Previous Issues Volume 1, Issue 3 - 2016
Aggressive Opportunistic Paracoccidioidomycosis in an AIDS Patient
Pedro Afonso Nogueira Moises Cardoso1, Paulo Naves Aguiar1, Heloisa Loureiro de Sa Neves Motta1, Erik Friedrich Alex de Souza1, Daniel de Alvarenga Salem Sugui2, Guilherme Almeida Rosa da Silva2, Rogerio Neves Motta2, Marcelo Costa Velho Mendes de Azevedo2, Walter de Araujo Eyer Silva2, Jorge Francisco da Cunha Pinto2, Fernando Raphael de Almeida Ferry2
1Medical Student at Universidade Federal do Estado do Rio de Janeiro (UNIRIO), Rio de Janeiro, Brazil.
2Professor from Universidade Federal do Estado do Rio de Janeiro (UNIRIO), Rio de Janeiro, Brazil.
Corresponding Author: Guilherme Almeida Rosa da Silva, Mariz e Barros 775, Tijuca-Rio de janeiro-Brazil, Tel: +55 21 22544759; E-Mail: [email protected]
Received Date: 15 Aug 2016 Accepted Date: 23 Aug 2016 Published Date: 25 Aug 2016
Copyright © 2016 da Silva GAR
Citation: da Silva GAR, Motta RN, Cardoso PANM, Aguiar PN, et al. (2016). Aggressive Opportunistic Paracoccidioidomycosis in an AIDS Patient. Mathews J Case Rep 1(3): 015.
ABSTRACT
Introduction: Paracoccidioidomycosis is a systemic mycosis transmitted by inhalation of the conidia form of Paracoccidiodes brasiliensis fungus. The disease is presented as acute, subacute or chronic forms, according to the cellular immune response of the host, and can be developed from a primary pulmonary focus or the reactivation of an existing focus, years after exposure. Case report: Male, age 41, carpenter, HIV positive with CD4+ lymphocyte count of 66 cells / ml, admitted at the Hospital Universitario Gaffree e Guinle (HUGG)- Brazil. He presented with fever, enlarged lymph nodes, diarrhea, and pancytopenia. Lymph node biopsy diagnosed as paracoccidioidomycosis. A Case advanced with severe thrombocytopenia, pulmonary bleeding and death, despite antifungal therapy instituted. Discussion: Paracoccidioidomycosis in patients with AIDS manifests itself with clinical and epidemiological patterns different from non-AIDS patients, and is more common in patients with CD4+ lymphocyte cell count less than 200 cells / ml. The best course of treatment and duration of the maintenance phase are yet to be determined by further studies.
INTRODUCTION
Paracoccidioidomycosis (PCM) or Lutz-Almeida-Splendore's disease is a granulomatous systemic chronic mycosis, with insidious progression, caused by the thermo-dimorphic Paracoccidiodes brasiliensis fungus. The most common form of infection occurs through inhalation of conidia present in the soil, usually in patients with epidemiological history of living in rural areas or earthmoving activity. These conidia produce the yeast form that parasite host tissues. Lungs and upper airway are the first segments to be affected, later following hematogenic or lymphatic dissemination [1]. PCM is the systemic mycosis with the highest mortality rate in Brazil. Considering the average mortality rate in the course of the disease, and the number of deaths / year attached to PCM, it is estimated an incidence of 3360 new cases / year of PCM in Brazil, around 1-3 cases per 100 000 inhabitants [1-3]. PCM can be expressed in acute, subacute and chronic forms. The acute and subacute forms, also called juvenile forms, correspond to 3 - 5% of the total incidence, more usual in children and teenagers. This clinical form has uniform distribution between genres with a slight predominance in young male adults. In this clinical presentation the main attributes are: lymphadenopathy, hepatosplenomegaly, unspecific digestive symptoms, joint involvement, and skin lesions. Anemia, fever, and weight loss, with fast deterioration of the general status, can also be found. Pulmonary involvement in juvenile forms is rare [1,4]. The unifocal-multifocal chronic adult form is more usual in males, and is responsible for over 90% of PCM cases. It is characterized by an indolent progression and a clinical condition that resembles to pulmonary tuberculosis, with weakness, weight loss, fever, sweat, coughing, and dyspnea symptoms. Reticulonodular infiltrate can be found in radiological lung exam: bat wing aspect, usually in the upper two-thirds of the lung, and distal hyper-transparency in the bases of the lung (unifocal form). When the disease expands to extra pulmonary sites, as the oral mucosa, adrenals, central nervous system, among others, it is classified as disseminated or multifocal [5]. According to these previous information, the main differential diagnosis of PCM is tuberculosis (TB), because of its clinical and radiological resemblance, so the diagnosis is based on microbiological criteria, established as isolation of fungus or mycobacteria. It is important to emphasize that the same individual can have both infections, this association occurring in 5.5 to 19% of cases, making the diagnosis even harder. Other differential diagnoses that should be considered are: sarcoidosis, histoplasmosis, idiopathic diffuse interstitial pneumonitis, chronic silicosis, coccidioidomycosis, chromoblastomycosis, cutaneous and visceral leishmaniasis, leprosy, skin, and laryngeal cancer [5]. The patient with Acquired Immunodeficiency Syndrome (AIDS) is more susceptible to the progression of acute PCM, reactivation of a chronic infection, and progression to disseminated forms, many times with unusual presentations. An Oswaldo Cruz Institute research, developed from 1996 to 2006, has shown that PCM, despite being the systemic mycosis with highest mortality rate in Brazil, has lower mortality when associated with AIDS. In this case, other causes are more prevalent: cryptococcosis, candidiasis, histoplasmosis, and aspergillosis, respectively. AIDS changes the appearance of systemic mycoses infection and may overlap other opportunistic infections, like TB. Therefore, it is possible that the PCM has to be underdiagnosed, especially when death occurs, in which autopsies are rarely performed [2].
CASE REPORT
Male, 41 years old, black, carpenter, born in Rio de Janeiro. HIV / AIDS diagnosed two years before. Presently in the irregular use of antiretroviral therapy (ARV) from diagnosis with: lamivudine (3TC) 150mg PO 2x / day, tenofovir (TDF) 300mg PO 1x / day, and efavirenz (EFV) 600mg PO 1x / day. Patient was transferred to the University Hospital Gaffree and Guinle (HUGG) with lymphadenopathy initiated five months before and abandonment of ARV. He was previously investigated for ganglionic tuberculosis and was in use of 4 four-inone tuberculosis tablets PO daily (rifampicin 150mg, isoniazid 75mg, pyrazinamide 400mg, and ethambutol 275mg - RIPE). Around seven days before admission, blood test showed CD4+ lymphocyte (LT - CD4+) count of 66 cells / ml and plasma HIV viral count of 135,519 copies / ml. Presented in good general condition, with pallor ++/4, tachycardia (124 beats per minute), tachypnea (23 incursions per minute), hypotensive (90x50 mmHg), febrile (38.5°C), with cervical, axillary, and inguinal lymphadenopathy, without jaundice or cyanosis. He complained of fever on evenings, unquantified weight loss, and low diarrhea in the last two months. Pulmonary exam with vesicular murmur decreased in right lung base, without significant alterations in the cardiovascular or abdominal examination. He presented a pattern of diffuse alveolar infiltrates on chest radiography. Prophylaxis began for Mycobacterium Avium Complex with clarithromycin 500mg IV 2x / day, and for toxoplasmosis and pneumocystis with trimethoprim/sulfamethoxazole (TMP/ SMX) 400 / 80mg PO 2x/day. Treatment for chronic diarrhea in immunosuppressed was initiated: metronidazole 500 mg PO 3x / day, albendazole 400 mg PO 1x / day and ciprofloxacin 400mg PO 2x / day, for five days (with TMP/SMX already installed). The use of RIPE was suspended due to drug-induced hepatitis. Admission exams were requested with blood count, biochemistry and urinalysis, shown in Table 1. The most significant data were: the presence of transaminases raising more than five times the reference value, assigned to RIPE, and anemia with severe leukopenia, which quickly evolved with thrombocytopenia to pancytopenia. There was a need for blood transfusions during hospitalization. The pancytopenia was attributed to advanced HIV infection, possibly combined with an opportunistic disease that seems to involve the reticuloendothelial system preferably.
Table 1: Most relevant Full Blood Count and Serum Biochemistry during admission.
| Day | 29/12 | 5/01 | 11/01 | 15/01 | 23/01 | 29/01 |
| Erythrocytes | 2,34 | 2,61 | 2,02 | 2,0 | 1,7 | 2,32 |
|---|---|---|---|---|---|---|
| Hemoglobin | 6,2 | 6,8 | 5,3 | 5,5 | 4,9 | 6,1 |
| Platelets | 145000 | 36000 | 16000 | 6000 | 2000 | 42000 |
| Leukocytes | 2000 | 1050 | 680 | 1790 | 15020 | 12150 |
| Band Cells | 2% | - | - | 6% | 13% | 13% |
| Neutrophils | 60% | - | - | 86% | 62% | 66% |
| Eosinophils | 0 | - | - | 0 | 8% | 1% |
| Lymphocytes | 28% | - | - | 4% | 15% | 13% |
| Monocytes | 8% | - | - | 4% | 10% | 5% |
| Sodium | - | 127 | 140 | 144 | 141 | 141 |
| Potassium | - | 4.06 | 3,7 | 4,2 | 4,7 | 3,46 |
| Chlorine | - | 95 | 107 | 106 | 103 | 105 |
| Serum Urea | - | 37 | 77 | 37 | 132 | 46 |
| Creatinine | - | 0,84 | 0.96 | 0,68 | 0,95 | 0,62 |
| AST | - | 235 | - | 49 | - | 47 |
| ALT | - | 48 | - | 127 | - | 19 |
| GGT | - | 491 | - | - | - | - |
| ALP | - | 1260 | - | - | - | - |
| Amylase | - | 153 | - | - | - | - |
| Lipase | - | 82 | - | - | - | - |
| LDH | - | 3889 | - | - | - | 1062 |
| CRP | - | - | - | 145,7 | 49 | 162 |
AST: Aspartate aminotransferase; ALT: Alanine aminotransferase; GGT: Gamma-glutamyl transpeptidase; LDH: Lactate Dehydrogenase; CRP:CReactive Protein; ALP: Alkaline phosphatase.
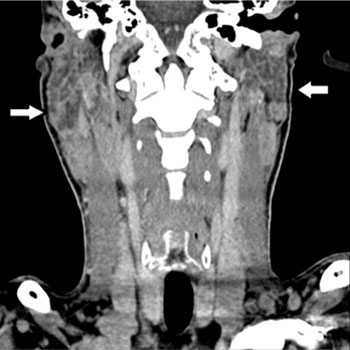
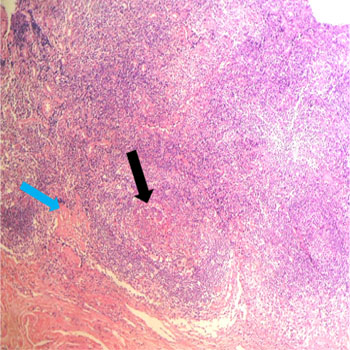
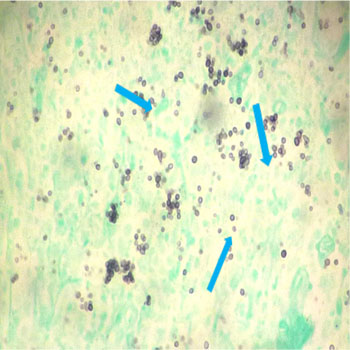
Due to the presence of generalized lymphadenopathy, computed tomography (CT) was requested for the neck, chest, abdomen, and pelvis. CT demonstrated: lymphadenopathy in bilateral neck chains (Figure 1), atelectasis in the right lung base, bilateral axillary lymphadenopathy, hepatosplenomegaly, and cystic images in the right kidney. Left inguinal lymph node open biopsy was performed. The case evolved with rapid clinical deterioration, with patient presenting nausea, vomiting, diarrhea, and inappetence. The result of the left inguinal lymph node biopsy revealed necrotizing granulomatous disease (Figure 2) with mold spores detected by staining of silver impregnation, compatible of paracoccidioidomycosis (Figure 3). Itraconazole was started in PO dose of 400mg / day.
Figure 1: Neck Computer Tomography Scan.
Lymphadenopathy in bilateral neck chains (white arrows).
Figures 2: Lymph node biopsy with Hematoxylin and Eosin Stain.
Hematoxylin and Eosin stain, x 100. Fibrosis (black arrow) and chronic granulomatous inflammation can be seen (blue arrow).
Figures 3: Lymph node biopsy with GMS Stain.
Grocott-Gomori's methenamine silver stain (GMS), 200 x. Spherical yeast-like cells with budding daughter cells, compatible with paracoccidiodomycosis can be observed.
Despite treatment, the patient maintained daily fever peaks and poor dietary compliance, with deteriorating dyspnea, dyslalia and visual alterations. The treatment regimen was substituted with amphotericin B deoxycolate in an initial dose of 0.3mg / kg / day IV diluted in 500 ml of dextrose, infused at 4 hours for cumulative doses, increasing by 5 mg / day every 3-5 days, in the absence of side effects such as hypokalemia and renal failure. One week after initiated treatment with amphotericin, the patient evolved to respiratory failure that made necessary intubation and mechanical ventilation with transfer to the intensive care unit (ICU). The Patient complicated with the pneumonia associated with mechanical ventilation, being treated with vancomycin 1 g IV 12 / 12h, imipenem 500mg IV 6/6h and polymyxin B 1,000,000 IU 12/12h. After tracheostomy for prolonged intubation, about three weeks later, the patient developed severe thrombocytopenia, massive pulmonary hemorrhage, and death.
DISCUSSION
The coinfection of PCM in AIDS patients is similar to severe manifestations of acute and subacute PCM, whose tropism for the reticuloendothelial system occurs, with the disease probable caused by reactivation of a latent focus of infection. In HIV-seronegative individuals, acute form is most commonly observed in children and young adults, consisting about 3 to 5% of all cases. However, in patients with AIDS, this is the most common form of the disease, regardless of age, and is more common in patients with LT - CD4+ cell counts below 200, like the case reported [7-10]. The symptoms most frequently exhibited by patients coinfected with HIV and PCM are: fever, present in up to 96% of patients; lymphadenopathy, up to 80% of patients; skin lesions, up to 65% of patients, and hepatomegaly in up to 64% cases. The immunosuppression is also associated with a more rapidly progressive disease installation. The most common radiological patterns are bilateral pulmonary reticulonodular infiltrates in 56.4%, and diffuse alveolar infiltrates in 25.6% of cases [10-13]. The PCM / AIDS coinfection also changes the epidemiology of PCM. The prevalence ratio between men and women falls from 15:1 to 3: 1, and there is a less frequent association with the epidemiological history of activity in rural areas10. In one case report, the most likely source of infection was the sharing of needles among intravenous drug users [16]. The PCM association to the abuse of alcohol and smoking was kept unchanged in AIDS patients, compared to those without AIDS, showing that these risk factors should always be considered [8,10,14,15]. In a retrospective series of eight cases of acute PCM, Paracoccidioides brasiliensis was responsible for bone marrow necrosis (BMN) seen at autopsy. BMN is a condition that affects the myeloid parenchyma and medullary stromal, affecting large areas of hematopoietic tissue, with cytopenia contributing to the death of some patients, similar as we have reported in our case [16]. Because of the clinical and epidemiological peculiarities of PCM in AIDS patients, Morejon et al. conclude that paracoccidioidomycosis should be classified as opportunistic and be included in the AIDS-defining illness list. The authors also suggest that, in regions most affected by the AIDS epidemic, patients with a disseminated form of the disease urge active investigation for HIV infection [10]. It is estimated that the PCM coexist with TB in AIDS patients between 5.5 and 19% of cases. The clinical and radiological similarities between these infections are such that the definitive diagnosis can only be obtained by identification of the infectious agent. In our case, samples of biopsies sent to pathology were negative for Ziehl-Neelsen, and there were no acid and alcohol fast bacilli in the bronchoalveolar lavage fluid, sputum test, blood or urine culture. However, the treatment, even though irregular, used for ganglionic tuberculosis (RIPE scheme about 2 months, followed by two months of RI before diagnostic investigation) makes it impossible to assert the non-existence of the two infections at some point. Also, the time of the sub-acute phase prior to the first hospitalization was imprecise [5]. Taking as a reference the study of case-control conducted by Morejon & Machado (2009) which followed 59 cases with PCM / AIDS association, most of the findings of this case are expected in patients with association between the two diseases, namely: fever, lymphadenopathy, pulmonary involvement, dyspnea, and diffuse alveolar interstitial infiltrate in simple radiograph. The study by Morejon et al. makes no mention of pancytopenia or neurological symptoms, however mentions central nervous system lesions in two patients (3.8% of cases) and bone lesions in 3 (5.7%), conditions that could attend with this kind of symptom. The disease pattern in our case was also consistent with one found on a series of 12 PCM / AIDS cases published by Paniago et al. (2005), although in her study the prevalence of symptoms and radiological findings were slightly different [10,17]. Treatment of PCM with antifungal agents in AIDS patients showed good results, with a significant reduction in mortality, in the study of 59 cases of coinfection treated at the University Hospital of Ribeir�o Preto Medical School. The most prescribed treatment for coinfected patients in this study was amphotericin B deoxycholate IV, with a cumulative dose ranging from 290 - 4.160mg, with subsequent consolidation therapy with other antifungal agents (fluconazole, ketoconazole, itraconazole or sulfamethoxazole-trimethoprim). Data from the study showed that the remission rates in coinfected patients were similar with the use of: sulfamethoxazole-trimethoprim 800 / 160mg 3 - 4x / day IV to clinical improvement, followed by 800 / 160mg PO 2x / day for up to 72 months, or fluconazole 400-600mg / day IV in the induction phase followed by 200 - 400mg / day PO in the consolidation and maintenance phase. In this study 83% of patients were taking antiretroviral therapy, and the authors attribute the reduction in mortality to early diagnosis and initiation of antifungal therapy. This study also states that the dose of sulfamethoxazole-trimethoprim used for prophylaxis for pneumocystis (400 / 80mg 2 times a day) does not reduce odds of the patient developing PCM as an opportunistic disease [10,17]. The antifungal therapy in our case, was initiated with itraconazole at a dose 400 mg / day PO, later being modified because of clinical deterioration to amphotericin B IV dose 0.3mg / kg / day (diluted in 500 ml of glucose solution, infused in 4 hours for cumulative dose) and increasing by 5 mg / day every 3 to 5 days, in the absence of side effects such as hypokalemia and renal insufficiency. The PCM consensus published in 2006 states that in hospitalized patients, antifungal medication should start intravenously, either with amphotericin B at a dose of 1 mg / kg / day or sulfamethoxazole-trimethoprim 800 / 160mg each 8 hours. Oral itraconazole is recommended as medication in mild to moderate disease, and did not fit the condition of our patient. In a study by Morejon & Martinez all patients began intravenous treatment, differing from the consensus, with the inclusion of intravenous therapy with fluconazole 400 - 600mg / day during the first days, as alternatives to amphotericin B and trimethoprim-sulfamethoxazole in the induction phase [1, 10]. Due to lack of data on coinfection AIDS/PCM, there is no recommendation based on the scientific level of evidence about the exact duration of antifungal therapy in these patients. In practice, it has been recommended according to paracoccidioidomycosis consensus, like another systemic mycosis occurrence in AIDS, the antifungal be maintained indefinitely until the levels of LT - CD4+ rise above 200 cells / mm3 for at least three to six months. Prospective studies are needed to validate this approach [1].
REFERENCES
- Shikanai-Yasuda MA, Telles-Filho FQ, Mendes RP, Colombo AL, et al. (2006). Consenso em paracoccidioidomicose. Revista da Sociedade Brasileira de Medicina Tropical. 39(3), 297-310.
- Prado M, Silva MB, Laurenti R, Travassos LR, et al. (2009). Mortality due to systemic mycoses as a primary cause of death or in association with AIDS in Brazil: a review from 1996 to 2006. Memorias do Instituto Oswaldo Cruz. 104(3), 513-521.
- Martinez R. (2010). Paracoccidioidomycosis: the dimension of the problem of a neglected disease. Revista da Sociedade Brasileira de Medicina Tropical. 43(4), 480.
- Wanke B and Aide MA. (2009). Capitulo 6 - Paracoccidioidomicose. Jornal Brasileiro de Pneumologia. 35(12), 1245-1249.
- Junior RQ, Grangeia TAG, Massucio RAC, De Capitani EM, et al. (2007). Association between paracoccidioidomycosis and tuberculosis: reality and misdiagnosis. Jornal Brasileiro de Pneumologia. 33(3), 295-300.
- Hotez PJ, Bottazzi ME, Franco-Paredes C, Ault SK, et al. (2008). The neglected tropical diseases of Latin America and the Caribbean: a review of disease burden and distribution and a road map for control and elimination. PLoS Neglected Tropical Diseases. 2(9), e300.
- Marques SA. (2003). Paracoccidioidomicose: atualização epidemiológica, clínica e terapêutica. Anais Brasileiros de Dermatologia. 78(2), 135-146.
- Benard G and Duarte AJS. (2000). Paracoccidioidomycosis: a model for evaluation of the effects of Human Immunodeficiency Virus infection of the natural history of endemic tropical diseases. Clinical Infectious Diseases. 31(4), 1032-1039.
- De Almeida SM. (2005). Central nervous system paracoccidioidomycosis: an overview. Brazilian Journal of Infectious Diseases. 9(2), 126-133.
- Morejon KML, Machado AA and Martinez R. (2009). Paracoccidioidomycosis in patients infected with and not infected with human immunodeficiency virus: a case-control study. The American Journal of Tropical Medicine and Hygiene. 80(3), 359-366.
- Shikanai-Yasuda MA, Conceicao YM, Kono A, Rivitti E, et al. (2008). Neoplasia and paracoccidioidomycosis. Mycopathologia. 165(4-5), 303-312.
- Zavascki AP, Bienardt JC and Severo LC. (2004). Paracoccidioidomycosis in organ transplant recipient: case report. Rev Inst Med Trop Sao Paulo. 46(5), 279.
- Shikanai-Yasuda MA, Duarte MI, Nunes DF, et al. (1995). Paracoccidioidomycosis in a renal transplant recipient. J Med Vet Mycol 33(6), 411-414.
- Ramos-e-Silva M. (2004). Facial and oral aspects of some venereal and tropical diseases. Acta Dermatovenereol Croat. 12(3), 173-180.
- Caseiro MM, Etzel A, Soares MC, Costa SO, et al. (2005). Septicemia caused by Paracoccidioides brasiliensis as the cause of death of an AIDS patient from Santos, Sao Paulo state, Brazil-a nonendemic area. Rev Inst Med Trop Sao Paulo. 47(4), 209-211.
- Resende LS, Mendes RP, Bacchi MM, Marques SA, et al. (2009). Bone marrow necrosis related to paracoccidioidomycosis: the first eight cases identified at autopsy. Histopathology. 2009; 54(4), 486-489.
- Paniago AM, Freitas AC, Aguiar ESA, Aguiar JIA, et al. (2005). Paracoccidioidomycosis in patients with human immunodeficiency virus: areview of 12 cases observed in an endemic region of Brazil. J Infect. 51(3), 248-252.