected Adults and Comparison to Prevalence among Adults in the US General Population. Clin Infect Dis. 52(3), 396-405.
- Mehta S, Hunter DJ, Nugusi FM, Spiegelman D, et al. (2009). Perinatal Outcomes, Including Mother-to-Child Transmission of HIV, and Child Mortality and Their Association with Maternal Vitamin D Status in Tanzania. J Infect Dis. 2009; 200(7), 1022-1030.
- Torres C, Nieto G, Vergara S, Carrero AJ, et al. (2008). Vitamin D Receptor Gene Haplotypes and Susceptibility to HIV-1 Infection in Injection Drug Users. J Infect Dis. 197(3), 405-410.
- Torres C, Sanchez-de-la-Torre M, Garcia-Moruja C, Carrero AJ, et al. (2010). Immunophenotype of Vitamin D Receptor Polymorphism Associated to Risk of HIV-1 Infection and Rate of Disease Progression. Current HIV Research. 8(6), 487-492.
- Mehta S, Mugusi FM, Spiegelman D, Villamor E, et al. (2011). Vitamin D Status and its Association with Morbidity Including Wasting and Opportunistic Illnesses in HIV-Infected Women in Tanzania. AIDS Patient Care and STDs. 25(10), 579- 585.
- French AL, Adeyemi OM, Agniel DM, Evans CT, et al. (2011). The Association of HIV Status with Bacterial Vaginosis and Vitamin D in the United States.Journal of Women's Health. J Womens Health (Larchmt). 20(10), 1497-1503.
- Nansera D, Graziano FM, Friedman DJ, Bobbs MK, et al. (2011). Vitamin D and calcium levels in Ugandan adults with human immunodeficiency virus and tuberculosis. Int J Tuberc Lung Dis. 15(11), 1522-1527.
- Triant VA, Brown TT, Lee H and Grinspoon SK. (2008). Fracture Prevalence among Human Immunodeficiency Virus (HIV)-Infected Versus Non-HIV-Infected Patients in a Large U.S. Healthcare System. J Clin Endo Metab. 93(9), 3499-3504.
- Jones S, Restrepo D, Kasowitz A, Korenstein D, et al. (2008). Risk factors for decreased bone density and effects of HIV on bone in the elderly. Osteoporosis Inter. 19(7), 913-918.
- Cazanave C, Dupon M, Lavignolle-Aurillac V, Barthe N, et al. (2008). Reduced bone mineral density in HIV-infected patients: prevalence and associated factors. AIDS. 22(3), 395- 402.
- McComsey GA , Kendall MA, Tebas P, Swindells S, et al. (2007). Alendronate with calcium and vitamin D supplementation is safe and effective for the treatment of decreased bone mineral density in HIV. AIDS. 21(18), 2473-2482.
- McComsey GA, Tebas P, Shane E, Yin MT, et al. (2010). Bone Disease in HIV Infection: A Practical Review and Recommendations for HIV Care Providers. Clin Infect Dis. 51(8), 937-946.