Previous Issues Volume 2, Issue 1 - 2017
Insights into Dermatological Applications and Cutaneous Toxicities of Bleomycin
Ahmed M. Kabel1,2*, Fatma M. Moharm3,4
1Department of Pharmacology, Faculty of Medicine, Tanta University, Tanta, Egypt.
2Department of Clinical Pharmacy, College of Pharmacy, Taif University, Taif, Saudi Arabia.
3Al Maha Care Clinics, Mecca, Saudi Arabia.
4Dermatology Specialist, Ministry of Health, Egypt.
Corresponding Author: Ahmed M. Kabel, Department of Pharmacology, Faculty of Medicine, Tanta University, Tanta, Egypt, Tel: +966564833526; E-Mail: [email protected]
Received Date: 07 Dec 2016
Accepted Date: 23 Feb 2017
Published Date: 24 Feb 2017
Copyright © 2017 Kabel AM
Citation: Kabel AM and Moharm FM. (2017). Insights into Dermatological Applications and Cutaneous Toxicities of Bleomycin. Mathews J Dermatol. 2(1): 010
Bleomycin is a chemotherapeutic agent used for treatment of many types of tumors including lymphomas, testicular cancer and breast cancer. Its cytotoxic effect may be attributed to its interaction with O2 and Fe2+ leading to scission of DNA. Its systemic application may be associated with serious pulmonary fibrosis. It may be used by intralesional injection for management of keloids, hypertrophic scars, cutaneous warts, hemangiomas, cutaneous malignancies, cutaneous leishmaniasis and condyloma accuminata. However, its use may lead to significant dermatologic toxicities including scratch dermatitis, Raynaud's phenomenon, hyperpigmentation, fibrosis, gangrene, neutrophilic eccrine hidradenitis, alopecia areata, oedema and nail changes.
Keywords
Bleomycin; Dermatology; Applications; Toxicity; Skin
INTRODUCTION
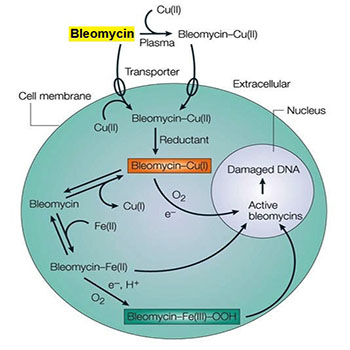
The bleomycins are DNA-cleaving antibiotics that were discovered as fermentation products of Streptococcus verticillus. The drug currently employed clinically is a mixture of the two copper-chelating peptides, bleomycins A2 and B2 [1]. Bleomycins have attracted interest because of their significant antitumor activity against squamous carcinoma of the cervix, lymphomas and testicular tumors. They are minimally myelo- and immunosuppressive but cause unusual cutaneous side effects and pulmonary fibrosis [2]. Because their toxicities do not overlap with those of other drugs and because of their unique mechanism of action, bleomycins maintain an important role in combination chemotherapy [3]. Although bleomycin has a number of interesting biochemical properties, its cytotoxic action results from their ability to cause oxidative damage to the deoxyribose of thymidylate and other nucleotides leading to single- and double-stranded breaks in DNA [4]. Studies in vitro indicate that bleomycin causes accumulation of cells in the G2 phase of the cell cycle and many of these cells display chromosomal aberrations including chromatid breaks, gaps and fragments, as well as translocations [5]. Bleomycin causes scission of DNA by interacting with O2 and Fe2+ (Figure 1). In the presence of O2 and a reducing agent, such as dithiothreitol, the metal-drug complex becomes activated and functions as a ferrous oxidase, transferring electrons from Fe2+ to molecular oxygen to produce activated species of oxygen [6]. Metallobleomycin complexes can be activated by reaction with the flavin enzyme, NADPH-cytochrome P450 reductase. Bleomycin binds to DNA through its amino-terminal peptide and the activated complex generates free radicals that are responsible for scission of the deoxyribose backbone of the DNA chain [7]. Bleomycin is degraded by a specific hydrolase found in various normal tissues. However, hydrolase activity is low in skin and lung, perhaps contributing to the toxicity at these sites [8].
Figure 1: Mechanism of action of bleomycin
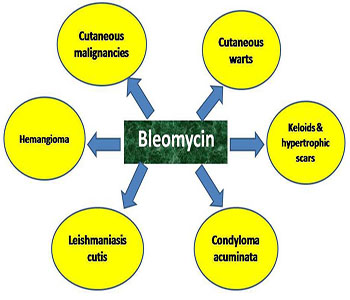
CLINICAL APPLICATIONS OF BLEOMYCIN IN DERMATOLOGY
Keloids and hypertrophic scars
Keloids and hypertrophic scars are abnormal wound responses to trauma, inflammation, surgery or burns in predisposed individuals. Both conditions are characterized by excessive deposition of collagen and glycoprotein [9]. Administration of bleomycin by intradermal injections or the multipuncture method has been shown to be effective against keloid and hypertrophic scars [10]. Thirteen patients were treated with bleomycin drip via multiple needle punctures, at a maximum dose of 6 mL. Complete flattening was noted in six cases, highly significant flattening in six cases, and significant flattening in one case [11]. Saray and Gulec [12] used dermojet injection of bleomycin to treat 14 patients with keloids and hypertrophic scars that had not responded to intralesional injection of triamcinolone acetonide. Multiple jet injections of 0.1 ml of bleomycin (1.5 IU/ml) were given to each lesion. The number of sessions used to successfully treat the lesions ranged from two to six. 73% of the lesions showed complete flattening, 7% showed highly significant flattening, 13% showed significant flattening and 7% showed moderate flattening. Aggarwal et al. [13] used bleomycin by multiple superficial puncture technique to treat fifty patients with keloids and hypertrophic scars. Three applications were given at interval of fifteen days followed by a fourth and final application two months after the last application. This resulted in complete flattening in 22 patients (44%), significant flattening in 11 patients (22%), adequate flattening in 7 patients (14%) and no flattening in 10 patients (20%). Naeini et al. [14] treated 45 patients with keloids and hypertrophic scars. They were divided into two groups; group A was treated by bleomycin tattoo and group B was treated with cryotherapy combined with intralesional triamcinolone injection. They were given four therapeutic sessions at one month interval. Therapeutic response in lesions less than 100mm2 was higher than 88% in both groups but in larger lesions, the response to bleomycin was significantly better than cryotherapy combined with intralesional triamcinolone injection.
Cutaneous warts
Because bleomycin inhibits DNA synthesis also in viruses, it is used for the treatment of warts resistant to other conventional treatments. Although intralesional injections are widely and very successfully employed, other methods have also been tried, e.g. instillation or direct application of bleomycin following pricking with a needle [15]. Other possible mechanisms underlying the efficacy of bleomycin are the induction of tumour necrosis factor (TNF), and the appearance of apoptotic cells in warts [16]. It causes acute tissue necrosis that may stimulate an immune response. Numerous reports have been published on the use of intralesional bleomycin for the treatment of warts with cure rates ranging from 14% to 99% [17].
Hemangioma
The exact mechanism of bleomycin action in hemangioma has not been clearly elucidated. Bleomycin has been shown to act as a sclerosing agent in lymphatic malformations. Direct free radical damage may also play a role. Bleomycin, also an antineoplastic agent, may induce apoptosis in rapidly proliferating, immature cells [18]. Because the majority of hemangioma patients are children and ulceration of thin skin is possible, weak concentrations of bleomycin, diluted to approximately 0.2 to 0.4 mg/kg in normal saline solution was used. Bleomycin is commonly delivered using a multipuncture technique (3 to 4 punctures per lesion) [19]. Studies suggest a 2 to 4 week interval between serial injections. Significant responses have been obtained within 4 to 10 courses, although a change in lesion size and appearance is often detectable after the third injection [20].
Cutaneous malignancies
Bleomycin injection is indicated in squamous cell carcinoma (SCC) of the head and neck (including mouth, tongue, tonsil, nasopharynx, oropharynx, sinus, palate, lip, buccal mucosa, gingivae, epiglottis and larynx), skin, penis, cervix and vulva [21]. Fourteen patients with advanced SCC of the skin or lip were treated with 1 to 4 cycles of combination chemotherapy consisting of cisplatin, 5-FU and bleomycin. Complete remission was seen for at least 1 month in 4 patients and partial remission for more than 1month in 7 patients [11]. Also, bleomycin has been used as an alternative for treatment of basal cell carcinoma (BCC). Benefits of bleomycin for BCC include ease of administration and time efficacy equal to conventional therapies [22].
Other applications of bleomycin in dermatology
There are two reports of intralesional bleomycin use in leishmaniasis cutis, both with a 100% cure rate [23]. Soyuer and Aktas [24] injected the patients with leishmaniasis cutis by 1.5 mL of 1% bleomycin solution and experienced swelling 72 hours after treatment. Administering bleomycin in cases of refractory condyloma acuminata is theoretically favorable because the drug successfully treats recalcitrant warts without significant side effects [23]. One study used 0.1mL of 1% bleomycin solution with a 25-gauge needle for injection of condyloma acuminatum. The total number of cycles was not reported, but the protocol resulted in a 70% cure rate [25].
Figure 2: Dermatological applications of bleomycin.
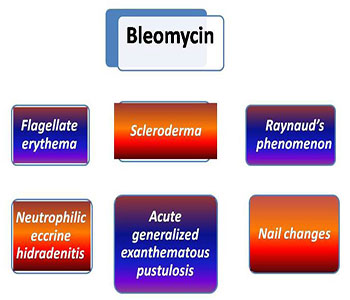
CUTANEOUS TOXICITY OF INTRALESIONAL BLEOMYCIN
Cutaneous side-effects including "flagellate" erythema (scratch dermatitis), hyperpigmentation, Raynaud's phenomenon, gangrene, fibrosis, neutrophilic eccrine hidradenitis (NEH), alopecia, oedema, nail changes and other miscellaneous reactions have been documented with the clinical use of bleomycin [26]. It was reported that cutaneous toxicity usually occurs at total doses of between 200 and 300 U, and pulmonary fibrosis occurs at doses more than 400 U. However, there is much variation in the data reported [11].
Flagellate erythema
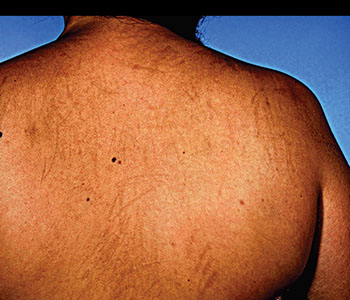
Flagellate erythema was originally described as a characteristic side-effect of bleomycin, which presents as erythematoviolaceous linear streaks on the trunk and/or shoulders. Additionally, an unusual case of reticular and linear hyperpigmentation was reported [27]. The incidence rate was shown to be 8-66% in patients treated with bleomycin. The reaction is considered to be dose dependent and normally occurs at total doses > 100 U and very frequently at doses > 200 U. By contrast, it can also occur after small doses as low as 14-15 U. The duration between administration of bleomycin and the onset of the clinical symptoms varies from 12-24 hours to 6 months [28]. Some patients complain of generalized pruritus, which eventually develops into flagellate erythema, just after receiving bleomycin. In some cases flagellate erythema is supposed to be caused by microtraumata such as scratching, which causes the drug to leak out of the blood vessels. Triggering factors other than scratching should also be considered [11].
Figure 3: Cutaneous toxicities of intralesional bleomycin.
Figure 4: Flagellate erythema due to bleomycin [29].
Raynaud"s phenomenon and gangrene
Several cases of Raynaud"s phenomenon have been reported during combination chemotherapy involving bleomycin and also following local intralesional injections of bleomycin for recalcitrant warts [30]. Raynaud's phenomenon is a common side-effect in human immunodeficiency virus-positive patients with Kaposi sarcoma who have been treated with bleomycin. In such patients, nailfold capillary microscopy shows a dramatic decrease in the number of capillaries. Raynaud's phenomenon can be induced independently of the method of administration or dosage of bleomycin, may be persistent or temporary, and is limited to the fingers that have been injected [31]. It has been suggested that bleomycin causes vascular endothelial cell injury, especially in small blood vessels, which may account for the development of Raynaud"s phenomenon [32]. Raynaud"s phenomenon leading to acral gangrene has been reported while gangrene without prior onset of Raynaud"s phenomenon has also been seen. In one case, histological examination revealed leucocytoclastic vasculitis, leading to ischaemic necrosis [33]. Accumulated evidence suggests that bleomycin enhances TNF-a synthesis, which in turn induces a procoagulant effect in blood vessels by increasing tissue factor and tissue plasminogen-activator inhibitor production, and by decreasing endothelial thrombomodulin and protein C activation. Thus, bleomycin-induced TNF-alpha may be involved in the development of Raynaud's phenomenon and gangrene [32].
Figure 5: Raynaud's phenomenon due to bleomycin [32].
Bleomycin-induced scleroderma
Systemic sclerodermatous changes have been reported to occur in patients with underlying cancer or haematological malignancies during or following systemic chemotherapy involving bleomycin. Histological examination showed dermal sclerosis [11]. Rats injected repeatedly with sublethal doses of bleomycin over a 58-week period developed severe dermal fibrosis, with structural abnormalities in the collagen fibres [34]. In mice, repeated local injection of bleomycin induced dermal sclerosis mimicking human scleroderma and has even been established as a mouse model for scleroderma. The possible mechanisms underlying fibrotic/sclerotic skin changes during bleomycin therapy are still under research [11].
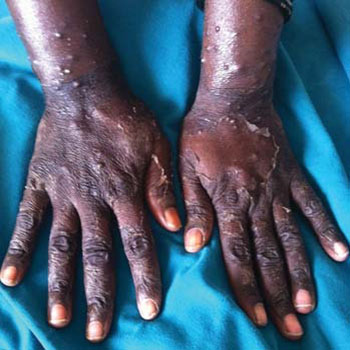
Neutrophilic eccrine hidradenitis (NEH)
Neutrophilic eccrine hidradenitis is an inflammatory dermatosis characterized clinically by erythematous plaques and nodules and histologically by the neutrophilic infiltrates of eccrine glands and the degeneration of eccrine cells (Figure 6). This condition is frequently associated with the combination of certain chemotherapeutic agents including bleomycin, cytarabine, doxorubicin, vincristine, cisplatin and dacarbazine [11]. There are various possible mechanisms including the effects of drug toxicity on sweat glands, or may be part of the neutrophilic dermatitis spectrum or may represent a paraneoplastic condition [35]. Histology of bleomycin-injected human skin also showed necrosis of the eccrine epithelium associated prominently with neutrophilic infiltrates, in a condition resembling NEH. The eccrine sweat gland apparatus may be susceptible to the toxic effects of bleomycin [36].
Figure 6: Neutrophilic eccrine hidradenitis [35].
Acute generalized exanthematous pustulosis
Acute generalized exanthematous pustulosis (AGEP) is characterized by an acute generalized erythema with a number of pustules and systemic symptoms such as high fever, most commonly after drug ingestion, especially of antibiotics [37]. A case of AGEP was reported in a patient treated with bleomycin, etoposide and cisplatin. The patient's condition worsened after a single instance of treatment using bleomycin, as confirmed by patch testing [38]. Additionally, there is an unusual report of a case of Stevens-Johnson syndrome resembling AGEP that was induced by peplomycin, a bleomycin analogue [39].
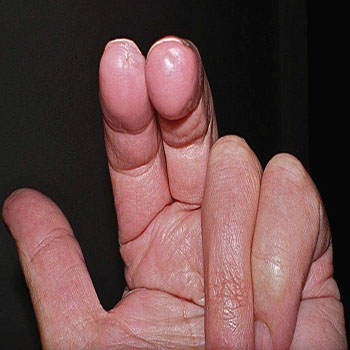
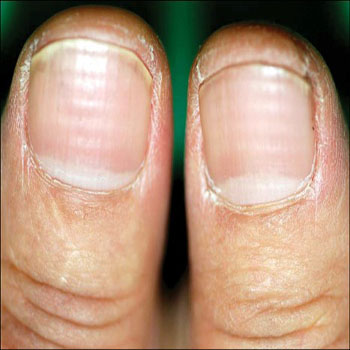
Bleomycin-induced nail changes
Bleomycin has been associated with nail pigmentation. The pigment is deposited in horizontal or vertical bands, which may be brown or blue and generally grow out with the nail. On the other hand, it was suggested that the pigmentation occurs in the nail bed [40]. Much drug-induced nail pigmentation is the result of increased melanin production by nail matrix melanocytes. The histology of this process has yet to be carefully examined. Nail dystrophy and nail loss were reported following intralesional bleomycin injections for periungual warts [11].
Figure 7: Bleomycin-induced nail changes [41].
CONCLUSION
Bleomycin has a crucial role in management of keloids, hypertrophic scars, warts, hemangiomas and cutaneous malignancies. However, its use may be limited by the possible occurrence of dermatologic toxicities including scratch dermatitis, Raynaud's phenomenon, hyperpigmentation, fibrosis, neutrophilic eccrine hidradenitis and nail changes.
REFERENCES
- Coughlin JM, Rudolf JD, Wendt-Pienkowski E, Ute G, et al. (2014). BlmB and TlmB Provide Resistance to the Bleomycin Family of Antitumor Antibiotics by N-Acetylating Metal-Free Bleomycin, Tallysomycin, Phleomycin, and Zorbamycin. Biochemistry. 53(44): 6901-6909.
- Ni S, Wang D, Qiu X, Pang L, et al. (2015). Bone marrow mesenchymal stem cells protect against bleomycin-induced pulmonary fibrosis in rat by activating Nrf2 signaling. International Journal of Clinical and Experimental Pathology. 8(7): 7752-7761.
- Patil N, Paulose RM, Udupa KS, Ramakrishna N, et al. (2016). Pulmonary Toxicity of Bleomycin-A Case Series from a Tertiary Care Center in Southern India. Journal of Clinical and Diagnostic Research: JCDR. 10(4): FR01-FR03.
- Chen JK and Murray V. (2016). The determination of the DNA sequence specificity of bleomycin-induced abasic sites. J Biol Inorg Chem. 21(3): 395-406.
- Cho YH, Lee JW, Woo HD, Lee S, et al. (2016). Protective Effect of Onion Extract on Bleomycin-Induced Cytotoxicity and Genotoxicity in Human Lymphocytes. Tchounwou PB, ed. International Journal of Environmental Research and Public Health. 13(2): 227.
- Pei H, Zheng Y, Kong R, Xia L, et al. (2016). Niche nanoparticle-based FRET assay for bleomycin detection via DNA scission. Biosens Bioelectron. 85: 76-82.
- Kabel AM, Omar MS and Elmaaboud MA. (2016). Amelioration of bleomycin-induced lung fibrosis in rats by valproic acid and butyrate: Role of nuclear factor kappa-B, proinflammatory cytokines and oxidative stress. International Immunopharmacology. 39: 335-342.
- Jona A, Miltenyi Z, Poliska S, Balint BL, et al. (2016). Effect of Bleomycin Hydrolase Gene Polymorphism on Late Pulmonary Complications of Treatment for Hodgkin Lymphoma. Morty RE, ed. PLoS ONE. 11(6): e0157651.
- Maghrabi IA and Kabel AM. (2014). Management of Keloids and Hypertrophic Scars: Role of Nutrition, Drugs, Cryotherapy and Phototherapy. Journal of Nutrition and Health. 2(2): 28-32.
- Kabel AM, Sabry HH, Sorour NE and Moharm FM. (2016). Comparative study between intralesional injection of bleomycin and 5-fluorouracil in the treatment of keloids and hypertrophic scars. Journal of Dermatology and Dermatologic Surgery. 20(1): 32-38.
- Yamamoto T. (2006). Bleomycin and the skin. Br J Dermatol. 155(5): 869-875.
- Saray Y and Gu lec AT. (2005). Treatment of keloids and hypertrophic scars with dermojet injections of bleomycin: a preliminary study. Int J Dermatol. 44(9): 777-784.
- Aggarwal H, Saxena A, Lubana PS, Mathur RK, et al. (2008). Treatment of keloids and hypertrophic scars using bleomycin. Cosm Dermatol. 7: 43-49.
- Naeini FF, Najafian J and Ahmadpour K. (2006). Bleomycin tattooing as a promising therapeutic modality in large keloids and hypertrophic scars. Dermatol Surg. 32: 1023- 1029.
- Saitta P, Krishnamurthy K and Brown LH. (2008). Bleomycin in dermatology: a review of intralesional applications. Dermatol Surg. 34(10): 1299-1313.
- Rabello FB, Souza CD and Junior JAF. (2014). Update on hypertrophic scar treatment. Clinics. 69(8): 565-573.
- Lee JY, Kim CW and Kim SS. (2015). Preliminary Study of Intralesional Bleomycin Injection for the Treatment of Genital Warts. Annals of Dermatology. 27(2): 239-241.
- Kasper M and Barth K. (2009). Bleomycin and its role in inducing apoptosis and senescence in lung cells-modulating effects of caveolin-1. Curr Cancer Drug Targets. 9(3): 341-353.
- Alghamdi KM and Khurram H. (2012). Successful treatment of plantar warts with very diluted bleomycin using a translesional multipuncture technique: pilot prospective study. J Cutan Med Surg. 16(4): 250-256.
- Muir T, Kirsten M, Fourie P, Dippenaar N, et al. (2004). Intralesional bleomycin injection (IBI) treatment for haemangiomas and congenital vascular malformations. Pediatr Surg Int. 19(12): 766-773.
- Tozon N, Pavlin D, Sersa G, Dolinsek T, et al. (2014). Electrochemotherapy with intravenous bleomycin injection: an observational study in superficial squamous cell carcinoma in cats. J Feline Med Surg. 16(4): 291-299.
- Meyer D and Gooding C. (2015). Intralesional Bleomycin as an Adjunct Therapeutic Modality in Eyelid and Extraocular Malignancies and Tumors. Middle East African Journal of Ophthalmology. 22(4): 410-414.
- Saitta P, Krishnamurthy K and Brown LH. (2008). Bleomycin in dermatology: a review of intralesional applications. Dermatol Surg. 34(10): 1299-313.
- Soyuer U and Aktas E. (1988). Intralesional bleomycin therapy for leishmaniasis cutis. Arch Dermatol. 124(10): 1571.
- Figueroa S and Gennaro AR. (1980). Intralesional bleomycin injection in treatment of condyloma acuminatum. Dis Colon Rectum. 23(8): 550-551.
- Trisliana Perdanasari A, Lazzeri D and Su W. (2014). Recent Developments in the Use of Intralesional Injections Keloid Treatment. Archives of Plastic Surgery. 41(6): 620-629.
- Grynszpan R, Niemeyer-Corbellini JP, Lopes MSS and Ramos-e-Silva M. (2013). Bleomycin-induced flagellate dermatitis. BMJ Case Reports. 2013: bcr2013-009764.
- VonHilsheimer GE and Norton SA. (2002). Delayed bleomycin-induced hyperpigmentation and pressure on the skin. J Am Acad Dermatol. 46(4): 642-643.
- Boussios S, Moschetta M, McLachlan J, Banerjee S. Bleomycin-Induced Flagellate Erythema in a Patient Diagnosed with Ovarian Yolk Sac Tumor. Case Reports in Oncological Medicine 2015; 2015: 574708.
- Vanhooteghem O, Richert B and de la Brassinne M. (2001). Raynaud phenomenon after treatment of verruca vulgaris of the sole with intralesional injection of bleomycin. Pediatr Dermatol. 18(3): 249-251.
- Lee H-Y, Lim K-H, Ryu Y and Song S-Y. (2014). Bleomycininduced flagellate erythema: A case report and review of the literature. Oncology Letters. 8(2): 933-935.
- McGrath SE, Webb A and Walker-Bone K. (2013). Bleomycin-induced Raynaud's phenomenon after single-dose exposure: risk factors and treatment with intravenous iloprost infusion. J Clin Oncol. 31(4): e51-2.
- Atas E1, Korkmazer N, Artik HA, Babacan O, et al. (2015). Raynaud's phenomenon in a child with medulloblastoma as a late effect of chemotherapy. J Cancer Res Ther. 11(3): 666.
- Mountz JD, Downs Minor MB, Turner R, Thomas MB, et al. (1983). Bleomycin-induced cutaneous toxicity in the rat: analysis of histopathology and ultrastructure compared with progressive systemic sclerosis (scleroderma). Br J Dermatol. 108(6): 679-686.
- Bhanu P, Santosh KV, Gondi S, Manjunath KG, et al. (2013). Neutrophilic eccrine hidradenitis: A new culpritcarbamazepine. Ind J Pharmacol. 45(1): 91-92.
- Changal K, Raina H, Changal Q and Raina M. (2014). Bleomycin-induced Flagellate Erythema: A Rare and Unique Drug Rash. West Ind Med J. 63(7): 807-809.
- Feldmeyer L, Heidemeyer K and Yawalkar N. (2016). Acute Generalized Exanthematous Pustulosis: Pathogenesis, Genetic Background, Clinical Variants and Therapy. Int J Mol Sci. 17(8): E1214.
- Altaykan A, Boztepe G, Erkin G, Ozkaya O, et al. (2004). Acute generalized exanthematous pustulosis induced by bleomycin and confirmed by patch testing. J Dermatol Treatment; 15(4): 231-234.
- Umebayashi Y, Enomoto H and Ogasawara M. (2004). Drug eruption due to peplomycin: An unusual form of Stevens-Johnson syndrome with pustules. J Dermatol. 31(10): 802-805.
- Kumar S, Dixit R, Karmakar S and Paul S. (2010). Unusual nail pigmentation following cyclophosphamide-containing chemotherapy regimen. Ind J Pharmacol. 42(4): 243- 244.
- Kim IS, Lee JW, Park KY, Li K, Seo SJ, Hong CK. Nail Change after Chemotherapy: Simultaneous Development of Beau's Lines and Mees' Lines. Ann Dermatol 2012; 24(2): 238-239.